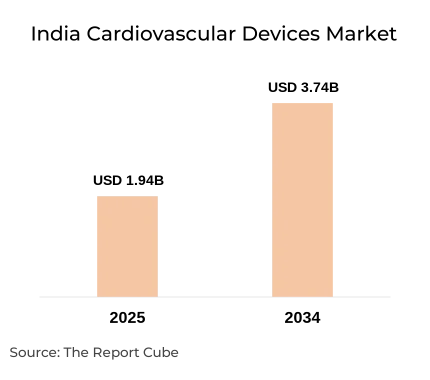
Build Your Custom Market Intelligence Report
Customize Your ReportIndia Cardiovascular Devices Market Statistics and Insights
Market Insight
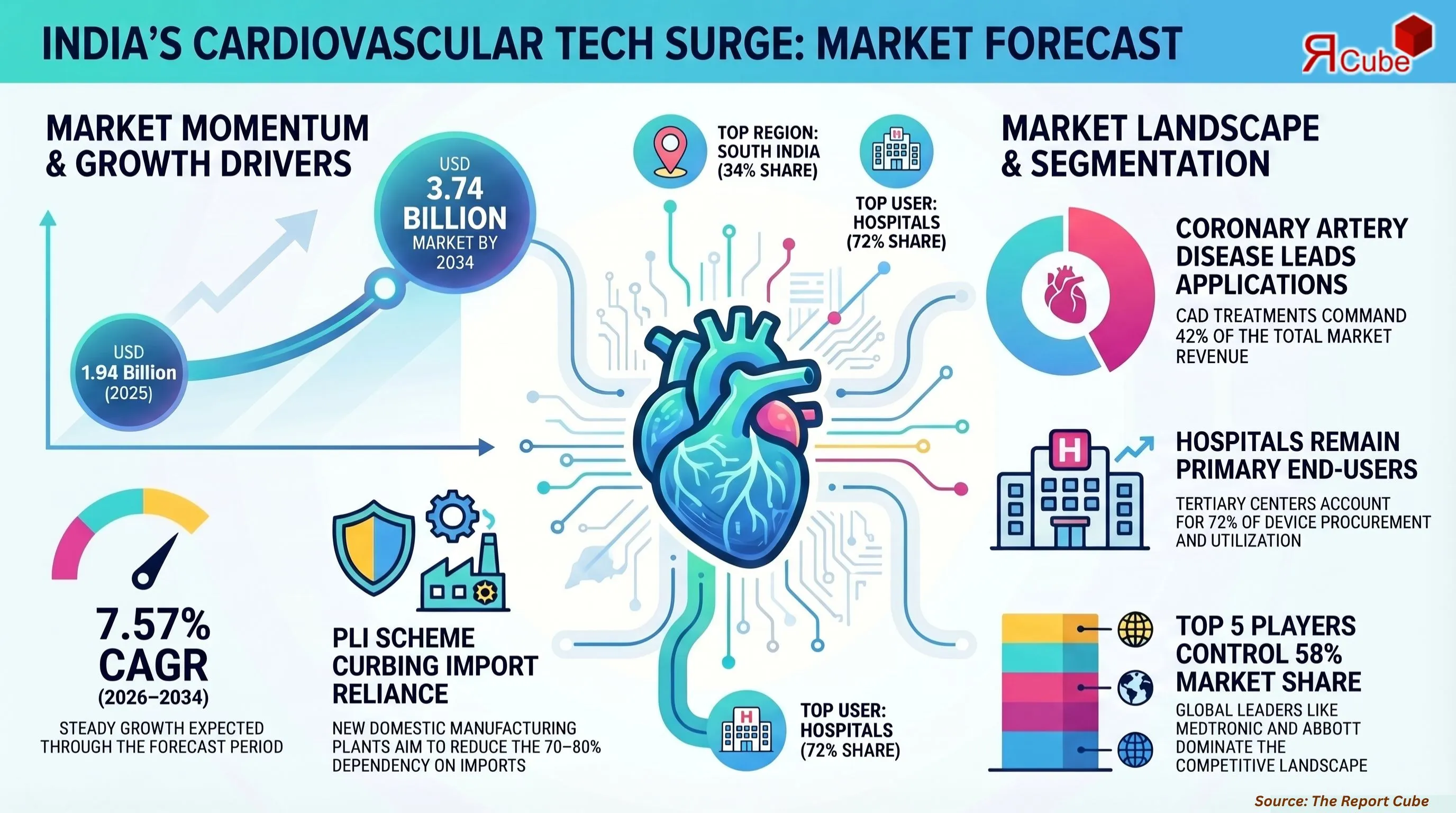
- The India Cardiovascular Devices Market was valued at USD 1.94 billion in 2025.
