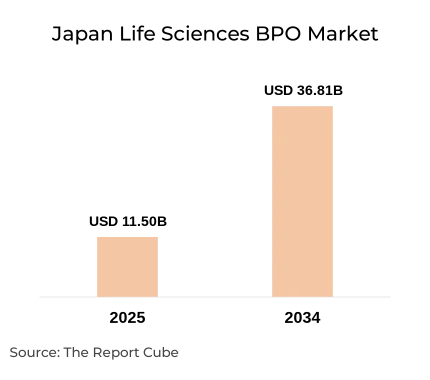
Build Your Custom Market Intelligence Report
Customize Your ReportJapan Life Sciences BPO Market Key Highlights
By Service Type:
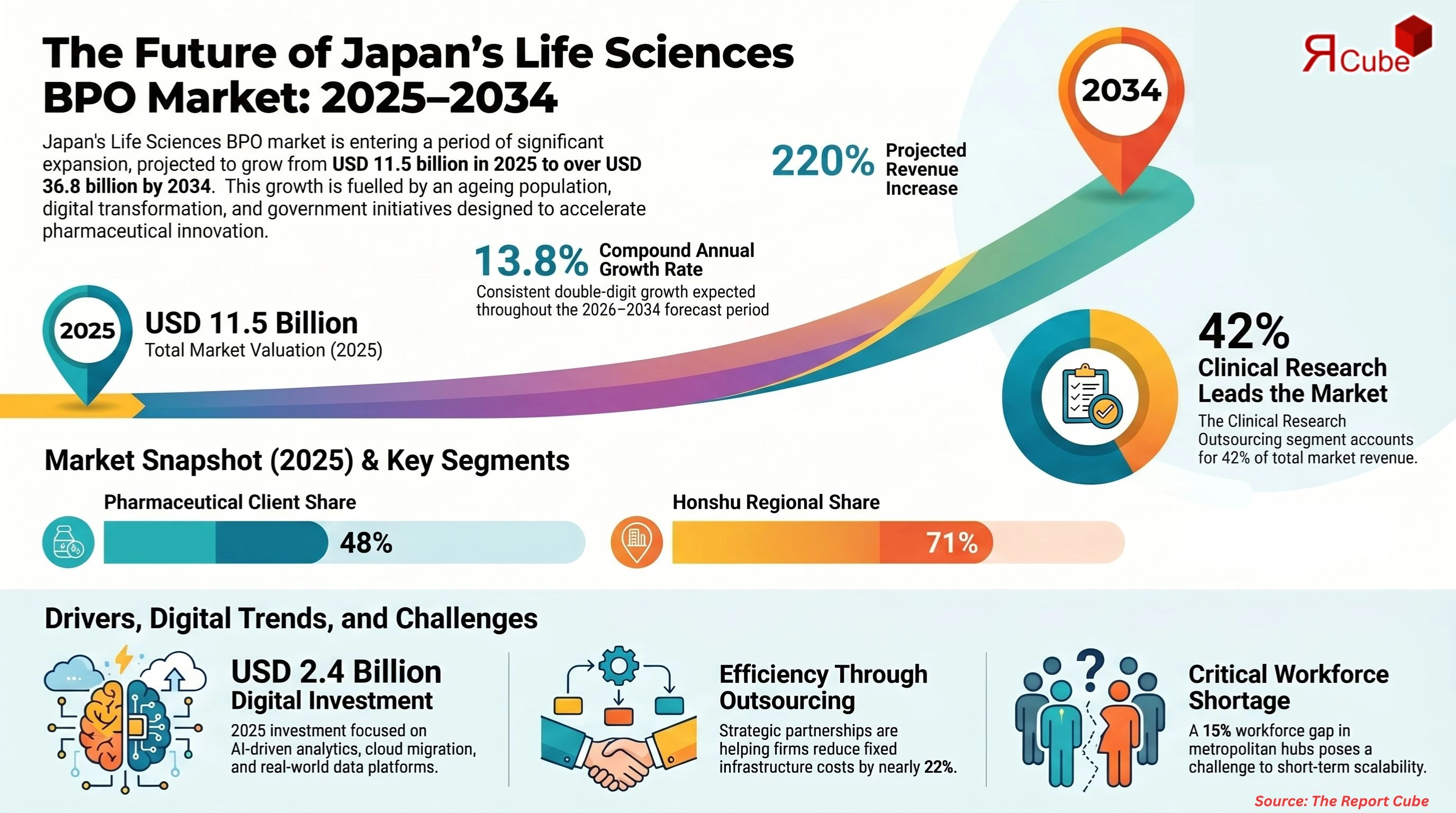
Clinical Research Outsourcing segment leads the market, with around 42% share.
