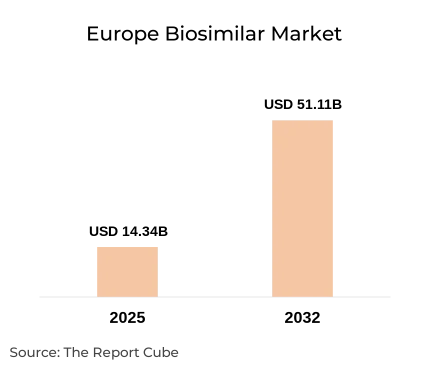
Build Your Custom Market Intelligence Report
Customize Your ReportEurope Biosimilar Market Insights & Analysis
The Europe Biosimilar Market is anticipated to register a CAGR of around 19.91% during the forecast period, 2026–2032. The market was valued at approximately USD 14.34 billion in 2025, and is projected to reach nearly USD 51.11 billion by 2032. The industry progression is instigated by mounting demand for reasonable biologics, growing chronic disease occurrence, and patent expirations of important reference biologics. Also, biosimilar products, which are biologic medical products largely similar to already approved branded biologics, have gained eminence owing to their cost-effectiveness & clinical efficiency.
Moreover, major drivers prompting the Europe Biosimilar Market include strong regulatory support from the European Medicines Agency (EMA), increasing pressure on healthcare budgets, and larger physician & patient acceptance. Also, recent initiatives like the EU’s Pharmaceutical Strategy to enhance access to inexpensive medicines & incentives for biosimilar production have augmented industry growth.
Additionally, leading biosimilar companies in Europe, such as Pfizer, Novartis (via Sandoz), and Samsung Bioepis, are considerably investing in biosimilar pipelines, motivating innovation & competitive pricing. For instance, the launch of Pfizer's biosimilar ustekinumab in several European nations in 2025 has augmented the number of autoimmune disease therapy possibilities. Celltrion Inc. further solidified its leadership in autoimmune treatments in 2025 with the introduction of CT-P43, a biosimilar of ustekinumab.
Furthermore, cooperative collaborations like the 2025 alliance between Organon & Samsung Bioepis to surge biosimilar distribution across Europe are key instances of strategic expansion. Also, the aim of government healthcare reforms & regulations, specifically in Germany & the UK, is to encourage the usage of biosimilars through substitution laws & tender procedures.
Consequently, the Europe Biosimilar Market appears to have a positive outlook, owing to innovation, supportive regulations, and increasing biosimilar adoption. Further, as patent cliffs draw near & the requirement for reasonably priced biologics rises, biosimilars are presumed to be fundamental in altering the European healthcare sector, thus enhancing the market share in the following years.
Europe Biosimilar Market Upgrades & Recent Developments
2025:
- Pfizer Inc. declared the introduction of ZIRABEV (a biosimilar of bevacizumab) in Germany & France, increasing its oncology biosimilar portfolio & accessibility across the European region.
- Celltrion Inc. introduced CT-P43, a biosimilar referencing ustekinumab, for autoimmune disease treatment, in leading European markets, strengthening its market share & biosimilar innovation headship.
Europe Biosimilar Market Dynamics
-
Driver: Patent Expirations of Major Biologics to Drive Market Growth
One of the leading driving forces in the Europe Biosimilar Industry is the patent expiration of blockbuster biologics, including Remicade (infliximab), Humira (adalimumab), and Herceptin (trastuzumab). Moreover, these expirations generate prospects for biosimilar producers to launch cost-effective substitutes, therefore decreasing treatment costs & expanding patient access. Also, regulatory clarity from the EMA further inspires companies to accelerate biosimilar development & launch, thus driving market demand.

Book your FREE 30-minute expert consultation today
Contact Us-
Challenge: Physician & Patient Awareness to Impede the Industry Expansion
Though biosimilars have acknowledged regulatory authorization, certain patient & physician groups remain uncertain about their safety & efficiency in comparison to original biologics. Moreover, this opposition might make it more challenging to prescribe & enter new markets. Further, even though confidence-boosting educational initiatives & experiential data are supportive, breaking through deeply ingrained brand loyalty is still demanding, thus hampering the market growth.
-
Opportunity: Rising Demand for Reasonable Biologics to Open New Avenues
The increasing occurrence of chronic diseases like cancer, diabetes, and autoimmune disorders has elevated the demand for biologic therapies. Nevertheless, the higher price of originator biologics restricts accessibility. Also, biosimilars provide a cost-efficient solution, providing a substantial growth opportunity for market companies aiming on biosimilar products in Europe, predominantly in publicly funded healthcare systems looking for cost containment.
-
Trend: Strategic Alliances & Co-Development Agreements to be Trending
The Europe Biosimilar Industry is observing a spike in partnerships among pharmaceutical companies to share R&D expenses & expand distribution. Also, recent instances such as Sanofi’s strategic association with Samsung Bioepis to co-develop biosimilars for oncology & immunology segments, promoting innovation & reforming market entry in the European countries..
Europe Biosimilar Market Segment-Wise Analysis
By Molecule:
- Infliximab
- Insulin Glargine
- Epoetin Alfa
- Etanercept
- Filgrastim
- Somatropin
- Rituximab
- Follitropin Alfa
- Adalimumab
Adalimumab captures the dominant market share across the Europe Biosimilar Market. A monoclonal antibody termed adalimumab is deployed to treat autoimmune diseases like Crohn's disease & rheumatoid arthritis. Also, several biosimilar copies of Humira, including Amgevita (Amgen) & Hyrimoz (Sandoz), have led the market since the drug's 2018 patent expired. Also, adalimumab's dominance is a result of its varied therapeutic usage & high deployment rates across Europe.