- The India Cardiovascular Devices Market is projected to reach USD 3.74 billion by 2034.
- The market is anticipated to register a CAGR of 7.57% during 2026–2034.
By Application:
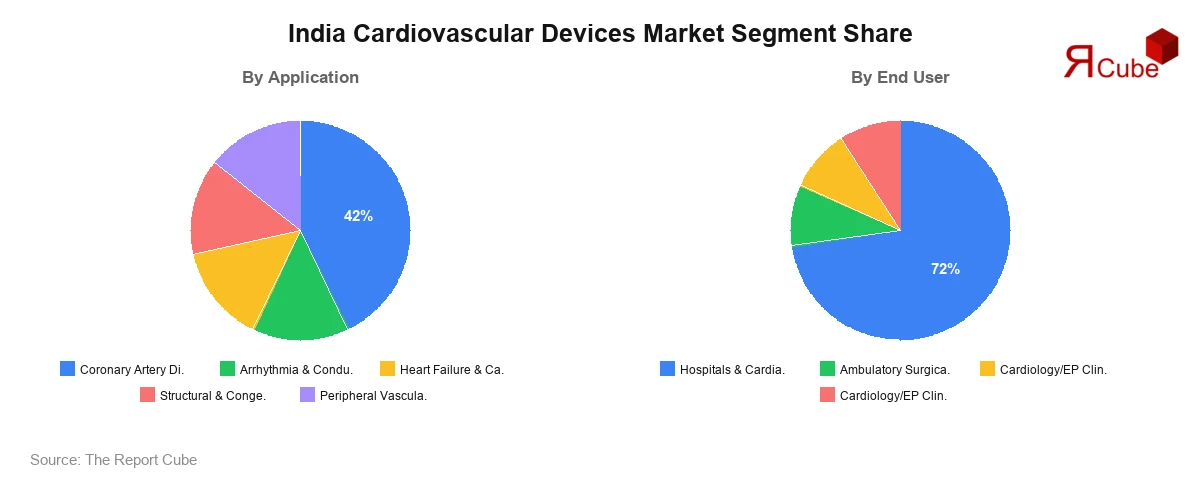
- Coronary Artery Disease leads the segment, commanding approximately 42% market share.
Key Players:
- More than 10 companies are actively engaged in producing India Cardiovascular Devices.
- Top 5 companies acquired around 58% of the market share.
- Medtronic plc, Abbott Laboratories, Boston Scientific Corporation, Edwards Lifesciences Corporation, Meril Life Sciences Pvt. Ltd., and others.
By End User:
- Hospitals & Cardiac Centres dominate the end-user segment, accounting for approximately 72% market share.
India Cardiovascular Devices Market Insights & Analysis
The India Cardiovascular Devices Market is anticipated to register a 7.57% CAGR during 2026–2034. The market size was valued at USD 1.94 billion in 2025 and is projected to reach USD 3.74 billion by 2034. The market's expansion is anchored in the country's severe and escalating cardiovascular disease burden, with ischemic heart disease and stroke cases rising 2.3-fold since 1990 and cardiovascular conditions now accounting for 14.1% of national disability-adjusted life years (DALYs).
India's Pradhan Mantri Jan Arogya Yojana (PM-JAY) scheme reimburses cardiac procedures including surgeries and implants for over 500 million beneficiaries, directly amplifying device procurement volumes across public and private facilities. The Production Linked Incentive (PLI) scheme has authorized 19 greenfield medical device manufacturing plants, reducing import dependency that previously stood at 70–80% for high-end cardiac products. Abbott Laboratories in September 2025 expanded its collaboration with local healthcare providers to broaden access to cardiovascular monitoring technologies, while Boston Scientific in August 2025 completed the acquisition of a local digital health startup focused on cardiac patient management, underscoring the deepening strategic interest of global players in the Indian cardiovascular space.
