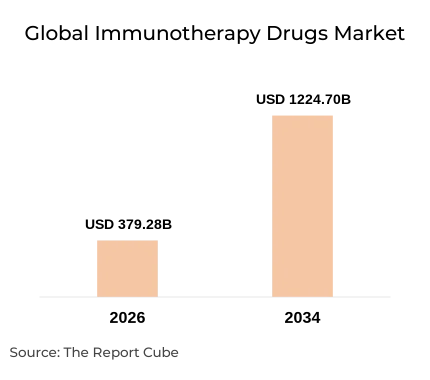
Build Your Custom Market Intelligence Report
Customize Your ReportKey Takeaways
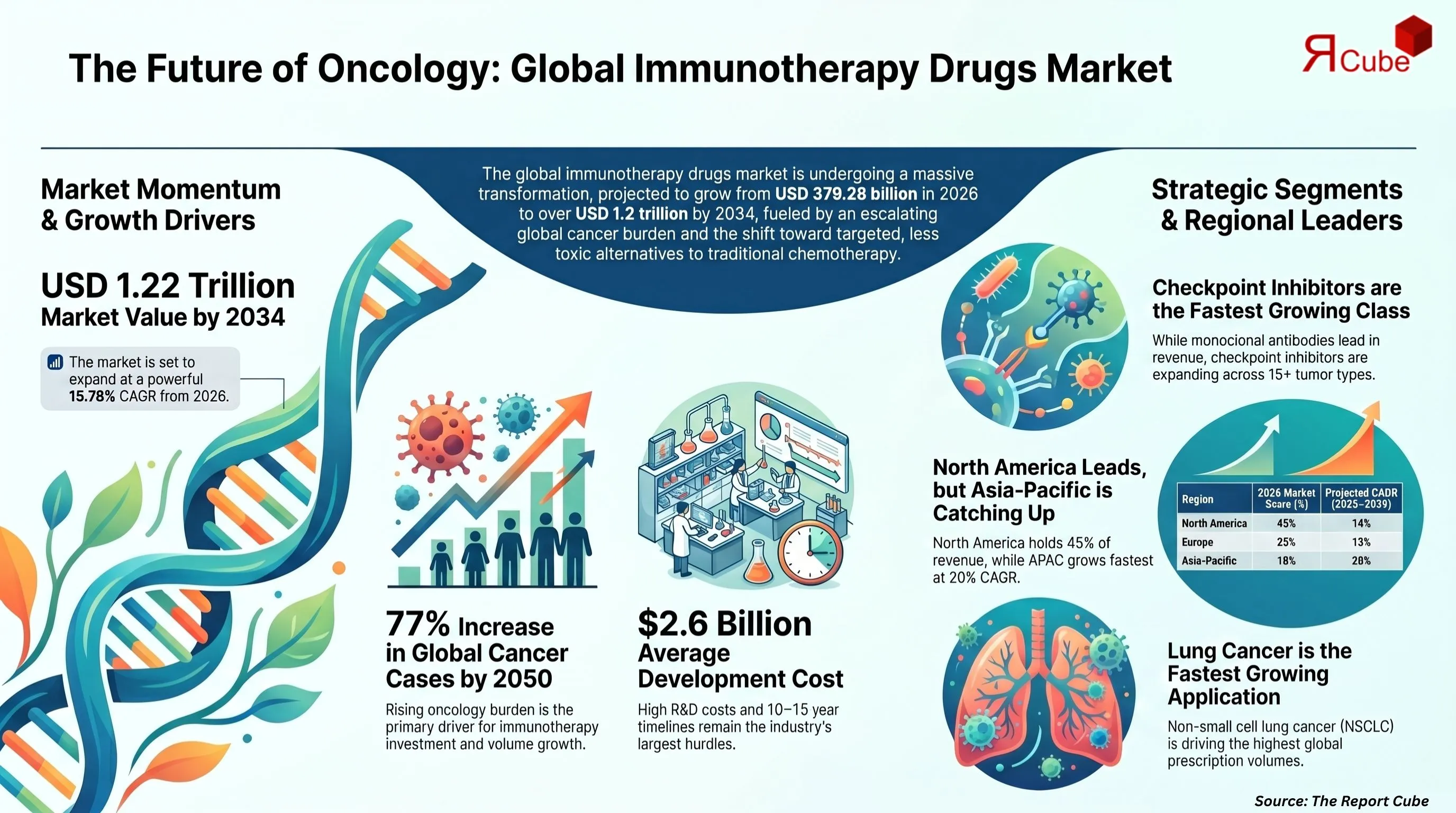
- Global immunotherapy drugs market projected to grow from USD 379.28 Billion (2026) to USD 1,224.70 Billion (2034)
- Strong growth trajectory with a CAGR of 15.78% (2026–2034)
- Monoclonal antibodies currently dominate the market by revenue
- Checkpoint inhibitors identified as the fastest-growing drug class
- Blood cancer remains the leading application segment
- Lung cancer is the fastest-growing application area
- North America leads the market with approximately 45% share
- Asia-Pacific emerges as the fastest-growing region, expanding at around 20% CAGR
Global Immunotherapy Drugs Market Insights & Analysis
Cancer remains the second leading cause of death globally. Standard treatments such as chemotherapy cause systemic toxicity that limits patient tolerance and outcomes, creating urgent demand for safer, targeted alternatives. With over 20 million new cancer cases diagnosed worldwide in 2022 (WHO, 2023), the gap between treatment demand and effective targeted therapy has reached a critical scale.
The global immunotherapy drugs market is projected to surge from USD 379.28 billion in 2026 to USD 1,224.70 billion by 2034, expanding at an impressive CAGR of 15.78%. This trajectory is driven by rising oncology prevalence, accelerating R&D investment, and a broadening biosimilar pipeline. Regulatory milestones - specifically FDA accelerated approvals for checkpoint inhibitors including PD-1/PD-L1 blockade agents - have fundamentally reshaped treatment protocols across multiple tumour types.
Among the key players in the oncology drugs market, Merck & Co., Bristol-Myers Squibb, F. Hoffmann-La Roche Ltd, AstraZeneca, and Novartis AG control the largest revenue shares in checkpoint inhibitor and monoclonal antibody segments. Patent cliffs for first-generation biologics between 2026 and 2029 are intensifying biosimilar competition, pressuring margins while simultaneously expanding patient access.
Global Immunotherapy Drugs Market Dynamics
