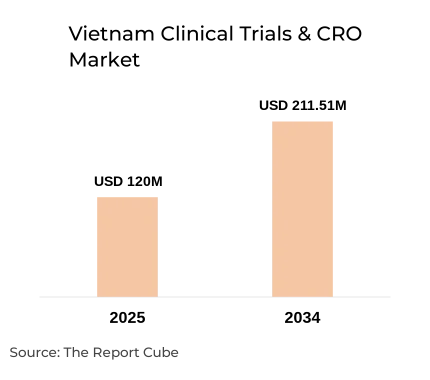
Build Your Custom Market Intelligence Report
Customize Your ReportVietnam Clinical Trials & CRO Market Key Highlights
By Trial Phase:
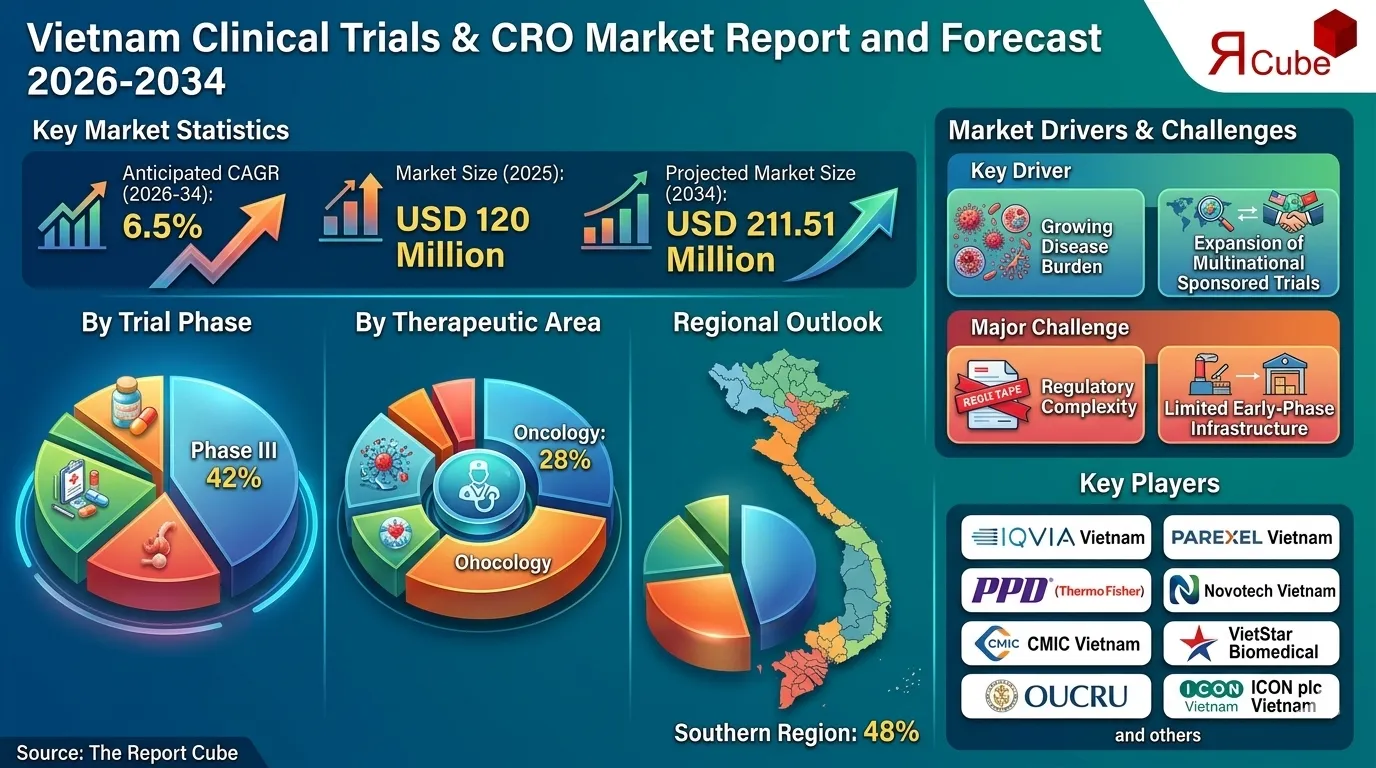
Phase III segment leads the market, with around 42% share.
By Therapeutic Area:
Oncology segment dominates the market, accounting for nearly 28% of total revenue.
Regional Outlook:
Southern region dominates the vietnam clinical trials & cro market, with around 48% share.
Government Initiatives & Policies
- Vietnam National Strategy for Pharmaceutical Industry Development to 2030, Vision 2045 (Updated 2025 Implementation Guidance): Encourages local clinical research capacity building, regulatory harmonization, and faster trial approvals.