India Cardiovascular Devices Market Dynamics
Key Market Driver PLI Scheme Accelerates Indigenous Cardiovascular Manufacturing
India's Production Linked Incentive scheme has fundamentally restructured the cardiovascular devices supply chain by catalyzing domestic manufacturing at scale. As of early 2025, the PLI program logged INR 1.61 lakh crore in total investment commitments across the broader medical devices sector, with 19 approved greenfield factories targeting high-end modalities previously sourced entirely from imports.
Companies such as Meril Life Sciences and Sahajanand Medical Technologies have expanded drug-eluting stent and heart valve production lines for both domestic consumption and export to African and Southeast Asian markets. Siemens Healthineers committed INR 91.9 crore to imaging production capacity, while Wipro GE pledged INR 8,000 crore toward device R&D and manufacturing. This policy-driven industrial transformation is simultaneously lowering device prices, improving supply reliability, and enabling Indian firms to move up the cardiovascular value chain toward more sophisticated implantable and interventional products.
Major Industry Challenge Regulatory Fragmentation Limits Uniform Market Penetration
India's multi-layered regulatory environment for cardiovascular devices presents a structural challenge that disproportionately affects both global entrants and domestic firms seeking to scale nationally. The Central Drugs Standard Control Organisation (CDSCO) governs device approvals at the national level, but state-level procurement norms, tendering processes, and facility accreditation standards vary considerably, creating inconsistency in how devices are evaluated, priced, and adopted across different geographies.
South Asian coronary anatomy, characterized by smaller vessel diameters and diffuse lesion patterns, necessitates device specifications that deviate from globally standardized designs, compelling manufacturers to invest in India-specific clinical validation studies. Rural healthcare facilities, which serve a large share of the population most affected by cardiovascular disease, frequently lack the catheterization laboratory infrastructure required to deploy interventional and implantable devices, restricting addressable market penetration beyond Tier 1 and Tier 2 cities.
Emerging Trend Shaping Outlook Telecardiology Reshaping Remote Cardiac Monitoring Infrastructure
The convergence of India's Digital Health Mission with rapid proliferation of low-cost wearable cardiac monitoring devices is establishing a new layer of cardiovascular care delivery that operates independently of hospital infrastructure. Portable ECG monitors, Bluetooth-enabled Holter devices, and AI-assisted arrhythmia detection wearables are being deployed through government-funded telehealth programs to serve patients in semi-urban and rural areas who previously had no access to cardiac diagnostics.
In July 2025, the SSI Mantra 3 robotic system executed India's first cardiac telesurgeries across a 250-km distance, demonstrating the technical viability of low-latency remote cardiac intervention. Home-care and remote monitoring programs, currently the fastest-growing end-user segment, are expected to sustain double-digit expansion through 2034 as hospitals and device manufacturers develop integrated home-based cardiac care models targeting elderly and chronic heart failure patients. This trend is progressively expanding the total addressable market for cardiovascular devices beyond traditional clinical settings.
Need Specific Data, Let’s Customize the Report for You
Request CustomizationIndia Cardiovascular Devices Market Segment-wise Analysis
The India Cardiovascular Devices Market encompasses a broad spectrum of devices spanning interventional, implantable, diagnostic, and remote monitoring categories, each aligned to distinct disease burdens and care-delivery settings. The market's segmentation by application and by end user reveals heterogeneous growth dynamics, with certain sub-segments driven by epidemiological pressures and others by infrastructural transformation. Both segments together capture the full clinical pathway from disease diagnosis through long-term disease management.
By Application
- Coronary Artery Disease
- Arrhythmia & Conduction Disorders
- Heart Failure & Cardiomyopathy
- Structural & Congenital Heart Defects
- Peripheral Vascular Disease
Coronary Artery Disease (CAD) is the dominant application segment, accounting for approximately 42% of total market revenue in 2025. India's urban-rural lifestyle convergence, marked by sedentary behavior, tobacco use, and energy-dense diets, has produced hypertension and dyslipidemia at a population scale that ensures sustained, high-volume demand for stents, angioplasty catheters, and coronary guidewires. Drug-eluting stents and bioresorbable vascular scaffolds from both domestic manufacturers and global players represent the highest-revenue sub-category within this segment.