By Client Type:
Pharmaceutical Companies segment dominates the market, accounting for nearly 48% of total revenue.
Regional Outlook:
Honshu region dominates the Japan life sciences bpo market, with around 71% share.
Government Initiatives & Policies
- Japan Revitalization Strategy 2026 – Pharmaceutical Innovation Acceleration Plan: The Ministry of Health, Labour and Welfare (MHLW) expanded fast-track clinical trial approvals and digital submission mandates in 2026 to reduce drug lag and strengthen outsourcing partnerships.
- AMED Advanced Medical R&D and DX Promotion Program (2026 Update): The Japan Agency for Medical Research and Development enhanced funding for decentralized trials, AI-driven drug discovery, and real-world data platforms to boost collaboration with CROs and BPO firms.
Japan Life Sciences BPO Market Insights & Analysis
The Japan Life Sciences BPO Market is anticipated to register a CAGR of 13.8% during 2026-2034. The market size was valued at USD 11.5 billion in 2025 and is projected to reach USD 36.81 billion by 2034. Japan’s aging population, advanced healthcare infrastructure, and strong pharmaceutical pipeline are reshaping outsourcing strategies across clinical, regulatory, manufacturing, and commercial functions.
Japan accounts for nearly 9% of global pharmaceutical sales and remains one of the top three drug markets worldwide. In 2025, more than 1,200 active clinical trials were registered across the country, with oncology, rare diseases, and regenerative medicine leading enrollment growth. Local pharmaceutical firms are increasingly partnering with contract research organizations (CROs) and IT-enabled service providers to reduce trial timelines by 18–24 months. The rise of decentralized clinical trials and electronic data capture systems has expanded demand for clinical data platforms and regulatory compliance support.
Foreign biopharma companies entering Japan face complex regulatory pathways and language-specific documentation requirements, which has accelerated regulatory affairs outsourcing and pharmacovigilance contracts. Investment in digital transformation reached approximately USD 2.4 billion across Japan’s life sciences ecosystem in 2025, with AI-driven analytics and cloud migration projects forming a large share. Companies such as CMIC Group and IQVIA Japan are strengthening full-service outsourcing models, enabling pharmaceutical clients to reduce fixed infrastructure costs by nearly 22% while maintaining compliance with PMDA standards.
Manufacturing outsourcing is also expanding as biologics and cell therapies grow. Japan’s regenerative medicine approvals have doubled over the past five years, creating opportunities for contract manufacturing outsourcing and cold-chain logistics support. Combined with government-backed digital health reforms, the market is transitioning from transactional outsourcing to strategic, long-term partnerships.
Japan Life Sciences BPO Market Dynamics
Key Driver: Expansion of Clinical Trials and Drug Innovation in Japan
Japan’s drug development pipeline is accelerating, driven by oncology, immunology, and rare disease research. By 2025, oncology alone represented nearly 38% of active clinical programs in Japan. Pharmaceutical firms are outsourcing Phase II and Phase III trials to manage operational complexity and accelerate regulatory approvals. The shift toward biologics and gene therapies requires specialized data management, pharmacovigilance monitoring, and regulatory submission expertise. Outsourcing enables sponsors to access localized regulatory knowledge and reduce operational overhead by up to 30%. The adoption of single IRB models and decentralized recruitment platforms further increases reliance on CRO partnerships. As global biopharma companies aim to launch therapies simultaneously in Japan and the US or EU, integrated outsourcing services are becoming essential for synchronized approvals.
