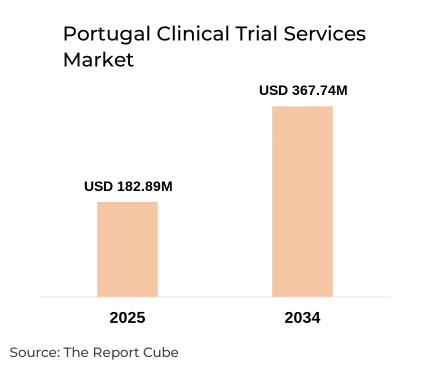
Build Your Custom Market Intelligence Report
Customize Your ReportPortugal Clinical Trial Services Market Statistics and Insights
Market Size Statistics
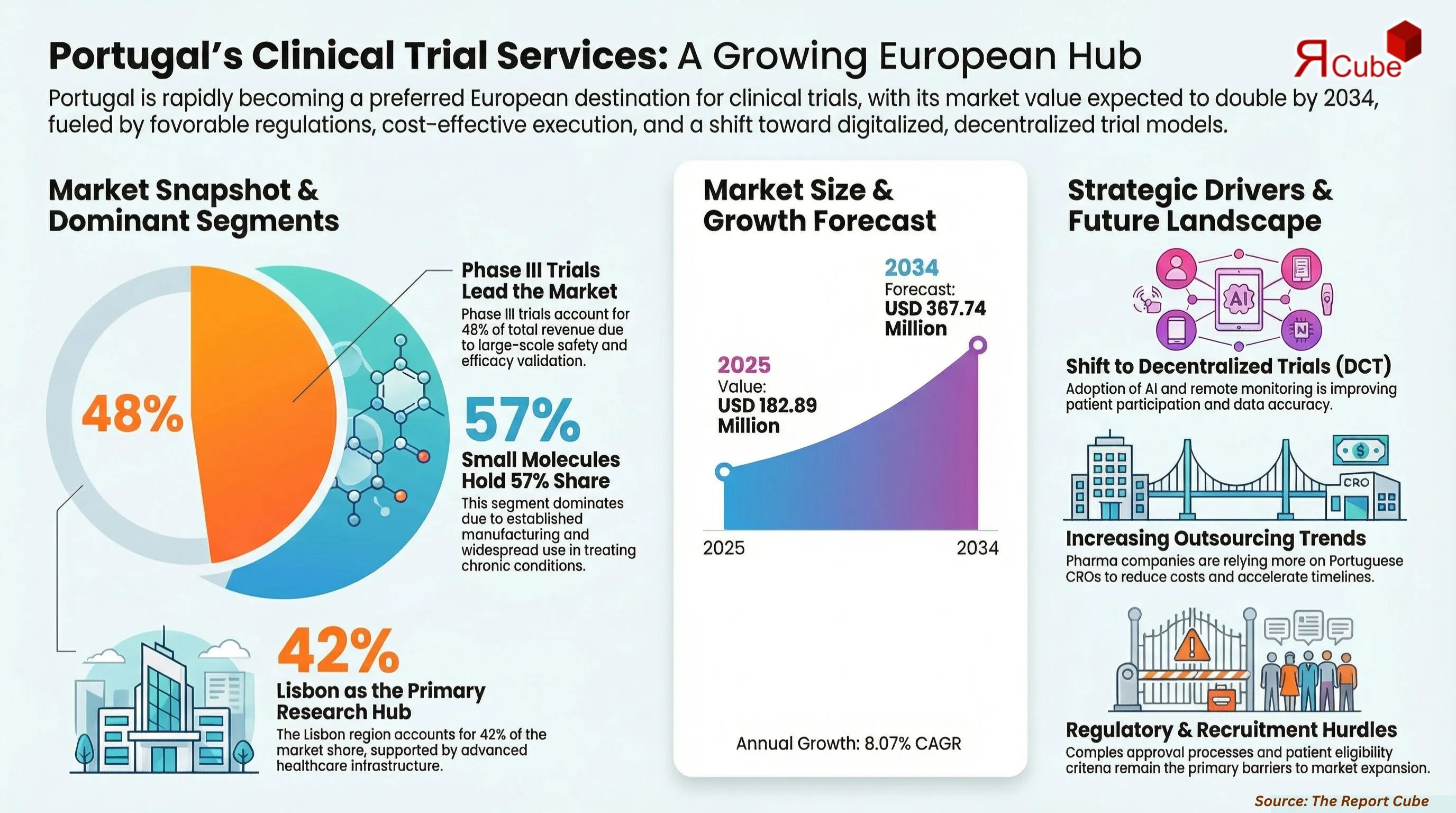
- Portugal Clinical Trial Services Market reached USD 182.89 million 2025
- Market projected to reach USD 367.74 million by 2034
- Market expected to grow at CAGR 8.07% during forecast period
By Phase
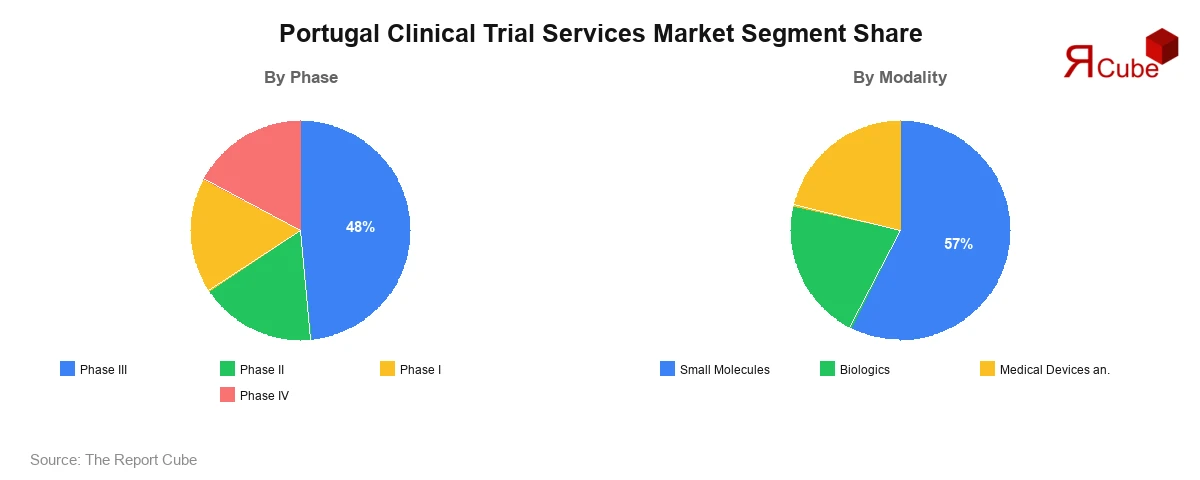
- Phase III segment holds dominant 48% share trials
Competitive Landscape
- More than 10 companies are actively engaged in producing Portugal Clinical Trial Services Market
- Top 5 companies acquired around 55% of the market share.
- BlueClinical, VectorB2B, Aibili, Evidenze Clinical Research Portugal, Scientific ToolBox Consulting, and others
By Modality
- Small molecules segment accounts for 57% driven demand
Portugal Clinical Trial Services Market Insights & Analysis
The Portugal Clinical Trial Services Market is anticipated to register a 8.07% CAGR during 2026-2034. The market size was valued at USD 182.89 million in 2025 and is projected to reach USD 367.74 million by 2034. The market is experiencing steady growth driven by Portugal’s increasing role as a preferred destination for clinical trials in Europe. Favorable regulatory frameworks, cost-effective trial execution, and access to diverse patient populations are attracting global pharmaceutical companies.
Government initiatives supporting research and innovation, along with EU funding programs, are strengthening clinical research infrastructure. Additionally, the presence of experienced contract research organizations (CROs) and skilled healthcare professionals is enhancing trial efficiency. The growing focus on precision medicine and personalized therapies is further driving demand for clinical trial services. Increasing collaborations between academic institutions and pharmaceutical companies are also contributing to market expansion.
Portugal Clinical Trial Services Market Dynamics
Key Market Driver: Increasing Clinical Research Investments and Outsourcing Trends
The rising trend of outsourcing clinical trials to specialized service providers is a key driver of the Portugal clinical trial services market. Pharmaceutical and biotechnology companies are increasingly relying on CROs to reduce operational costs and accelerate drug development timelines. Portugal offers competitive advantages, including lower trial costs compared to Western Europe and access to well-established healthcare systems. Additionally, increasing investments in research and development are boosting clinical trial activity. The country’s participation in multinational clinical studies is also expanding, further strengthening its position as a clinical research hub in Europe.
