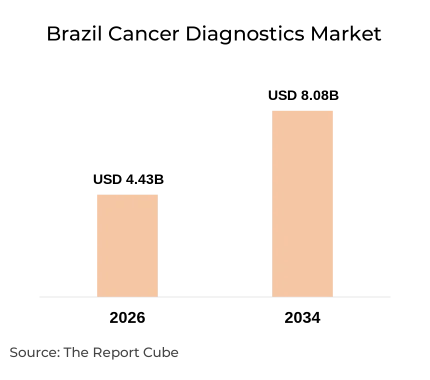
Build Your Custom Market Intelligence Report
Customize Your ReportKey Takeaways
- The Brazil cancer diagnostics market is valued at USD 4.43 billion in 2026 and is projected to reach USD 8.08 billion by 2034, expanding at a CAGR of 7.80%.
- Early Detection is the leading segment by type, fuelled by ANS Resolution 465/2021 mandating private insurer coverage of cancer screening tests.
- Services - driven by domestic lab networks Fleury, Dasa, and Hermes Pardini - dominate product-level spending due to SUS procurement constraints on capital equipment.
- Molecular diagnostics and NGS adoption are accelerating, led by Illumina, Roche, and Thermo Fisher Scientific repositioning their Brazilian commercial operations.
- A chronic pathologist shortage - fewer than 3,000 active practitioners for 215 million people - is creating urgent demand for AI-assisted digital pathology platforms.
- Infrastructure inequity remains the single largest structural constraint: fewer than 15% of accredited oncology diagnostic centres serve regions home to 30%+ of Brazil's population.
Brazil Cancer Diagnostics Market Insights & Analysis
Brazil carries one of Latin America's highest cancer burdens. INCA (Instituto Nacional de Câncer) projects over 704,000 new cancer cases annually through 2025, yet diagnostic infrastructure across the country's interior states remains severely underdeveloped. Late-stage diagnoses account for a disproportionate share of oncology spending - treating a Stage III–IV cancer costs three to five times more per patient than intervening at Stage I. This structural gap is reshaping investment in diagnostics at pace.
The Brazil cancer diagnostics market reached USD 4.43 billion in 2026 and is on track to nearly double to USD 8.08 billion by 2034, growing at a CAGR of 7.80% (Report Publisher, 2026). That trajectory reflects two simultaneous forces: the expansion of government-led screening through SUS (Sistema Único de Saúde) and the rapid growth of private oncology diagnostic labs in São Paulo, Rio de Janeiro, and Belo Horizonte.
The technology mix is shifting as well. Workflows that once relied exclusively on histopathology are moving toward molecular and genomic diagnostics - a transition that upgrades both the clinical value and the unit economics of each test. For companies operating in the Latin America cancer diagnostics market, Brazil represents the region's single largest and fastest-evolving opportunity.
Brazil Cancer Diagnostics Market Dynamics
Brazil's cancer diagnostics sector operates under competing pressures - technological advancement at private urban centres versus chronic underfunding at public diagnostic units. São Paulo state alone accounts for an estimated 40%+ of national diagnostic volume, while the North and Northeast lag by a decade in infrastructure depth. Four forces define where this market moves next.
Key Market Driver - Rising Cancer Incidence & Government Screening Mandates
Brazil's cancer burden falls unevenly across cancer types. Cervical, breast, colorectal, and prostate cancers together account for over 50% of new diagnoses annually, and most present at Stage II or later. INCA data show that five-year survival rates for late-detected breast cancer in Brazil sit below 60%, compared with 90%+ when detected at Stage I - a performance gap that regulators can no longer ignore.
The most powerful demand trigger in the Brazil cancer diagnostics market is policy-driven: ANS Resolution 465/2021 mandates that private insurers cover specific cancer screening tests, including mammography, PAP smears, colonoscopy, and PSA panels. This single regulatory intervention has driven a measurable spike in early-detection test volumes at accredited private labs. SUS Ministry of Health budget lines for oncology diagnostics are expanding in parallel, with new procurement tenders for imaging and pathology instrumentation across state hospital networks.
The quantifiable effect: private-lab screening test volumes in São Paulo grew by an estimated double-digit percentage in the two years following the ANS mandate - a linkage competitor rarely model directly. Brands and providers operating in the Brazil healthcare market must account for this policy-to-volume mechanism in any demand forecast.