Book your FREE 30-minute expert consultation today
Contact UsIndustry Trends: Digital Transformation and Real-World Evidence Integration
Digital transformation is reshaping Japan’s life sciences outsourcing ecosystem. Cloud-based clinical data platforms and AI-enabled analytics tools are reducing data cleaning time by 40% compared to legacy systems. Real-world evidence (RWE) projects, supported by Japan’s electronic health record infrastructure, are increasingly outsourced to analytics-focused BPO providers. In 2025, nearly 55% of mid-to-large pharmaceutical companies in Japan reported active RWE collaborations. IT and analytics outsourcing, including CRM platforms and safety reporting automation, is expanding at double-digit growth rates. Hybrid outsourcing models combining functional service providers with full-service contracts are gaining traction, offering flexibility in project scaling. These trends are positioning Japan as a technology-integrated life sciences outsourcing hub in Asia.
Major Challenge: Regulatory Complexity and Skilled Workforce Constraints
Japan maintains stringent regulatory standards through the Pharmaceuticals and Medical Devices Agency (PMDA), which requires extensive documentation and post-marketing surveillance. Regulatory submission cycles can extend beyond 12 months for certain therapeutic classes. BPO providers must maintain bilingual regulatory teams and advanced compliance systems, increasing operational complexity. Additionally, Japan faces a shortage of clinical research professionals, with workforce gaps estimated at 15% in metropolitan hubs such as Tokyo and Osaka. Competition for experienced biostatisticians and pharmacovigilance experts drives salary inflation and operational strain. These structural constraints may moderate short-term scalability for smaller outsourcing firms.
Opportunity: Growth in Rare Disease and Regenerative Medicine Outsourcing
Japan’s orphan drug incentives and accelerated approval pathways are creating a strong opportunity for specialized BPO services. Rare disease trials often require international coordination, complex patient recruitment, and long-term safety monitoring. Outsourcing providers offering end-to-end regulatory and clinical management can capture significant value. Regenerative medicine approvals have increased steadily, and advanced therapy manufacturing requires cold-chain logistics and quality assurance expertise. Collaboration between domestic CROs and global technology firms is expected to expand capacity. This niche segment is forecast to grow above 16% annually, outpacing the overall market average.
Japan Life Sciences BPO Market Segment-wise Analysis
By Service Type:
- Clinical Research Outsourcing
- Contract Manufacturing Outsourcing
- Contract Sales & Marketing Outsourcing
- Pharmacovigilance & Safety Outsourcing
- Regulatory Affairs Outsourcing
- IT & Analytics Outsourcing
- Medical Information & Contact Center Outsourcing
- Others
Clinical Research Outsourcing leads the Japan Life Sciences BPO Market, accounting for approximately 42% of total revenue in 2025. Phase II and Phase III services represent the largest share within this category due to higher patient enrollment requirements and extended monitoring periods. Sponsors are increasingly outsourcing data management and biostatistics services to reduce analysis turnaround times by nearly 35%. Site management services are expanding across academic hospitals in Tokyo and Osaka, improving patient recruitment efficiency. Regulatory submissions support also forms a significant component, especially for foreign sponsors navigating Japan-specific documentation. The increasing volume of oncology and rare disease trials is strengthening long-term CRO partnerships.
By Client Type:
- Pharmaceutical Companies
- Biotechnology Companies
- Medical Device Companies
- Generics Manufacturers
- Contract Research & Manufacturing Organizations
- Others
Pharmaceutical Companies dominate the client landscape, contributing nearly 48% of market revenue in 2025. Large domestic and multinational pharmaceutical firms rely heavily on outsourcing to streamline R&D expenditure. Japan’s top 20 pharmaceutical companies collectively invest over USD 15 billion annually in research and development. Outsourcing reduces infrastructure investment while enabling flexible scaling across therapeutic pipelines. Biotech firms are also expanding their share as venture-backed startups leverage CRO networks for faster proof-of-concept trials. However, pharmaceutical giants remain the primary revenue contributors due to high-volume, multi-phase clinical programs.