Arrhythmia & Conduction Disorders is the fastest-growing application, driven by the expanding adoption of implantable cardioverter-defibrillators (ICDs), cardiac resynchronization therapy (CRT) devices, and electrophysiology catheters. Growth in this sub-segment is supported by PM-JAY reimbursements covering rhythm management procedures. Heart Failure & Cardiomyopathy is gaining share as ventricular assist devices and advanced heart failure monitoring systems, once restricted to premium tertiary hospitals, become accessible through public-private partnerships. Structural & Congenital Heart Defects is supported by TAVR system adoption and Meril Life Sciences' Myval transcatheter heart valve, which has secured CE-mark access while targeting domestic affordability. Peripheral Vascular Disease accounts for a smaller but steadily growing revenue share as diabetic vasculopathy burden intensifies across India's urban population.
By End User
- Hospitals & Cardiac Centres
- Ambulatory Surgical Centres
- Cardiology/EP Clinics
- Home-care & Remote Monitoring Programs
Hospitals & Cardiac Centres dominate the end-user landscape with approximately 72% revenue share, a reflection of the concentration of catheterization laboratories, cardiac surgery suites, and electrophysiology facilities in private and public tertiary institutions in metropolitan areas. Major hospital groups in Bengaluru, Chennai, and Delhi-NCR have expanded dedicated cardiac intervention units, sustaining high-volume procurement of stents, catheters, pacemakers, and imaging systems.
Ambulatory Surgical Centres are an emerging end-user category as minimally invasive procedures, angioplasty, ablation, and peripheral interventions, migrate from inpatient to day-care settings, reducing cost and improving patient throughput. Cardiology/EP Clinics are expanding access in Tier 2 and Tier 3 cities where tertiary hospitals remain scarce, driving demand for compact diagnostic and monitoring devices. Home-care & Remote Monitoring Programs represent the most rapidly expanding end-user segment, underpinned by the Digital Health Mission, government telehealth funding, and partnerships between manufacturers and hospital networks to deliver chronic cardiac disease management outside clinical walls.
Regional Projection of India Cardiovascular Devices Market
The India Cardiovascular Devices Market is analyzed across the following regional geographies:
- North India
- South India
- West India
- East India
South India leads the India Cardiovascular Devices Market with approximately 34% of national revenue, anchored by the concentration of super-specialty cardiac hospitals and medical device manufacturing clusters in Chennai, Bengaluru, and Hyderabad. The region's well-developed private healthcare ecosystem, high physician density, and favorable state-level health insurance coverage drive disproportionately high procedure volumes and device procurement.
North India is the second-largest regional segment, with Delhi-NCR serving as a hub for interventional cardiology and cardiac surgery, and government-funded tertiary hospitals under the All India Institute of Medical Sciences network stimulating high-volume device consumption. West India, led by Maharashtra and Gujarat, is experiencing accelerated growth as medical device SEZ developments and PLI-backed manufacturing investments cluster around Pune and Ahmedabad. East and Central India remain underpenetrated but are targeted by telecardiology programs and mobile cardiac diagnostic deployments as infrastructure investment expands.
India Cardiovascular Devices Market: Recent Development (2025)
- Abbott Laboratories expanded cardiac monitoring technology access via local partnerships, recording approximately 9% revenue growth in India operations in FY2025.
- Boston Scientific completed acquisition of a digital cardiovascular health startup, strengthening its remote monitoring and AI-enabled patient engagement portfolio for India.
- Meril Life Sciences' Myval transcatheter heart valve secured CE research access while scaling affordable structural heart device supply for domestic and export markets.
- Sahajanand Medical Technologies reported a 15% increase in drug-eluting stent production output, driven by expanded PLI-backed manufacturing lines and African export contracts.
India Cardiovascular Devices Market Future Outlook (2034)
The India Cardiovascular Devices Market is positioned for sustained and structurally underpinned expansion through 2034, supported by a 7.57% CAGR that is projected to elevate total market value to USD 3.74 billion. Growth opportunities will be concentrated in electrophysiology devices, transcatheter structural heart solutions, and wearable remote monitoring platforms as chronic disease management evolves beyond hospital settings.