Moreover, its acceptance is further stimulated by government-backed switching programs & tender systems in countries such as the UK & Germany. Additionally, market growth has been assessed by decreased costs & enhanced accessibility brought about by the heightened competition.
By Indication:
- Autoimmune Diseases
- Blood Disorder
- Diabetes
- Oncology
- Growth Deficiency
- Female Infertility
The Oncology segment remains the leading segment in the Europe Biosimilar Market, driven by the increasing incidence of cancer & the high price of oncology biologics. Transtuzumab, bevacizumab, and rituximab biosimilars have largely expanded the industry by offering reasonably priced cancer treatments. Also, the strong acceptance of biosimilars in cancer could be attributed to the embracing of national frameworks by nations such as France & Italy. Furthermore, the segment’s market reach & affordability are further elevated by the coordinated procurement of biosimilars, which is supported by hospital-based distribution channels in oncology care.
Country Projection of the Europe Biosimilar Industry
The Europe Biosimilar Market is geographically diversified, covering:
- Italy
- Germany
- The UK
- France
- Spain
- Rest of Europe
Germany dominates the Europe Biosimilar Market, accounting for the potential market share in the forecast years. The country’s well-established biosimilar approval processes, advanced reimbursement policies, and supportive healthcare infrastructure are the causes for this. Further, owing to its competitive tendering approaches and early implementation of biosimilar-friendly policies, Germany has become a prevalent launch location for big companies. Also, businesses with solid biosimilar activities in Germany include Pfizer Inc. & Novartis AG (Sandoz).
Moreover, to surge adoption, the country has also established national goals for physician incentives for biosimilar acceptance. Germany also sets standards for the size of the Biosimilar Market globally, and serves as a model for other EU nations, having a significant impact on the market share in Europe as a whole.
Top Companies in Europe Biosimilar Market
Ask Our Lead Analyst a Question About This Report
Contact UsWhat Does Our Europe Biosimilar Market Research Study Entail?
- The Europe Biosimilar Market Research Report highlights the forecast growth rate (CAGR) by anticipating the market size and share.
- The market analysis puts light upon the primary industry trends, driving aspects, potential opportunities, growth challenges, and other major factors.
- The Europe Biosimilar Market Research Report entails details about the most critical shifts in market share in the prominent regions.
- Considering the statistics & the developments by the primary market competitors, our report also strives to demonstrate the most sought-after strategies of the key players.
Table of Contents
- Introduction
- Objective of the study
- Product Definition
- Market Segmentation
- Study Variables
- Research Methodology
- Secondary Data Points
- Companies Interviewed
- Primary Data Points
- Break Down of Primary Interviews
- Secondary Data Points
- Executive Summary
- Market Dynamics
- Drivers
- Challenges
- Opportunity Assessment
- Recent Trends and Developments
- Policy and Regulatory Landscape
- Europe Europe Market Overview (2020-2032)
- Market Size, By Value (in USD Billions)
- Market Share, By Molecule
- Infliximab
- Insulin Glargine
- Epoetin Alfa
- Etanercept
- Filgrastim
- Somatropin
- Rituximab
- Follitropin Alfa
- Adalimumab
- Market Share, By Indication
- Autoimmune Diseases
- Blood Disorder
- Diabetes
- Oncology
- Growth Deficiency
- Female Infertility
- Market Share, By Manufacturing Type
- In-house Manufacturing
- Contract Manufacturing
- Market Share, By Country
- Italy
- Germany
- The UK
- France
- Spain
- Rest of Europe
- Market Share, By Company
- Revenue Shares
- Competition Characteristics
- Italy Europe Market Overview, 2020-2032F
- Market Size, By Value (in USD Billions)
- By Molecule
- By Indication
- By Manufacturing Type
- Germany Europe Market Overview, 2020-2032F
- Market Size, By Value (in USD Billions)
- By Molecule
- By Indication
- By Manufacturing Type
- The UK Europe Market Overview, 2020-2032F
- Market Size, By Value (in USD Billions)
- By Molecule
- By Indication
- By Manufacturing Type
- France Europe Market Overview, 2020-2032F
- Market Size, By Value (in USD Billions)
- By Molecule
- By Indication
- By Manufacturing Type
- Spain Europe Market Overview, 2020-2032F
- Market Size, By Value (in USD Billions)
- By Molecule
- By Indication
- By Manufacturing Type
- Competitive Outlook (Company Profile - Partila List)
- Pfizer Inc.
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- Celltrion Inc.
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- Novartis AG
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- Amgen Inc.
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- Eli Lilly and Company
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- Samsung Bioepis
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- Sanofi SA
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- Dr. Reddy’s Laboratories Ltd.
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- Boehringer Ingelheim
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- Others
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- Pfizer Inc.
- Contact Us & Disclaimer
Top Key Players & Market Share Outlook
- Pfizer Inc.
- Celltrion Inc.
- Novartis AG
- Amgen Inc.
- Eli Lilly and Company
- Samsung Bioepis
- Sanofi SA
- Dr. Reddy’s Laboratories Ltd.
- Boehringer Ingelheim
- Others
Frequently Asked Questions