Major Industry Challenge - Infrastructure Gaps & Diagnostic Inequity Across Regions
Brazil's geographic scale directly undermines uniform diagnostic access. The Amazon basin and Northeast states contain over 30% of the national population but fewer than 15% of accredited oncology diagnostic centres, according to CFM (Conselho Federal de Medicina) data. Patients in these regions average 120+ days between symptomatic presentation and confirmed cancer diagnosis - nearly triple the São Paulo metropolitan benchmark of 45 days.
IBGE regional population data make clear that this is not a marginal problem. The Northeast and North together represent a significant addressable volume of unmet diagnostic demand, quantifiable as a market opportunity rather than merely a social policy challenge. INCA's mobile diagnostic unit programme has extended reach into some underserved states, but coverage remains insufficient relative to population need. Private-sector telehealth and remote pathology initiatives are beginning to bridge the gap, making this underserved cohort commercially reachable for the first time. Companies with exposure to the Brazil medical devices market should treat diagnostic infrastructure expansion in Tier 2 and Tier 3 cities as a primary growth vector through 2034.
Emerging Trend - Molecular Diagnostics & NGS Adoption
Brazilian oncologists have historically relied on immunohistochemistry and standard pathology panels - methods that cannot identify targetable genomic alterations. Targeted therapy protocols for NSCLC, breast, and colorectal cancers now require companion diagnostics that standard pathology cannot deliver, creating urgent clinical demand for NGS (next-generation sequencing) and liquid biopsy.
ANVISA (Agência Nacional de Vigilância Sanitária) has approved dedicated regulatory pathways for molecular diagnostic kits, but commercial launch timelines lag approval dates by six to eighteen months - a bottleneck that constrains market penetration even where clinical demand exists. Key players are moving aggressively: Illumina has launched NGS partnerships with major São Paulo oncology centres for somatic tumour profiling; Thermo Fisher Scientific is deploying Ion Torrent platforms through an expanded Brazilian distributor network; and Roche is advancing Foundation Medicine partnerships in the country's largest academic medical centres. Liquid biopsy clinical trial activity on ClinicalTrials.gov confirms that Brazil is now an active NGS development market, not merely an adoption follower.

Need Specific Data, Let’s Customize the Report for You
Request CustomizationEmerging Opportunity - AI-Assisted Diagnostics & Digital Pathology
Brazil's pathologist workforce shortage - fewer than 3,000 active practitioners for a population of 215 million - limits diagnostic throughput at public facilities even when biopsy slides are physically available. A backlog of unread slides at SUS-linked hospitals delays treatment initiation by an average of 3–6 weeks in under-resourced states, according to CFM estimates.
AI-assisted image analysis and digital pathology scanning platforms offer a direct throughput solution. Koninklijke Philips NV's IntelliSite Pathology Solution is currently in pilot deployment across Brazilian hospital networks; Siemens Healthineers has signed a digital pathology pilot agreement with the Rio de Janeiro state health secretariat. São Paulo and Minas Gerais state governments have issued tenders for digital pathology scanners. The Brazilian Society of Clinical Pathology (SBPC/ML) has issued a formal position statement validating digital pathology workflows - removing the last major institutional barrier to procurement.
Brazil Cancer Diagnostics Market - Segment-wise Analysis
By Type - Diagnosis, Monitoring, Early Detection & Therapy Selection
|
Type |
Growth Outlook |
Primary Driver |
|---|---|---|
|
Diagnosis |
Moderate |
High cancer incidence |
|
Monitoring |
Moderate |
Survivorship care expansion |
|
Early Detection |
High |
ANS screening mandates |
|
Therapy Selection |
Fastest |
Targeted therapy adoption |
Historically, Brazilian oncology spending concentrated at the Diagnosis stage - confirming a cancer already suspected. ANS mandate expansion and private-insurer pressure are now measurably redistributing spending toward Early Detection and Therapy Selection. Companion diagnostics for HER2, EGFR, and PD-L1 testing are the fastest-growing sub-segment, directly linked to targeted therapy prescription volumes at private oncology centres. For a full view of treatment-side demand, see the Brazil oncology market report.
By Product - Services, Instruments (Pathology-based, Others)
|
Product |
Growth Outlook |
Key Suppliers |
|---|---|---|
|
Services |
Steady |
Fleury, Dasa, Hermes Pardini |
|
Pathology-based Instruments |
Moderate |
Roche, Leica (Danaher) |
|
Instruments |
High |
BD, Abbott, Qiagen, Thermo Fisher |
|
Others |
Moderate |
Multiple |
Services dominate product-level spending because SUS procurement rules require competitive tendering for capital equipment above BRL 80,000, extending instrument acquisition timelines by 12–24 months. Brazil's domestic reference lab networks - Fleury, Dasa, and Hermes Pardini - form the structural backbone of this segment, operating as the primary channel through which diagnostic testing reaches patients across the country. The product mix is gradually rebalancing as private oncology groups invest in in-house pathology and sequencing capacity. For reagent and instrument segment detail, see the Brazil in-vitro diagnostics market report.