Immunotherapy drugs occupy a market where clinical efficacy alone does not determine commercial success - reimbursement timelines, manufacturing scalability, and adverse-event profiles all shape uptake. Of the 100+ immunotherapy agents approved globally as of 2024, fewer than 40% achieve broad payer coverage within three years of approval (IQVIA Institute, 2023).
Key Market Driver: Escalating Global Cancer Burden and Unmet Therapeutic Need
Conventional chemotherapy and radiation offer incomplete responses for many solid tumours, leaving patients with recurrent disease and diminishing treatment options. The International Agency for Research on Cancer (IARC) projects 35 million new cancer cases annually by 2050 - a 77% increase from 2022 levels. This escalating oncology burden is the primary structural driver behind immunotherapy drug investment and market volume growth through 2034.
Rising incidence of lung, blood, and melanoma cancers - the three highest-revenue indication categories for immunotherapy - is converting latent patients into active treatment candidates. Government oncology screening programmes, including the US Cancer Moonshot initiative and EU4Health, are expanding diagnosis rates. A measurable correlation exists between increased early-stage diagnosis and checkpoint inhibitors market prescription rates, as earlier-stage disease responds better to immune-based therapies.
Major Industry Challenge: High Development Costs and Reimbursement Barriers
Immunotherapy drug development requires 10–15 years from discovery to approval and an average investment of USD 2.6 billion per approved asset (Tufts Center for the Study of Drug Development, 2022). Even post-approval, payer acceptance in mid-income markets takes 2–4 years, delaying revenue recognition and limiting patient access in markets representing 40%+ of future volume growth.
The average cost per course of checkpoint inhibitor therapy ranges from USD 100,000 to USD 200,000 per patient annually (JAMA Oncology, 2023). Health Technology Assessment (HTA) rejections and conditional approvals in EU member states delay commercial rollout by an average of 18 months beyond EMA approval. However, the biologic patent expiry window of 2026–2030 presents a structural opportunity to widen access without proportional cost increases.