- Ministry of Health Circular on Clinical Trial Governance and Digital Submission (2025 update): Expands centralized ethics review, strengthens GCP compliance, and accelerates study approval timelines.
Vietnam Clinical Trials & CRO Market Insights & Analysis
The Vietnam Clinical Trials & CRO Market is anticipated to register a CAGR: 6.5% during 2026-34. The market size was valued at USD 120 million in 2025 and is projected to reach USD 211.51 million by 2034. Vietnam is emerging as a strategic destination for Asia-Pacific clinical research, supported by cost efficiency, a large treatment-naïve patient pool, and improving regulatory clarity.
Vietnam’s population of over 100 million, combined with rising prevalence of non-communicable diseases such as cancer, diabetes, and cardiovascular disorders, creates strong demand for advanced therapies and structured clinical research programs. Oncology incidence in Vietnam has increased by approximately 8-10% annually over the past five years, positioning oncology trials as a dominant therapeutic focus. Sponsors are increasingly leveraging Vietnam for multi-center Phase III trials due to competitive site costs that are 30-40% lower than in developed Asian markets such as South Korea or Singapore.
Regulatory modernization is another structural growth lever. The Ministry of Health has streamlined ethics committee reviews and introduced partial digital dossier submission processes, reducing average trial approval timelines from 12 months to approximately 6-8 months in major hospitals. This improvement enhances Vietnam’s attractiveness for global sponsors seeking faster site activation in Southeast Asia.
In addition, global CROs including IQVIA Vietnam, Parexel Vietnam, ICON plc Vietnam, and PPD (Thermo Fisher) are expanding local monitoring and data management teams. Domestic players such as VietStar Biomedical and Nanogen Pharma are strengthening capabilities in vaccine trials and biosimilar research. Academic institutions such as OUCRU and leading medical universities are also supporting infectious disease platform trials, further elevating Vietnam’s global clinical research profile.
Vietnam Clinical Trials & CRO Market Dynamics
Key Driver: Growing Disease Burden and Expansion of Multinational Sponsored Trials
The steady rise in chronic disease prevalence is driving sponsor interest in Vietnam. Cardiovascular diseases account for nearly 31% of total mortality nationwide, while cancer cases exceed 180,000 new diagnoses annually. Pharmaceutical and biotechnology companies are increasing investments in late-phase trials to support regional drug approvals. Vietnam’s treatment-naïve populations enable rapid patient recruitment, reducing enrollment timelines by 15-20% compared to more saturated markets. Furthermore, the government’s emphasis on strengthening hospital research units has expanded the number of GCP-compliant sites, particularly in Ho Chi Minh City and Hanoi. As multinational sponsors diversify supply chains and research footprints, Vietnam’s strategic location within ASEAN enhances its role in regional Phase II and III trials.
Book your FREE 30-minute expert consultation today
Contact UsIndustry Trends: Rise of Oncology Precision Medicine and Decentralized Trial Models
Precision oncology and biomarker-driven trials are reshaping Vietnam’s CRO landscape. Companies such as Gene Solutions are advancing circulating tumor DNA (ctDNA) diagnostics to support companion diagnostic research. Integration of genomic sequencing and AI-based analytics is improving patient stratification and enhancing trial efficiency. Decentralized clinical trial components, including remote patient monitoring and electronic data capture systems, are gradually being adopted by urban hospitals. Hybrid trial models that combine on-site visits with telemedicine consultations are reducing dropout rates and improving retention. Data management and biostatistics services are experiencing above-average growth as sponsors demand real-time analytics and regional data harmonization across ASEAN markets.
Major Challenge: Regulatory Complexity and Limited Early-Phase Infrastructure
While regulatory timelines have improved, early-phase (Phase I) trial infrastructure remains limited. Specialized Phase I units with intensive pharmacokinetic monitoring capabilities are concentrated in a few urban hospitals. This restricts Vietnam’s competitiveness in first-in-human studies compared to regional peers. Ethical review processes, although faster, can still vary between institutions, leading to administrative inconsistencies. Moreover, language barriers and documentation requirements may increase coordination efforts for international sponsors. Investment in dedicated research wards, standardized IRB frameworks, and expanded investigator training is essential to strengthen early-phase trial competitiveness.
Opportunity: Positioning Vietnam as a Regional Hub for Infectious Disease and Vaccine Trials
Vietnam’s experience in infectious disease surveillance, particularly through partnerships with global health organizations, creates opportunities for platform trials and vaccine development studies. Institutions such as OUCRU have demonstrated advanced genomic sequencing capacity and outbreak-response capabilities. Southeast Asia remains a hotspot for emerging infections, making Vietnam strategically valuable for global health research. Expansion of laboratory services, pharmacovigilance systems, and regulatory harmonization with ASEAN standards could increase inbound sponsor activity by 20-25% over the forecast period. Growth in medical device trials and real-world evidence studies also provides diversification beyond pharmaceutical research.