Major Industry Challenge: Regulatory Complexity and Patient Recruitment Barriers
Despite favorable conditions, regulatory complexity and patient recruitment challenges remain significant hurdles. Navigating approval processes and compliance requirements can be time-consuming, potentially delaying trial initiation. Additionally, recruiting suitable patients for clinical trials can be difficult due to limited awareness and strict eligibility criteria. Competition among trial sites for patient enrollment further intensifies this challenge. Ensuring patient retention throughout the study duration is also critical. Addressing these issues requires streamlined regulatory processes, improved patient engagement strategies, and enhanced collaboration between stakeholders in the clinical research ecosystem.
Emerging Trend Shaping Outlook: Growth of Decentralized Trials and Digital Health Integration
The adoption of decentralized clinical trials and digital health technologies is transforming the clinical trial landscape in Portugal. Remote monitoring, telemedicine, and wearable devices are enabling more flexible and patient-centric trial designs. These technologies improve patient participation, reduce dropout rates, and enhance data accuracy. Additionally, the integration of AI and data analytics is optimizing trial design, patient recruitment, and data management. The growing use of electronic data capture systems and cloud-based platforms is further improving efficiency. This trend is expected to drive innovation and improve the overall effectiveness of clinical trial services in Portugal.
Need Specific Data, Let’s Customize the Report for You
Request CustomizationPortugal Clinical Trial Services Market Segment-wise Analysis
By Phase
- Phase III
- Phase II
- Phase I
- Phase IV
The Phase III segment dominates the Portugal Clinical Trial Services Market, accounting for approximately 48% of total revenue share. This dominance is driven by the large-scale nature of Phase III trials, which involve extensive patient populations and significant investment. Pharmaceutical companies rely heavily on Phase III studies to validate the safety and efficacy of new drugs before regulatory approval. Phase II trials also contribute significantly, focusing on evaluating treatment effectiveness. Phase I trials are smaller but critical for assessing safety and dosage, while Phase IV trials are conducted post-approval to monitor long-term effects. The increasing number of late-stage clinical trials is supporting market growth.
By Modality
- Small Molecules
- Biologics
- Medical Devices and IVD
The small molecules segment leads the market with an estimated 57% share, driven by their widespread use in drug development and established manufacturing processes. Small molecule drugs are commonly used in treating various chronic and acute conditions, making them a key focus of clinical trials. Biologics are witnessing rapid growth due to increasing demand for targeted therapies and advancements in biotechnology. Medical devices and in vitro diagnostics (IVD) are also gaining traction, supported by technological innovation and growing demand for diagnostic solutions. The diversification of clinical research across modalities is contributing to overall market expansion.
Regional Projection of Portugal Clinical Trial Services Market
- Lisbon Region
- Northern Portugal
- Central Portugal
- Southern Portugal
Lisbon Region leads the market, accounting for approximately 42% share due to the presence of major hospitals, research institutions, and clinical trial centers. The region benefits from advanced healthcare infrastructure and strong collaboration between academia and industry. Northern Portugal also contributes significantly, supported by leading universities and healthcare facilities in cities like Porto. Central and Southern Portugal are gradually emerging as growth regions with increasing investments in healthcare and research infrastructure.
Portugal Clinical Trial Services Market: Recent Develpoment (2025)
- BlueClinical expanded clinical research operations enhancing trial management capabilities across multiple therapeutic areas in Portugal
- IQVIA strengthened partnerships with Portuguese healthcare institutions to accelerate clinical trial execution and data analytics services
- ICON introduced advanced digital trial solutions improving patient recruitment and monitoring efficiency across clinical studies
- Evidenze Clinical Research Portugal enhanced service offerings focusing on decentralized trials and innovative research methodologies
Portugal Clinical Trial Services Market Future Outlook (2034)
The Portugal Clinical Trial Services Market is expected to grow at a CAGR of 8.07%, reaching USD 367.74 million by 2034. Growth will be driven by increasing outsourcing of clinical trials, expansion of research infrastructure, and rising demand for innovative therapies. Technological advancements and AI-driven analytics will enhance trial efficiency and patient recruitment. The adoption of decentralized trials and digital health solutions will further transform the market. Additionally, strong government support and international collaborations will position Portugal as a key hub for clinical research in Europe.
