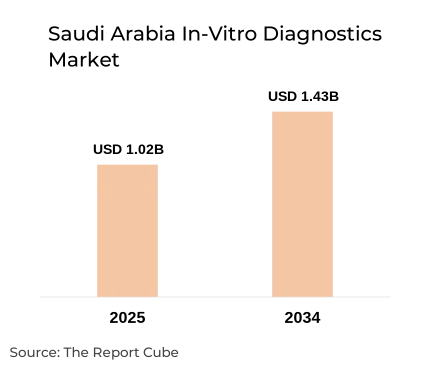
Build Your Custom Market Intelligence Report
Customize Your ReportSaudi Arabia In-Vitro Diagnostics Market Statistics and Insights
Market Size Statistics
- Saudi Arabia In-Vitro Diagnostics Market valued USD 1.02 billion 2025
- Market projected to reach USD 1.43 billion 2034
- Market expected to grow at 3.80% CAGR
By Test Type
- Clinical Chemistry dominates with 34% market share
Competition
- More than 10 companies are actively engaged in producing Saudi Arabia In-Vitro Diagnostics Market
- Top 5 companies acquired around 63% of the market share.
- Abbott Laboratories, Becton Dickinson (BD), Bio-Rad Laboratories, bioMérieux, Danaher (Beckman Coulter & Cepheid), and others
By Product
- Reagents & Kits lead with 61% market share
Saudi Arabia In-Vitro Diagnostics Market Insights & Analysis
The Saudi Arabia In-Vitro Diagnostics Market is anticipated to register a CAGR of 3.80% during 2026-2034. The market size was valued at USD 1.02 billion in 2025 and is projected to reach USD 1.43 billion by 2034. The market reflects stable growth driven by rising chronic disease prevalence, expanding laboratory networks, and healthcare modernization under Vision 2030. Increasing incidence of diabetes, cardiovascular diseases, and infectious conditions is significantly driving diagnostic testing volumes. In 2025, clinical laboratory test volumes recorded consistent growth across public and private healthcare facilities.
Government investments in centralized laboratory infrastructure and digital health integration are strengthening diagnostic capabilities. Expansion of private hospital chains and specialized diagnostic centers is further supporting demand for advanced molecular and immuno-diagnostic solutions. Adoption of automated laboratory systems and integrated diagnostic platforms enhances efficiency and turnaround times. These structural drivers collectively sustain steady market growth and reinforce a positive long-term outlook through 2034.
Saudi Arabia In-Vitro Diagnostics Market Dynamics
Key Market Driver: Rising Chronic Disease Burden and Screening Programs
Increasing prevalence of non-communicable diseases remains the primary growth driver. In 2025, diabetes prevalence exceeded 17% among adults, significantly boosting demand for glucose monitoring and clinical chemistry tests. National screening programs for infectious diseases and early cancer detection are expanding molecular diagnostic adoption. Growth in preventive health check-ups further increases routine hematology and immuno-diagnostic testing. Expansion of healthcare insurance coverage also supports higher diagnostic utilization. These factors substantially enhance market share across reagents and clinical chemistry segments.
Major Industry Challenge: Import Dependency and Pricing Pressure
The market remains heavily reliant on imported diagnostic equipment and reagents, exposing suppliers to currency fluctuations and global supply chain disruptions. Centralized procurement policies and tender-based pricing frameworks may compress margins. Maintenance and calibration requirements for advanced diagnostic instruments increase operational complexity. Regulatory approvals for new diagnostic assays require strict compliance with national standards. These structural challenges shape market analysis and necessitate local manufacturing partnerships and cost optimization strategies.
Emerging Trend Shaping Outlook: Automation and Molecular Diagnostic Expansion
Automation in laboratory workflows is improving productivity and reducing human error. In 2025, adoption of fully automated analyzers increased across tertiary hospitals. Molecular diagnostics, including PCR-based testing, are gaining prominence in infectious disease and oncology detection. AI-assisted data interpretation tools are enhancing diagnostic accuracy and reporting efficiency. Integration of laboratory information systems (LIS) with hospital networks strengthens digital health ecosystems. These developments are shaping market trends and contributing to incremental growth.