Vietnam Clinical Trials & CRO Market Segment-wise Analysis
By Trial Phase:
- Phase I
- Phase II
- Phase III
- Phase IV
Phase III accounts for approximately 42% of total market revenue in 2025, reflecting Vietnam’s strong position in late-stage multinational trials. Large patient pools enable efficient enrollment across oncology, cardiology, and infectious diseases. Phase III trials often involve 300-1,000 participants, requiring extensive site management and monitoring services, which boosts CRO revenue streams. Global pharmaceutical companies prefer Vietnam for cost-effective multicenter studies prior to ASEAN regulatory submissions. Phase IV post-marketing studies are also expanding, particularly in oncology biologics and diabetes therapies, as sponsors gather real-world safety and effectiveness data.
By Therapeutic Area:
- Oncology
- Cardiology
- Infectious Diseases
- Central Nervous System
- Endocrinology and Metabolic Disorders
- Respiratory Diseases
- Others
Oncology leads with nearly 28% share, driven by increasing cancer incidence and introduction of targeted therapies. Lung, liver, and breast cancers are among the most studied indications. Precision diagnostics integration, including genomic profiling, supports biomarker-based enrollment. Cardiology follows closely due to high cardiovascular disease prevalence. Infectious disease trials maintain relevance, supported by Vietnam’s strong epidemiological research networks. Growth in metabolic disorder studies is accelerating as diabetes prevalence approaches 7% of the adult population, increasing demand for innovative therapeutics and combination regimens.
Regional Projection of Vietnam Clinical Trials & CRO Industry
- Southern
- Northern
- Central
- Others
The Southern region commands approximately 48% share, led by Ho Chi Minh City’s concentration of tertiary hospitals and research institutes. Major CRO offices and pharmaceutical headquarters are located here, facilitating sponsor coordination. Northern Vietnam, particularly Hanoi, follows with strong academic research institutions and public hospitals conducting infectious disease and vaccine studies. Central Vietnam is gradually expanding capabilities, supported by regional medical universities. Infrastructure concentration in urban centers directly correlates with higher trial density and CRO service demand.
Vietnam Clinical Trials & CRO Market Recent Developments
- 2025: AstraZeneca Vietnam partnered with the Ministry of Health to launch the “Vietnam Lung Health Program 2025-2027,” strengthening nationwide respiratory screening and supporting long-term clinical research ecosystem development.
- 2025: Gene Solutions hosted K-CONNECT APAC 2025 in Da Nang, showcasing AI-driven ctDNA-MRD and multi-cancer early detection platforms to expand precision oncology clinical collaborations.
- 2025: OUCRU signed an MoU with Vietnam’s Health Strategy and Policy Institute to promote joint research and evidence-based policy engagement, reinforcing clinical research integration nationwide.
Need insights for a specific region within this market?
Request Regional DataWhy Choose This Report?
- Provides a comprehensive overview of the overall market analysis, encompassing key trends, consumer behavior analysis, and risk assessment to support strategic decision-making.
- Provides accurate, up-to-date insights into market size, segmentation, and emerging opportunities, helping to minimize risk & capitalizing on growth.
- Gives deep understanding of target audience preferences, investment habits, and communication channels for enhanced product development & marketing effectiveness.
- Delivers competitive analysis & benchmarking, uncovering the strengths & weaknesses of market competitors to guide strategies.
- Consolidate comprehensive market intelligence, reducing reasoning & streamlining research efforts.
- Facilitates customized market segmentation & risk mitigation strategies, fine-tuned to the business objectives.
- Aids in identifying both market challenges & untapped opportunities within the industry to drive long-term business growth.
- Provides valuable information based on actual customer data & search trends.