Why Choose This Report?
- Provides a comprehensive overview of the overall market analysis, encompassing key trends, consumer behavior analysis, and risk assessment to support strategic decision-making.
- Provides accurate, up-to-date insights into market size, segmentation, and emerging opportunities, helping to minimize risk & capitalizing on growth.
- Gives deep understanding of target audience preferences, investment habits, and communication channels for enhanced product development & marketing effectiveness.
- Delivers competitive analysis & benchmarking, uncovering the strengths & weaknesses of market competitors to guide strategies.
- Consolidate comprehensive market intelligence, reducing reasoning & streamlining research efforts.
- Facilitates customized market segmentation & risk mitigation strategies, fine-tuned to the business objectives.
- Aids in identifying both market challenges & untapped opportunities within the industry to drive long-term business growth.
- Provides valuable information based on actual customer data & search trends.
Table of Contents
- Introduction
- Objective of the study
- Product Definition
- Market Segmentation
- Study Variables
- Research Methodology
- Secondary Data Points
- Companies Interviewed
- Primary Data Points
- Breakdown of Primary Interviews
- Secondary Data Points
- Executive Summary
- Market Dynamics
- Drivers
- Challenges
- Opportunity Assessment
- Recent Trends and Developments
- Policy and Regulatory Landscape
- Portugal Clinical Trial Services Market Overview (2021-2034)
- Market Size, By Value (in USD Billions)
- Market Share, By Service Type
- Clinical Trial Management and Monitoring
- Laboratory Services
- Clinical Trial Data Management Services
- Clinical Trial Supply and Logistic Services
- Consulting
- Patient Recruitment and Retention
- Medical Writing
- Safety and Pharmacovigilance
- Others
- Market Share, By Phase
- Phase III
- Phase II
- Phase I
- Phase IV
- Market Share, By Modality
- Small Molecules
- Biologics
- Medical Devices and IVD
- Market Share, By Therapeutic Area
- Oncology
- Neurology
- Respiratory Disorders
- Metabolic Disorders/Endocrinology
- Cardiovascular System Disorders
- Dermatology
- Gastrointestinal Diseases
- Immunological Disorders
- Infectious Diseases
- Psychiatry
- Ophthalmology
- Hematology
- Genitourinary and Women's Health
- Others
- Market Share, By Delivery Model
- Full-service Outsourcing (FSO) Model
- Functional Service Provider (FSP) Model
- Hybrid Model
- Market Share, By Study Design
- Interventional
- Observational
- Market Share, By End User
- Pharmaceutical and Biopharmaceutical Companies
- Medical Device Companies
- Academic Institutes
- Market Share, By Region
- Lisbon Region
- Northern Portugal
- Central Portugal
- Southern Portugal
- Market Share, By Competitors
- Competition Characteristics
- Revenue Shares
- Portugal Clinical Trial Management and Monitoring Clinical Trial Services Market Overview, 2021-2034F
- By Value (USD Million)
- By Phase- Market Size & Forecast 2021-2034, USD Million
- By Modality- Market Size & Forecast 2021-2034, USD Million
- By Therapeutic Area- Market Size & Forecast 2021-2034, USD Million
- By Delivery Model- Market Size & Forecast 2021-2034, USD Million
- By Study Design- Market Size & Forecast 2021-2034, USD Million
- By End User- Market Size & Forecast 2021-2034, USD Million
- Portugal Laboratory Services Clinical Trial Services Market Overview, 2021-2034F
- By Value (USD Million)
- By Phase- Market Size & Forecast 2021-2034, USD Million
- By Modality- Market Size & Forecast 2021-2034, USD Million
- By Therapeutic Area- Market Size & Forecast 2021-2034, USD Million
- By Delivery Model- Market Size & Forecast 2021-2034, USD Million
- By Study Design- Market Size & Forecast 2021-2034, USD Million
- By End User- Market Size & Forecast 2021-2034, USD Million
- Portugal Clinical Trial Data Management Services Clinical Trial Services Market Overview, 2021-2034F
- By Value (USD Million)
- By Phase- Market Size & Forecast 2021-2034, USD Million