Regional Projection of Japan Life Sciences BPO Industry
- Hokkaido
- Honshu
- Shikoku
- Kyushu
Honshu dominates the regional landscape with around 71% market share in 2025. Tokyo, Osaka, and Yokohama host the majority of pharmaceutical headquarters, academic medical centers, and CRO offices. Approximately 78% of Japan’s clinical trial sites are located in Honshu, ensuring strong service demand. Kyushu follows with growing biotechnology clusters and regenerative medicine facilities. Hokkaido and Shikoku maintain smaller shares but are emerging as cost-effective trial locations. Infrastructure concentration and proximity to regulatory agencies continue to support Honshu’s leadership position.
Japan Life Sciences BPO Market Recent Developments
- 2025: CMIC Group entered a strategic service agreement with the Japanese Society for Inherited Metabolic Diseases to address drug loss in Japan, strengthening rare disease clinical development and market access capabilities across domestic and international partnerships.
- 2025: Syneos Health signed an agreement with ACTIVATO to enhance clinical trial execution efficiency in Japan through independent IRB services and optimized enrollment models for multi-center studies.
- 2025: IQVIA Japan announced long-term global clinical and commercial partnerships with Veeva Systems, enabling integrated data platforms and accelerating launch execution for innovative therapies in Japan.
- 2025: M3, Inc. expanded its commercialization footprint through Cosmotec Inc., forming an exclusive partnership with EverBridge Group to strengthen medical device market access and digital engagement strategies in Japan.
Need insights for a specific region within this market?
Request Regional DataWhy Choose This Report?
- Provides a comprehensive overview of the overall market analysis, encompassing key trends, consumer behavior analysis, and risk assessment to support strategic decision-making.
- Provides accurate, up-to-date insights into market size, segmentation, and emerging opportunities, helping to minimize risk & capitalizing on growth.
- Gives deep understanding of target audience preferences, investment habits, and communication channels for enhanced product development & marketing effectiveness.
- Delivers competitive analysis & benchmarking, uncovering the strengths & weaknesses of market competitors to guide strategies.
- Consolidate comprehensive market intelligence, reducing reasoning & streamlining research efforts.
- Facilitates customized market segmentation & risk mitigation strategies, fine-tuned to the business objectives.
- Aids in identifying both market challenges & untapped opportunities within the industry to drive long-term business growth.
- Provides valuable information based on actual customer data & search trends.
Table of Contents
- Introduction
- Objective of the Study
- Product and Category Definition
- Market Segmentation
- Study Variables
- Research Methodology
- Secondary Data Points
- Breakdown of Secondary Sources
- Primary Data Points
- Breakdown of Primary Interviews
- Secondary Data Points
- Executive Summary
- Market Dynamics
- Drivers
- Challenges
- Opportunity Assessment
- Recent Trends and Developments
- Regulatory and Policy Landscape
- Japan Life Sciences BPO Market Overview (2021-2034)
- Market Size, By Value (USD Billion)
- Market Share, By Service Type
- Clinical Research Outsourcing
- Phase I Services
- Phase II Services
- Phase III Services
- Phase IV Services
- Site Management Services
- Data Management Services
- Biostatistics Services
- Regulatory Submissions Support
- Pharmacovigilance & Safety Services
- Contract Manufacturing Outsourcing
- Active Pharmaceutical Ingredient Manufacturing
- Finished Dosage Form Manufacturing
- Packaging & Labeling Services
- Cold-Chain & Logistics Support
- Contract Sales & Marketing Outsourcing
- Sales Force Outsourcing
- Key Account Management Support
- Medical Affairs Support
- Market Access & Pricing Support