Need Specific Data, Let’s Customize the Report for You
Request CustomizationEmerging Trend: Combination Immunotherapy Protocols and Personalised Medicine
Single-agent immunotherapy achieves durable responses in only 20–40% of treated patients, depending on tumour type and PD-L1 expression levels. This efficacy ceiling is pushing oncologists and developers toward combination protocols - pairing checkpoint inhibitors with monoclonal antibodies, chemotherapy, or CAR-T therapies.
FDA-approved combination regimens - including nivolumab plus ipilimumab for non-small cell lung cancer (NSCLC) and pembrolizumab plus chemotherapy for triple-negative breast cancer - have established combination protocols as the new standard-of-care benchmark. Biomarker testing across PD-L1, tumour mutational burden (TMB), and MSI-H markers is expanding, with Illumina and QIAGEN scaling companion diagnostics in emerging markets. Patient stratification through the precision oncology market reduces trial costs and improves approval probability by directing drugs to responsive subpopulations.
Emerging Opportunity: Immunotherapy Expansion in Asia-Pacific and Emerging Markets
Asia-Pacific accounted for less than 18% of global immunotherapy drug revenue in 2024, despite hosting over 50% of the world's cancer patients. Structural barriers - fragmented reimbursement, low biosimilar awareness, and limited oncology infrastructure - have suppressed market penetration far below clinical demand.
China's National Drug Administration (NMPA) has instituted fast-track approvals for PD-1 inhibitors from domestic manufacturers including BeiGene and Hengrui. India's Pharma Vision 2047 biosimilar production targets are reducing therapy costs across regional markets. South Korea and Japan stand as early adopters of cell-based immunotherapy, with established reimbursement frameworks supporting uptake. For a broader view of regional oncology spend, see the Asia-Pacific oncology drugs market analysis.
Global Immunotherapy Drugs Market - Segment-wise Analysis
By Drug Type
Drug type determines manufacturing complexity, pricing power, and indication breadth. The monoclonal antibodies market currently leads all drug classes. Key products - trastuzumab (Herceptin, Roche), rituximab (Rituxan), and bevacizumab - face mounting biosimilar pressure post-2026. Checkpoint inhibitors represent the fastest-growing class: pembrolizumab (Keytruda, Merck) and nivolumab (Opdivo, Bristol-Myers Squibb) now hold approved indications across 15+ tumour types.
|
Drug Type |
Est. 2026 Share (%) |
2034 Growth Outlook |
Biosimilar Risk |
Key Players |
|---|---|---|---|---|
|
Monoclonal Antibodies |
38% |
Steady |
High (2026–2029) |
Roche, AbbVie, Amgen |
|
Checkpoint Inhibitors |
22% |
Fastest-growing |
Low (patents to 2031+) |
Merck, BMS, AstraZeneca |
|
Vaccines (Therapeutic) |
18% |
High |
Low |
Merck, Sanofi, Pfizer |
|
Interferons |
12% |
Moderate–declining |
Medium |
Sanofi, Bayer |
|
Interleukins |
10% |
Moderate |
Low |
Novartis, Pfizer |
By Application
Blood cancer holds the largest application segment at approximately 28% of 2026 revenue, driven by CD20/CD19 monoclonal antibodies and CAR-T cell therapy market targeting AML, CLL, and DLBCL. Key players include AbbVie (Imbruvica) and Novartis (Kymriah).
The lung cancer therapeutics market represents the fastest-growing application, with NSCLC driving the highest checkpoint inhibitor volumes globally and Merck's pembrolizumab as the dominant agent. Breast cancer is a growing indication for HER2-targeted antibodies including trastuzumab and pertuzumab, where triple-negative breast cancer is driving combination regimen adoption.