Need Specific Data, Let’s Customize the Report for You
Request Regional DataSaudi Arabia In-Vitro Diagnostics Market Segment-wise Analysis
The market share distribution varies by test type and product category, reflecting diagnostic demand patterns across healthcare facilities.
By Test Type
- Clinical Chemistry
- Molecular Diagnostics
- Immuno-Diagnostics
- Hematology
- Other Test Types
Clinical Chemistry accounts for approximately 34% of total revenue due to high routine testing volumes. Molecular Diagnostics represent nearly 23% share supported by infectious disease and oncology screening. Immuno-Diagnostics contribute around 19% share reflecting hormone and antigen testing demand. Hematology holds approximately 15% share driven by routine blood analysis. Other Test Types collectively account for nearly 9%. This segmentation defines the structural composition of the market size.
By Product
- Instruments
- Reagents & Kits
Reagents & Kits dominate with approximately 61% of total revenue due to recurring consumption in routine testing. Instruments account for nearly 39% share supported by capital investment in automated analyzers and diagnostic platforms. Continuous technological upgrades in instrumentation further strengthen this segment. This segmentation framework supports comprehensive market analysis.
Regional Projection of Saudi Arabia In-Vitro Diagnostics Market
The market demonstrates concentrated demand across major healthcare hubs supported by hospital density and laboratory infrastructure.
- Riyadh
- Jeddah
- Dammam
- Mecca
- Medina
Riyadh leads with approximately 36% of total market demand due to concentration of tertiary hospitals and centralized laboratories. Jeddah contributes nearly 24% share supported by private healthcare expansion. Dammam accounts for around 17% reflecting industrial and urban population growth. Mecca represents approximately 13% share influenced by seasonal healthcare demand. Medina contributes nearly 10%, supported by regional healthcare network development. These regional dynamics reinforce the market outlook through 2034.
Saudi Arabia In-Vitro Diagnostics Market: Recent Development
- 2025: Abbott Laboratories expanded automated immunoassay systems across hospitals.
- 2025: Bio-Rad Laboratories introduced advanced molecular diagnostic platforms.
- 2025: Becton Dickinson strengthened reagent supply agreements nationwide.
- 2025: Danaher expanded local service support for diagnostic instruments.
Saudi Arabia In-Vitro Diagnostics Market Future Outlook (2034)
The Saudi Arabia In-Vitro Diagnostics Market is projected to sustain a CAGR of 3.80%, reaching USD 1.43 billion by 2034. Growth will be driven by chronic disease management, screening program expansion, and laboratory automation adoption. Advancements in molecular diagnostics, AI-based data analytics, and integrated laboratory systems will enhance diagnostic precision and efficiency. Expansion of private diagnostic centers and health insurance coverage will further stimulate testing volumes. As healthcare infrastructure modernization continues under Vision 2030, sustained market growth is anticipated. Strategic localization of reagent production and digital health integration will maintain a stable market outlook through the forecast period.
Why Choose This Report?
- Provides a comprehensive overview of the overall market analysis, encompassing key trends, consumer behavior analysis, and risk assessment to support strategic decision-making.
- Provides accurate, up-to-date insights into market size, segmentation, and emerging opportunities, helping to minimize risk & capitalizing on growth.
- Gives deep understanding of target audience preferences, investment habits, and communication channels for enhanced product development & marketing effectiveness.
- Delivers competitive analysis & benchmarking, uncovering the strengths & weaknesses of market competitors to guide strategies.
- Consolidate comprehensive market intelligence, reducing reasoning & streamlining research efforts.
- Facilitates customized market segmentation & risk mitigation strategies, fine-tuned to the business objectives.
- Aids in identifying both market challenges & untapped opportunities within the industry to drive long-term business growth.
- Provides valuable information based on actual customer data & search trends.