- Pharmacovigilance & Safety Outsourcing
- Case Processing Services
- Signal Detection & Risk Management
- Regulatory Safety Reporting
- Regulatory Affairs Outsourcing
- Dossier Preparation & Submission
- Labeling & Artwork Management
- Compliance & Lifecycle Management
- IT & Analytics Outsourcing
- Clinical Data Platforms
- Regulatory & Safety Systems
- Commercial Analytics & CRM Platforms
- Real-World Evidence & Big Data Analytics
- Cloud & Infrastructure Services
- Medical Information & Contact Center Outsourcing
- Others
- Clinical Research Outsourcing
- Market Share, By Client Type
- Pharmaceutical Companies
- Biotechnology Companies
- Medical Device Companies
- Generics Manufacturers
- Contract Research & Manufacturing Organizations
- Others
- Market Share, By Service Delivery Model
- Project-Based Outsourcing
- Full-Service Outsourcing
- Functional Service Provider Outsourcing
- Hybrid Outsourcing Models
- Market Share, By Region
- Hokkaido
- Honshu
- Shikoku
- Kyushu
- Market Share, By Company
- Revenue Shares & Analysis
- Competitive Landscape
- Hokkaido Life Sciences BPO Market
- Market Size, By Value (USD Billion/Million)
- Market Share, By Service Type
- Market Share, By Client Type
- Market Share, By Service Delivery Model
- Honshu Life Sciences BPO Market
- Market Size, By Value (USD Billion/Million)
- Market Share, By Service Type
- Market Share, By Client Type
- Market Share, By Service Delivery Model
- Shikoku Life Sciences BPO Market
- Market Size, By Value (USD Billion/Million)
- Market Share, By Service Type
- Market Share, By Client Type
- Market Share, By Service Delivery Model
- Kyushu Life Sciences BPO Market
- Market Size, By Value (USD Billion/Million)
- Market Share, By Service Type
- Market Share, By Client Type
- Market Share, By Service Delivery Model
- Competitive Outlook and Company Profiles
- CMIC Group
- Company Overview
- Product Portfolio
- Strategic Alliances/Partnerships
- Recent Developments
- EPS Holdings
- Company Overview
- Product Portfolio
- Strategic Alliances/Partnerships
- Recent Developments
- Linical Co., Ltd.
- Company Overview
- Product Portfolio
- Strategic Alliances/Partnerships
- Recent Developments
- IQVIA Japan
- Company Overview
- Product Portfolio
- Strategic Alliances/Partnerships
- Recent Developments
- PPD-SNBL
- Company Overview
- Product Portfolio
- Strategic Alliances/Partnerships
- Recent Developments
- Parexel International
- Company Overview
- Product Portfolio
- Strategic Alliances/Partnerships
- Recent Developments
- Syneos Health
- Company Overview
- Product Portfolio
- Strategic Alliances/Partnerships
- Recent Developments
- Accenture Japan
- Company Overview
- Product Portfolio
- Strategic Alliances/Partnerships
- Recent Developments
- Cognizant Japan
- Company Overview
- Product Portfolio
- Strategic Alliances/Partnerships
- Recent Developments
- TCS Japan
- Company Overview
- Product Portfolio
- Strategic Alliances/Partnerships
- Recent Developments
- Infosys Japan
- Company Overview
- Product Portfolio
- Strategic Alliances/Partnerships
- Recent Developments
- DNP CoArise
- Company Overview
- Product Portfolio
- Strategic Alliances/Partnerships
- Recent Developments
- Medidata Japan
- Company Overview
- Product Portfolio
- Strategic Alliances/Partnerships
- Recent Developments
- Bushu Pharma
- Company Overview
- Product Portfolio
- Strategic Alliances/Partnerships
- Recent Developments
- M3, Inc.
- Company Overview
- Product Portfolio
- Strategic Alliances/Partnerships
- Recent Developments
- Suzu-yo & Co.
- Company Overview
- Product Portfolio
- Strategic Alliances/Partnerships
- Recent Developments
- Others
- CMIC Group
- Contact Us and Disclaimer
Top Key Players & Market Share Outlook
- CMIC Group
- EPS Holdings
- Linical Co., Ltd.
- IQVIA Japan
- PPD-SNBL
- Parexel International
- Syneos Health
- Accenture Japan
- Cognizant Japan
- TCS Japan
- Infosys Japan
- DNP CoArise
- Medidata Japan
- Bushu Pharma
- M3, Inc.
- Suzu-yo & Co.
Frequently Asked Questions