|
Application |
Est. 2026 Share (%) |
Fastest-Growing? |
Primary Drug Class |
Key Players |
|---|---|---|---|---|
|
Blood Cancer |
28% |
No (established) |
Monoclonal Ab, CAR-T |
AbbVie, Novartis, J&J |
|
Lung Cancer |
22% |
Yes |
Checkpoint Inhibitors |
Merck, AstraZeneca, Roche |
|
Breast Cancer |
14% |
Moderate |
Monoclonal Antibodies |
Roche, Pfizer, AbbVie |
|
Melanoma |
10% |
No (maturing) |
Checkpoint Inhibitors |
BMS, Merck |
|
Cervical Cancer |
9% |
Moderate |
Vaccines |
Merck, Sanofi, J&J |
|
Prostate Cancer |
7% |
Moderate |
Vaccines, Monoclonal Ab |
J&J, Sanofi |
|
Gastric Cancer |
6% |
Moderate-high |
Checkpoint Inhibitors |
AstraZeneca, Ono Pharma |
|
Glioblastoma |
4% |
Low (pipeline) |
Vaccines, CAR-T (trial) |
Novartis, Amgen (trial) |
Regional Projection of Global Immunotherapy Drugs Market
Immunotherapy drug adoption is not uniform. Reimbursement infrastructure, oncology prevalence patterns, and domestic biopharmaceutical capacity create significant regional revenue disparities.
- North America - The dominant region, generating more than 45% of global revenue. The US market is driven by FDA approvals, private payer coverage, and high per-treatment pricing. Canada is expanding public reimbursement for checkpoint inhibitors. Key players include Merck & Co., AbbVie, Amgen, and Pfizer. See the North America oncology drugs market for country-level detail.
- Europe - The second-largest region. EMA centralised approvals and multi-country HTA processes, including EUNETHRA joint clinical assessment from 2025, shape access timelines. Germany, France, and the UK record the highest per-capita immunotherapy spend. Roche, Novartis, and AstraZeneca dominate European manufacturing. (Source: European Medicines Agency)
- Asia-Pacific - The fastest-growing region at approximately 20% CAGR. China leads with domestic PD-1 inhibitor manufacturers (BeiGene, Hengrui); Japan has established early CAR-T reimbursement; India is scaling biosimilar manufacturing. Rising incidence of gastric and lung cancers is accelerating regional demand.
- Latin America - Moderate growth. Brazil and Mexico are the primary markets; price sensitivity limits checkpoint inhibitor adoption, while government tendering systems favour lowest-cost biosimilars.
- Middle East & Africa - A nascent but emerging market. GCC states including UAE and Saudi Arabia are investing in oncology infrastructure. Therapeutic vaccine programmes for cervical cancer show the strongest near-term uptake.
|
Region |
Est. 2026 Share (%) |
CAGR 2026–2034 |
Key Growth Driver |
|---|---|---|---|
|
North America |
45% |
14% |
Broad payer coverage, high pricing |
|
Europe |
25% |
13% |
EMA approval pipeline |
|
Asia-Pacific |
18% |
20% (fastest) |
Domestic manufacturers, rising incidence |
|
Latin America |
7% |
16% |
Biosimilar access expansion |
|
Middle East & Africa |
5% |
15% |
Oncology infrastructure build-out |
Global Immunotherapy Drugs Market - Recent Developments (2024–2025
The immunotherapy drugs competitive landscape shifted materially in 2024–2025. Seven new indications for PD-1/PD-L1 agents were approved by the FDA in 2024 alone, adding addressable patient populations worth an estimated USD 8 billion annually (FDA Hematology/Oncology Approvals, 2024).
- 2025 - Merck & Co.: Pembrolizumab (Keytruda) received FDA approval for an additional NSCLC combination regimen with chemotherapy, expanding first-line patient eligibility.
- 2024 - AstraZeneca: Durvalumab (Imfinzi) approved in combination with tremelimumab for hepatocellular carcinoma, entering a competitive space previously dominated by Roche's atezolizumab.
- 2024 - Novartis: Tisagenlecleucel (Kymriah) manufacturing capacity expanded at the Stein, Switzerland facility, targeting higher CAR-T cell therapy market supply volumes for European markets.
- 2024 - Pfizer: Completed acquisition of Seagen Inc. for USD 43 billion, adding an antibody-drug conjugate (ADC) portfolio to its oncology pipeline.
- 2024 - Companion Diagnostics Expansion: Abbott and PerkinElmer expanded PD-L1 testing agreements with multiple checkpoint inhibitor manufacturers, supporting companion diagnostic requirements across EU markets.
