India Pharmaceutical Packaging Market Set to Hit USD 11.21 Billion by 2034, Fuelled by Regulatory Overhaul and Biologics Surge
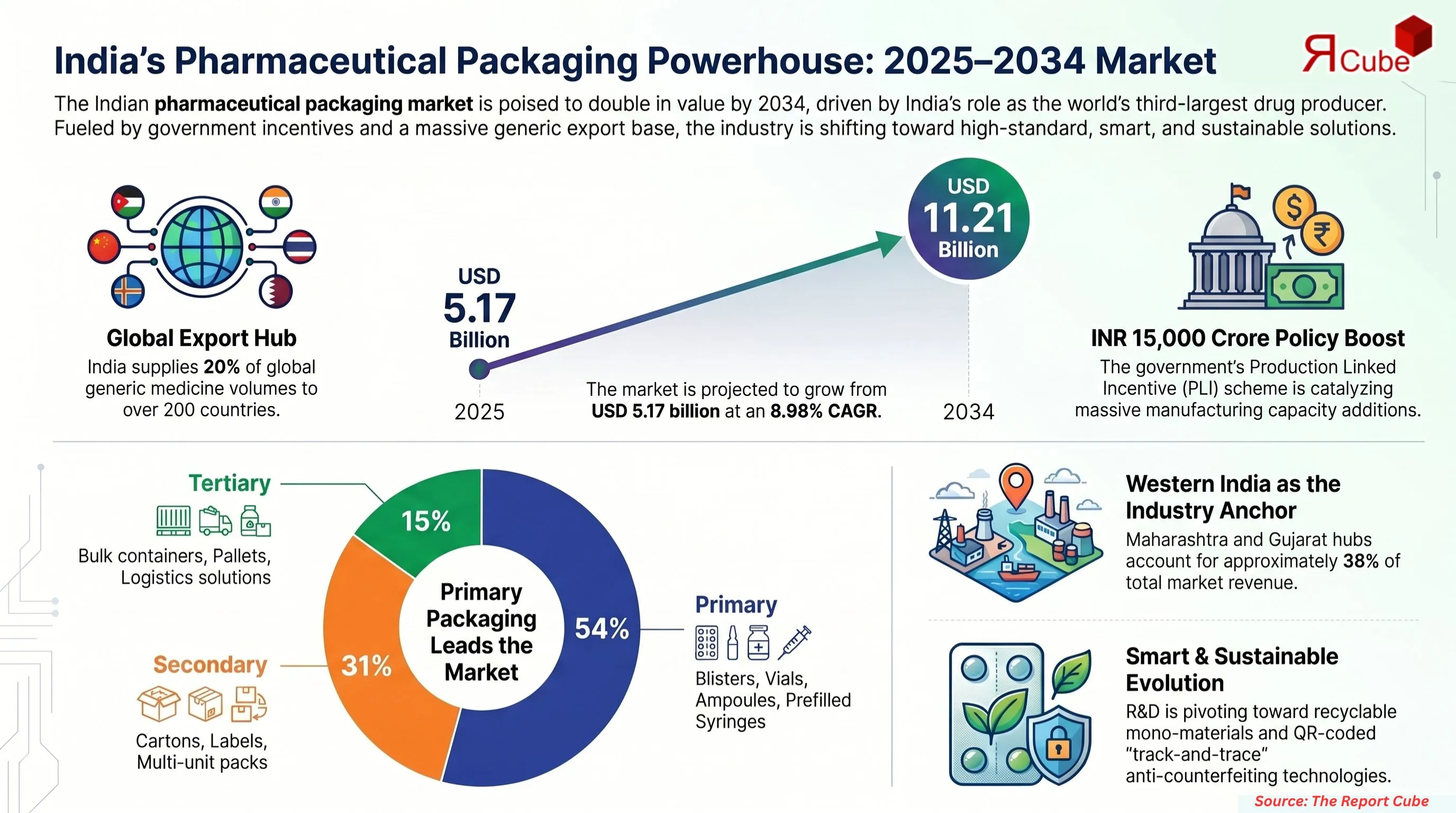
The India pharmaceutical packaging market, currently valued between USD 5.17 billion in 2025, is on track to reach USD 11.21 billion by 2034, advancing at a compound annual growth rate (CAGR) of approximately 8.98% over the 2026–2034 forecast period. This growth is being driven by a simultaneous convergence of mandatory regulatory reform, the rapid expansion of India's biologics sector, and an accelerating national sustainability mandate.
2026 Regulatory Mandates Raise the Packaging Floor
The year 2026 marks a structural reset for pharma packaging in India. The Central Drugs Standard Control Organization (CDSCO), through Notification No. F.1-54/2024-DA, has made several features compulsory for the first time, including high-contrast printing on blister foils, Braille inscriptions on prescription drug cartons, and QR codes with voice-assisted dosage instructions for the top 300 drug brands under the Schedule H2 mandate. Serialization and end-to-end traceability are now regulatory imperatives, not optional upgrades.
Simultaneously, the New Drug Manufacturing Rules (effective March 2026) introduce a formalized three-tier post-approval change classification system, Major, Moderate, and Minor, compelling manufacturers to maintain audit-ready documentation linking every packaging modification to clinical stability data.
For small and medium enterprises, retrofitting lines to meet these standards is estimated to increase capital expenditure by 15% to 25%.
Need Specific Data, Let’s Customize the Report for You
Request CustomizationBiologics and Prefilled Syringes Redefine Packaging Value
The global injectables market, projected to grow from USD 700 billion in 2024 to USD 1.2 trillion by 2030, is reshaping what pharmaceutical packaging means. In India, the transition to prefilled syringes (PFS), ready-to-use (RTU) vials, and integrated drug delivery systems is accelerating, driven largely by the surge in GLP-1 agonist therapies for diabetes and obesity management.
The inauguration of the Corning–SGD Pharma joint venture in Telangana, targeting 2.2 billion Type I borosilicate vials per annum by early 2026, signals India's commitment to anchoring this high-value segment domestically. Cyclic olefin polymers (COP and COC) are also gaining traction as glass alternatives for protein-sensitive biologics.
Sustainability Mandates Accelerate Material Transition
Extended Producer Responsibility (EPR) rules now require 30% recycled content in pharmaceutical plastic packaging, rising to 60% by 2027–2028. Bio-based resins and composites represent the fastest-growing material segment, projected at a 9.65% CAGR through 2034. Companies such as EPL Limited already derive 38% of revenues from sustainable tube formats incorporating up to 70% post-consumer recycled resin.
Market Consolidation Intensifies
The all-stock merger of Amcor and Berry Global, valued at USD 8.4 billion and creating a combined entity with USD 24.3 billion in annual revenue, has set a new benchmark for scale-driven competition in Indian pharma packaging. Amcor's new Gujarat healthcare facility and targeted synergies of USD 650 million by 2027 underscore that capability depth, not just cost, now defines market leadership.
About This Analysis
This release is based on comprehensive market data from pharmaceutical industry reports, CDSCO regulatory filings, and Indian Pharmacopoeia 2026 (IP 2026) documentation covering 3,340 drug quality standards.