Future Outlook - Brazil Cancer Diagnostics Market to 2034
Brazil's cancer diagnostics market in 2034 will not resemble today's instrument-light, service-heavy structure. Three forces - policy-led screening normalization, genomic medicine mainstreaming, and AI-assisted diagnostic throughput - will redefine the market's shape by the end of the forecast period.
SUS universal cancer screening expansion for breast, cervical, and colorectal cancers will significantly increase public-sector test volumes, narrowing the current public/private revenue split. NGS adoption will move from concentrated urban oncology centres toward regional hospital networks as ANVISA approvals translate into commercial availability. AI digital pathology will address the pathologist shortage at scale, enabling SUS-linked hospitals to process biopsy backlogs that currently cost the system weeks of treatment delay.
The market reaches USD 8.08 billion by 2034 at a 7.80% CAGR (Report Publisher, 2026). Without early detection investment, the SUS faces compounding cost pressure: treating late-stage cancers costs an estimated 3-4× more per patient than Stage I intervention. That arithmetic alone makes diagnostic investment a healthcare economic necessity, not a discretionary spend. For the global context, see the WHO / IARC Global Cancer Report.
Table of Contents
- Introduction
- Objective of the study
- Product Definition
- Market Segmentation
- Study Variables
- Research Methodology
- Secondary Data Points
- Companies Interviewed
- Primary Data Points
- Breakdown of Primary Interviews
- Secondary Data Points
- Executive Summary
- Market Dynamics
- Drivers
- Challenges
- Opportunity Assessment
- Recent Trends and Developments
- Policy and Regulatory Landscape
- Brazil Dental Care Market Overview and Forecast Analysis (2021-2034)
- Market Size, By Value, By growth rate (CAGR/USD Billions)
- Demand - Supply Trends
- Market Share, By Coverage
- Private
- Public insurance
- Market Share, By Technology
- Imaging
- Computed Tomography (CT)
- Mammography
- Ultrasound
- Others Imaging
- Magnetic Resonance Imaging (MRI)
- Positron Emission Tomography (PET)
- Others
- Market Share, By Application
- Breast Cancer
- Cervical Cancer
- Colorectal Cancer
- Prostate Cancer
- Others
- Market Share, By Type
- Diagnosis
- Monitoring
- Early Detection
- Therapy Selection
- Market Share, By Product
- Services
- Instruments
- Pathology-based Instruments
- Others
- Market Share, By End Use
- Hospitals
- Laboratories
- Others
- Market Share, By Competitors
- Competition Characteristics
- Revenue Shares
- Brazil Private Dental Care Market Overview, 2021-2034F
- By Value (USD Million)
- By Technology- Market Size & Forecast 2021-2034, USD Million
- By Application- Market Size & Forecast 2021-2034, USD Million
- By Type- Market Size & Forecast 2021-2034, USD Million
- By Product- Market Size & Forecast 2021-2034, USD Million
- By End Use- Market Size & Forecast 2021-2034, USD Million
- Brazil Public Insurance Dental Care Market Overview, 2021-2034F
- By Value (USD Million)
- By Technology- Market Size & Forecast 2021-2034, USD Million
- By Application- Market Size & Forecast 2021-2034, USD Million
- By Type- Market Size & Forecast 2021-2034, USD Million
- By Product- Market Size & Forecast 2021-2034, USD Million
- By End Use- Market Size & Forecast 2021-2034, USD Million
- Competitive Outlook (Company Profile - Partial List)
- Hologic Inc
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- Qiagen NV
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- Illumina Inc
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- Roche Holding AG
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- Thermo Fisher Scientific Inc
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- Siemens Healthineers AG ADR
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- oninklijke Philips NV
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- Becton Dickinson & Co
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- Abbott Laboratories
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- Others
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- Hologic Inc
- Contact Us & Disclaimer
Top Key Players & Market Share Outlook
- Hologic Inc
- Qiagen NV
- Illumina Inc
- Roche Holding AG
- Thermo Fisher Scientific Inc
- Siemens Healthineers AG ADR
- oninklijke Philips NV
- Becton Dickinson & Co
- Abbott Laboratories
- Others
Frequently Asked Questions