Global Immunotherapy Drugs Market - Future Outlook (2034)
The immunotherapy drugs market faces a structural inflection point between 2026 and 2030. Biosimilar entry for first-generation biologics will erode revenue per unit, while new indications, mRNA vaccines, and ADC combinations expand the addressable patient base. Goldman Sachs Research (2024) estimates that biologic patent cliffs will expose USD 200 billion or more in global drug revenues between 2025 and 2030.
Post-COVID-19 mRNA platform maturation - led by Pfizer/BioNTech and Moderna - is accelerating clinical readouts for personalised cancer vaccines, with early Phase III results expected between 2026 and 2028. The mRNA therapeutics market represents one of the two primary upside scenarios for sustaining a 16%+ CAGR through 2034. The ADC pipeline is equally significant: Pfizer/Seagen and AstraZeneca/Daiichi Sankyo programmes account for 15+ ADCs in Phase III as of 2025.
Artificial intelligence in antibody design - through AbSci and Exscientia partnerships with AstraZeneca and Sanofi - is compressing development timelines from five years to under two. IQVIA projects biosimilar penetration to reach 45% of biologic volume in the US by 2030, reducing per-unit revenues while expanding total treated patient volumes by 60% or more.
Why Choose This Report?
- Covers USD 379.28B (2026) to USD 1,224.70B (2034) market at 15.78% CAGR with methodology disclosed.
- Segment data across 5 drug types and 8 cancer application areas with share and growth outlook per sub-segment.
- Regional projections for North America, Europe, Asia-Pacific, Latin America, and MEA with country-level detail.
- Competitive profiles for 14 key players including Merck & Co., Roche, AstraZeneca, Pfizer, Novartis, AbbVie, and Amgen.
- Regulatory intelligence: FDA, EMA, and NMPA approval timelines and pipeline impact.
- Biosimilar risk assessment by drug class: patent expiry schedule 2026–2030.
- Customisation options: segment-level data extracts, country deep-dives, and competitive benchmarking available on request.
Table of Contents
- Introduction
- Objective of the study
- Product Definition
- Market Segmentation
- Study Variables
- Research Methodology
- Secondary Data Points
- Companies Interviewed
- Primary Data Points
- Breakdown of Primary Interviews
- Secondary Data Points
- Executive Summary
- Market Dynamics
- Drivers
- Challenges
- Opportunity Assessment
- Recent Trends and Developments
- Policy and Regulatory Landscape
- Global Immunotherapy Drugs Market Overview and Forecast Analysis (2021-2034)
- Market Size, By Value, By growth rate (CAGR/USD Billions)
- Demand - Supply Trends
- Market Share, By Drug Type
- Monoclonal Antibodies
- Interferons
- Interleukins
- Vaccines
- Checkpoint Inhibitors
- Market Share, By Application
- Blood Cancer
- Cervical Cancer
- Breast Cancer
- Glioblastoma
- Lung Cancer
- Gastric Cancer
- Prostate Cancer
- Melanoma
- Market Share, By End User
- Hospitals
- Clinics
- Other
- Market Share, By Region
- North America
- Europe
- Asia-Pacific
- Latin America
- Middle East & Africa
- Market Share, By Company
- Revenue Shares
- Competition Characteristics
- North America Immunotherapy Drugs Market Overview (2021-2034)
- Market Size, By Value (in USD Billions)
- Market Share, By Drug Type
- Market Share, By Application
- Market Share, By End User
- By Country
- The US
- Canada
- Mexico
- The US Immunotherapy Drugs Market Overview (2021-2034)
- Market Share, By Drug Type
- Market Share, By Application
- Market Share, By End User
- Canada Immunotherapy Drugs Market Overview (2021-2034)
- Market Share, By Drug Type
- Market Share, By Application
- Market Share, By End User
- Mexico Immunotherapy Drugs Market Overview (2021-2034)
- Market Share, By Drug Type
- Market Share, By Application
- Market Share, By End User
- South America Immunotherapy Drugs Market Overview (2021-2034)
- Market Size, By Value (in USD Billions)
- Market Share, By Drug Type
- Market Share, By Application
- Market Share, By End User
- By Country
- Brazil
- Argentina
- Rest of South America
- Brazil Immunotherapy Drugs Market Overview (2021-2034)
- Market Share, By Drug Type