Table of Contents
- Introduction
- Objective of the Study
- Product and Category Definition
- Market Segmentation
- Study Variables
- Research Methodology
- Secondary Data Points
- Breakdown of Secondary Sources
- Primary Data Points
- Breakdown of Primary Interviews
- Secondary Data Points
- Executive Summary
- Market Dynamics
- Drivers
- Challenges
- Opportunity Assessment
- Recent Trends and Developments
- Regulatory and Policy Landscape
- Vietnam Clinical Trials & CRO Market Overview (2021-2034)
- Market Size, By Value (USD Million)
- Market Share, By Trial Phase
- Phase I
- Phase II
- Phase III
- Phase IV
- Market Share, By Therapeutic Area
- Oncology
- Cardiology
- Infectious Diseases
- Central Nervous System
- Endocrinology and Metabolic Disorders
- Respiratory Diseases
- Others
- Market Share, By Sponsor Type
- Pharmaceutical and Biotechnology Companies
- Medical Device Companies
- Academic and Research Institutions
- Government and Non-profit Organizations
- Market Share, By Service Type
- Clinical Trial Management
- Site Management and Monitoring
- Project Management
- Data Management and Biostatistics
- Regulatory Affairs
- Medical Writing
- Safety and Pharmacovigilance
- Laboratory Services
- Others
- Market Share, By Study Design
- Interventional Studies
- Observational Studies
- Expanded Access and Other Designs
- Market Share, By End User
- Hospitals
- Specialty Clinics
- Academic Medical Centers
- Contract Research Organizations
- Others
- Market Share, By Region
- Northern
- Central
- Southern
- Others
- Market Share, By Company
- Revenue Shares and Analysis
- Competitive Landscape
- Northern Vietnam Clinical Trials & CRO Market
- Market Size, By Value (USD Million)
- Market Share, By Trial Phase
- Market Share, By Therapeutic Area
- Market Share, By Sponsor Type
- Market Share, By Service Type
- Market Share, By Study Design
- Market Share, By End User
- Central Vietnam Clinical Trials & CRO Market
- Market Size, By Value (USD Million)
- Market Share, By Trial Phase
- Market Share, By Therapeutic Area
- Market Share, By Sponsor Type
- Market Share, By Service Type
- Market Share, By Study Design
- Market Share, By End User
- Southern Vietnam Clinical Trials & CRO Market
- Market Size, By Value (USD Million)
- Market Share, By Trial Phase
- Market Share, By Therapeutic Area
- Market Share, By Sponsor Type
- Market Share, By Service Type
- Market Share, By Study Design
- Market Share, By End User
- Competitive Outlook and Company Profiles
- IQVIA Vietnam
- Company Overview
- Product Portfolio
- Strategic Alliances/Partnerships
- Recent Developments
- Parexel Vietnam
- Company Overview
- Product Portfolio
- Strategic Alliances/Partnerships
- Recent Developments
- PPD (Thermo Fisher)
- Company Overview
- Product Portfolio
- Strategic Alliances/Partnerships
- Recent Developments
- Novotech Vietnam
- Company Overview
- Product Portfolio
- Strategic Alliances/Partnerships
- Recent Developments
- CMIC Vietnam
- Company Overview
- Product Portfolio
- Strategic Alliances/Partnerships
- Recent Developments
- VietStar Biomedical
- Company Overview
- Product Portfolio
- Strategic Alliances/Partnerships
- Recent Developments
- OUCRU
- Company Overview
- Product Portfolio
- Strategic Alliances/Partnerships
- Recent Developments
- ICON plc Vietnam
- Company Overview
- Product Portfolio
- Strategic Alliances/Partnerships
- Recent Developments
- SGS Vietnam
- Company Overview
- Product Portfolio
- Strategic Alliances/Partnerships
- Recent Developments
- Nanogen Pharma
- Company Overview
- Product Portfolio
- Strategic Alliances/Partnerships
- Recent Developments
- Dokumeds Vietnam
- Company Overview
- Product Portfolio
- Strategic Alliances/Partnerships
- Recent Developments
- Gene Solutions
- Company Overview
- Product Portfolio
- Strategic Alliances/Partnerships
- Recent Developments
- Syneos Health
- Company Overview
- Product Portfolio
- Strategic Alliances/Partnerships
- Recent Developments
- Fortrea Vietnam
- Company Overview
- Product Portfolio
- Strategic Alliances/Partnerships
- Recent Developments
- AstraZeneca Vietnam
- Company Overview
- Product Portfolio
- Strategic Alliances/Partnerships
- Recent Developments
- GSK Pharma Vietnam
- Company Overview
- Product Portfolio
- Strategic Alliances/Partnerships
- Recent Developments
- Sanofi Vietnam
- Company Overview
- Product Portfolio
- Strategic Alliances/Partnerships
- Recent Developments
- Others
- IQVIA Vietnam
- Contact Us and Disclaimer
Top Key Players & Market Share Outlook
- IQVIA Vietnam
- Parexel Vietnam
- PPD (Thermo Fisher)
- Novotech Vietnam
- CMIC Vietnam
- VietStar Biomedical
- OUCRU
- ICON plc Vietnam
- SGS Vietnam
- Nanogen Pharma
- Dokumeds Vietnam
- Gene Solutions
- Syneos Health
- Fortrea Vietnam
- AstraZeneca Vietnam
- GSK Pharma Vietnam
- Sanofi Vietnam
Frequently Asked Questions