AI-driven cardiac diagnostics, encompassing machine learning-assisted echocardiography interpretation, arrhythmia classification algorithms embedded in wearables, and predictive heart failure management tools, will become integral to device ecosystems rather than supplementary software layers. Domestic manufacturers are expected to capture a significantly larger share of interventional device categories as PLI investments mature, reducing import dependence to below 50% by 2034. Export of Indian-manufactured cardiovascular devices to emerging economies in Africa, Southeast Asia, and the Middle East will provide an additional growth vector, leveraging India's established cost-performance advantage in stents, pacemakers, and monitoring consumables.
Why Choose This Report?
- Provides a comprehensive overview of the overall market analysis, encompassing key trends, consumer behavior analysis, and risk assessment to support strategic decision-making.
- Provides accurate, up-to-date insights into market size, segmentation, and emerging opportunities, helping to minimize risk & capitalizing on growth.
- Gives deep understanding of target audience preferences, investment habits, and communication channels for enhanced product development & marketing effectiveness.
- Delivers competitive analysis & benchmarking, uncovering the strengths & weaknesses of market competitors to guide strategies.
- Consolidate comprehensive market intelligence, reducing reasoning & streamlining research efforts.
- Facilitates customized market segmentation & risk mitigation strategies, fine-tuned to the business objectives.
- Aids in identifying both market challenges & untapped opportunities within the industry to drive long-term business growth.
- Provides valuable information based on actual customer data & search trends.
Table of Contents
- Introduction
- Objective of the study
- Product Definition
- Market Segmentation
- Study Variables
- Research Methodology
- Secondary Data Points
- Companies Interviewed
- Primary Data Points
- Breakdown of Primary Interviews
- Secondary Data Points
- Executive Summary
- Market Dynamics
- Drivers
- Challenges
- Opportunity Assessment
- Recent Trends and Developments
- Policy and Regulatory Landscape
- India Cardiovascular Devices Market Overview (2021-2034)
- Market Size, By Value (in USD Billions)
- Market Share, By Product Type
- Diagnostic & Monitoring Devices
- Therapeutic & Surgical Devices
- Market Share, By Application
- Coronary Artery Disease
- Arrhythmia & Conduction Disorders
- Heart Failure & Cardiomyopathy
- Structural & Congenital Heart Defects
- Peripheral Vascular Disease
- Market Share, By End User
- Hospitals & Cardiac Centres
- Ambulatory Surgical Centres
- Cardiology/EP Clinics
- Home-care & Remote Monitoring Programs
- Market Share, By Region
- North India
- South India
- West India
- East India
- Market Share, By Competitors
- Competition Characteristics
- Revenue Shares
- India Diagnostic & Monitoring Devices Cardiovascular Devices Market Overview, 2021-2034F
- By Value (USD Million)
- By Application- Market Size & Forecast 2021-2034, USD Million
- By End User- Market Size & Forecast 2021-2034, USD Million
- India Therapeutic & Surgical Devices Cardiovascular Devices Market Overview, 2021-2034F
- By Value (USD Million)
- By Application- Market Size & Forecast 2021-2034, USD Million
- By End User- Market Size & Forecast 2021-2034, USD Million
- Competitive Outlook (Company Profile - Partial List)
- Medtronic plc
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- Abbott Laboratories
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- Boston Scientific Corporation
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- Edwards Lifesciences Corporation
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- Terumo Corporation
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- Meril Life Sciences Pvt. Ltd.
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- Sahajanand Medical Technologies Ltd.
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- Opto Circuits (India) Ltd.
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- Translumina Therapeutics LLP
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- Others
- Medtronic plc
- Contact Us & Disclaimer
Top Key Players & Market Share Outlook
- Medtronic plc
- Abbott Laboratories
- Boston Scientific Corporation
- Edwards Lifesciences Corporation
- Terumo Corporation
- Meril Life Sciences Pvt. Ltd.
- Sahajanand Medical Technologies Ltd.
- Opto Circuits (India) Ltd.
- Translumina Therapeutics LLP
- Others
Frequently Asked Questions