- Market Share, By Application
- Market Share, By End User
- Argentina Immunotherapy Drugs Market Overview (2021-2034)
- Market Share, By Drug Type
- Market Share, By Application
- Market Share, By End User
- Europe Immunotherapy Drugs Market Overview (2021-2034)
- Market Size, By Value (in USD Billions)
- By Drug Type
- By Application
- By End User
- By Country
- Germany
- The UK
- France
- Spain
- Italy
- Rest of Europe
- Germany Immunotherapy Drugs Market Overview (2021-2034)
- Market Share, By Drug Type
- Market Share, By Application
- Market Share, By End User
- France Immunotherapy Drugs Market Overview (2021-2034)
- Market Share, By Drug Type
- Market Share, By Application
- Market Share, By End User
- The UK Immunotherapy Drugs Market Overview (2021-2034)
- Market Share, By Drug Type
- Market Share, By Application
- Market Share, By End User
- Spain Immunotherapy Drugs Market Overview (2021-2034)
- Market Share, By Drug Type
- Market Share, By Application
- Market Share, By End User
- Italy Immunotherapy Drugs Market Overview (2021-2034)
- Market Share, By Drug Type
- Market Share, By Application
- Market Share, By End User
- The Middle East & Africa Immunotherapy Drugs Market Overview (2021-2034)
- Market Size, By Value (in USD Billions)
- By Drug Type
- By Application
- By End User
- By Country
- The UAE
- Saudi Arabia
- South Africa
- Rest of the Middle East & Africa
- The UAE Immunotherapy Drugs Market Overview (2021-2034)
- Market Share, By Drug Type
- Market Share, By Application
- Market Share, By End User
- Saudi Arabia Immunotherapy Drugs Market Overview (2021-2034)
- Market Share, By Drug Type
- Market Share, By Application
- Market Share, By End User
- South Africa Immunotherapy Drugs Market Overview (2021-2034)
- Market Share, By Drug Type
- Market Share, By Application
- Market Share, By End User
- Asia-Pacific Immunotherapy Drugs Market Overview (2021-2034)
- Market Size, By Value (in USD Billions)
- By Drug Type
- By Application
- By End User
- By Country
- China
- India
- Japan
- South Korea
- Australia
- Rest of Asia-Pacific
- China Immunotherapy Drugs Market Overview (2021-2034)
- Market Share, By Drug Type
- Market Share, By Application
- Market Share, By End User
- India Immunotherapy Drugs Market Overview (2021-2034)
- Market Share, By Drug Type
- Market Share, By Application
- Market Share, By End User
- Japan Immunotherapy Drugs Market Overview (2021-2034)
- Market Share, By Drug Type
- Market Share, By Application
- Market Share, By End User
- South Korea Immunotherapy Drugs Market Overview (2021-2034)
- Market Share, By Drug Type
- Market Share, By Application
- Market Share, By End User
- Austraila Immunotherapy Drugs Market Overview (2021-2034)
- Market Share, By Drug Type
- Market Share, By Application
- Market Share, By End User
- Competitive Outlook (Company Profile - Partial List)
- Abbott
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- PerkinElmer Inc.
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- Illumina, Inc.
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- QIAGEN
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- F. Hoffmann-La Roche Ltd
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- Novartis AG
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- AstraZeneca
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- Pfizer Inc.
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- Sanofi
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- Johnson & Johnson Services, Inc.
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- AbbVie Inc.
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- Allergan
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- Merck & Co., Inc.
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- Amgen Inc.
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- Others
- Abbott
- Disclaimer
Top Key Players & Market Share Outlook
- Abbott
- PerkinElmer Inc.
- Illumina, Inc.
- QIAGEN
- F. Hoffmann-La Roche Ltd
- Novartis AG
- AstraZeneca
- Pfizer Inc.
- Sanofi
- Johnson & Johnson Services, Inc.
- AbbVie Inc.
- Allergan
- Merck & Co., Inc.
- Amgen Inc.
- Others
Frequently Asked Questions