- By Modality- Market Size & Forecast 2021-2034, USD Million
- By Therapeutic Area- Market Size & Forecast 2021-2034, USD Million
- By Delivery Model- Market Size & Forecast 2021-2034, USD Million
- By Study Design- Market Size & Forecast 2021-2034, USD Million
- By End User- Market Size & Forecast 2021-2034, USD Million
- Portugal Clinical Trial Supply and Logistic Services Clinical Trial Services Market Overview, 2021-2034F
- By Value (USD Million)
- By Phase- Market Size & Forecast 2021-2034, USD Million
- By Modality- Market Size & Forecast 2021-2034, USD Million
- By Therapeutic Area- Market Size & Forecast 2021-2034, USD Million
- By Delivery Model- Market Size & Forecast 2021-2034, USD Million
- By Study Design- Market Size & Forecast 2021-2034, USD Million
- By End User- Market Size & Forecast 2021-2034, USD Million
- Portugal Consulting Clinical Trial Services Market Overview, 2021-2034F
- By Value (USD Million)
- By Phase- Market Size & Forecast 2021-2034, USD Million
- By Modality- Market Size & Forecast 2021-2034, USD Million
- By Therapeutic Area- Market Size & Forecast 2021-2034, USD Million
- By Delivery Model- Market Size & Forecast 2021-2034, USD Million
- By Study Design- Market Size & Forecast 2021-2034, USD Million
- By End User- Market Size & Forecast 2021-2034, USD Million
- Portugal Patient Recruitment and Retention Clinical Trial Services Market Overview, 2021-2034F
- By Value (USD Million)
- By Phase- Market Size & Forecast 2021-2034, USD Million
- By Modality- Market Size & Forecast 2021-2034, USD Million
- By Therapeutic Area- Market Size & Forecast 2021-2034, USD Million
- By Delivery Model- Market Size & Forecast 2021-2034, USD Million
- By Study Design- Market Size & Forecast 2021-2034, USD Million
- By End User- Market Size & Forecast 2021-2034, USD Million
- Portugal Medical Writing Clinical Trial Services Market Overview, 2021-2034F
- By Value (USD Million)
- By Phase- Market Size & Forecast 2021-2034, USD Million
- By Modality- Market Size & Forecast 2021-2034, USD Million
- By Therapeutic Area- Market Size & Forecast 2021-2034, USD Million
- By Delivery Model- Market Size & Forecast 2021-2034, USD Million
- By Study Design- Market Size & Forecast 2021-2034, USD Million
- By End User- Market Size & Forecast 2021-2034, USD Million
- Portugal Safety and Pharmacovigilance Clinical Trial Services Market Overview, 2021-2034F
- By Value (USD Million)
- By Phase- Market Size & Forecast 2021-2034, USD Million
- By Modality- Market Size & Forecast 2021-2034, USD Million
- By Therapeutic Area- Market Size & Forecast 2021-2034, USD Million
- By Delivery Model- Market Size & Forecast 2021-2034, USD Million
- By Study Design- Market Size & Forecast 2021-2034, USD Million
- By End User- Market Size & Forecast 2021-2034, USD Million
- Portugal Others Clinical Trial Services Market Overview, 2021-2034F
- By Value (USD Million)
- By Phase- Market Size & Forecast 2021-2034, USD Million
- By Modality- Market Size & Forecast 2021-2034, USD Million
- By Therapeutic Area- Market Size & Forecast 2021-2034, USD Million
- By Delivery Model- Market Size & Forecast 2021-2034, USD Million
- By Study Design- Market Size & Forecast 2021-2034, USD Million
- By End User- Market Size & Forecast 2021-2034, USD Million
- Competitive Outlook (Company Profile - Partial List)
- BlueClinical
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- VectorB2B
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- Aibili
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- Evidenze Clinical Research Portugal
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- Scientific ToolBox Consulting
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- IQVIA
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- ICON
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- Others
- BlueClinical
- Contact Us & Disclaimer
Top Key Players & Market Share Outlook
- BlueClinical
- VectorB2B
- Aibili
- Evidenze Clinical Research Portugal
- Scientific ToolBox Consulting
- IQVIA
- ICON
- Others
Frequently Asked Questions