Table of Contents
- Introduction
- Objective of the study
- Product Definition
- Market Segmentation
- Study Variables
- Research Methodology
- Secondary Data Points
- Companies Interviewed
- Primary Data Points
- Breakdown of Primary Interviews
- Secondary Data Points
- Executive Summary
- Market Dynamics
- Drivers
- Challenges
- Opportunity Assessment
- Recent Trends and Developments
- Policy and Regulatory Landscape
- Saudi Arabia In-Vitro Diagnostics Market Overview (2021-2034)
- Market Size, By Value (in USD Billions)
- Market Share, By Test Type
- Clinical Chemistry
- Molecular Diagnostics
- Immuno-Diagnostics
- Hematology
- Other Test Types
- Market Share, By Product
- Instruments
- Reagents & Kits
- Market Share, By Usability
- Disposable IVD Devices
- Re-Usable IVD Devices
- Market Share, By Application
- Infectious Disease
- Diabetes
- Oncology
- Cardiology
- Other Applications
- Market Share, By End-User
- Diagnostic Laboratories
- Hospitals & Clinics
- Home-Care & POC Settings
- Other End-Users
- Market Share, By Region
- Riyadh
- Jeddah
- Dammam
- Mecca
- Medina
- Market Share, By Competitors
- Competition Characteristics
- Revenue Shares
- Saudi Arabia Clinical Chemistry In-Vitro Diagnostics Market Overview, 2021-2034F
- By Value (USD Million/Billion)
- By Product- Market Size & Forecast 2021-2034, USD Million/Billion
- By Usability- Market Size & Forecast 2021-2034, USD Million/Billion
- By Application- Market Size & Forecast 2021-2034, USD Million/Billion
- By End-User- Market Size & Forecast 2021-2034, USD Million/Billion
- Saudi Arabia Molecular Diagnostics In-Vitro Diagnostics Market Overview, 2021-2034F
- By Value (USD Million/Billion)
- By Product- Market Size & Forecast 2021-2034, USD Million/Billion
- By Usability- Market Size & Forecast 2021-2034, USD Million/Billion
- By Application- Market Size & Forecast 2021-2034, USD Million/Billion
- By End-User- Market Size & Forecast 2021-2034, USD Million/Billion
- Saudi Arabia Immuno-Diagnostics In-Vitro Diagnostics Market Overview, 2021-2034F
- By Value (USD Million/Billion)
- By Product- Market Size & Forecast 2021-2034, USD Million/Billion
- By Usability- Market Size & Forecast 2021-2034, USD Million/Billion
- By Application- Market Size & Forecast 2021-2034, USD Million/Billion
- By End-User- Market Size & Forecast 2021-2034, USD Million/Billion
- Saudi Arabia Hematology In-Vitro Diagnostics Market Overview, 2021-2034F
- By Value (USD Million/Billion)
- By Product- Market Size & Forecast 2021-2034, USD Million/Billion
- By Usability- Market Size & Forecast 2021-2034, USD Million/Billion
- By Application- Market Size & Forecast 2021-2034, USD Million/Billion
- By End-User- Market Size & Forecast 2021-2034, USD Million/Billion
- Saudi Arabia Other Test Types In-Vitro Diagnostics Market Overview, 2021-2034F
- By Value (USD Million/Billion)
- By Product- Market Size & Forecast 2021-2034, USD Million/Billion
- By Usability- Market Size & Forecast 2021-2034, USD Million/Billion
- By Application- Market Size & Forecast 2021-2034, USD Million/Billion
- By End-User- Market Size & Forecast 2021-2034, USD Million/Billion
- Competitive Outlook (Company Profile - Partial List)
- Abbott Laboratories
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- Al Borg Diagnostics
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- Al Razi Medical Labs
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- Becton Dickinson (BD)
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- Bio-Rad Laboratories
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- bioMérieux
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- Danaher (Beckman Coulter & Cepheid)
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- Dammam Regional Laboratory
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- DiaSorin
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- Others
- Abbott Laboratories
- Contact Us & Disclaimer
Top Key Players & Market Share Outlook
- Abbott Laboratories
- Al Borg Diagnostics
- Al Razi Medical Labs
- Becton Dickinson (BD)
- Bio-Rad Laboratories
- bioMérieux
- Danaher (Beckman Coulter & Cepheid)
- Dammam Regional Laboratory
- DiaSorin
- Others
Frequently Asked Questions






