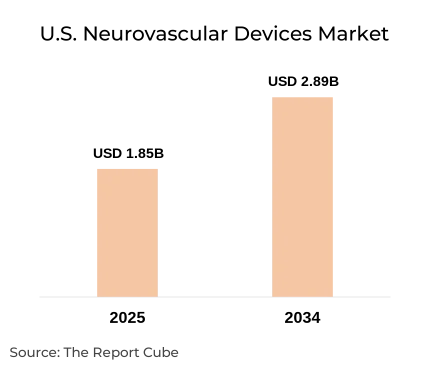
Build Your Custom Market Intelligence Report
Customize Your ReportU.S. Neurovascular Devices Market Statistics and Insights
Market Insight
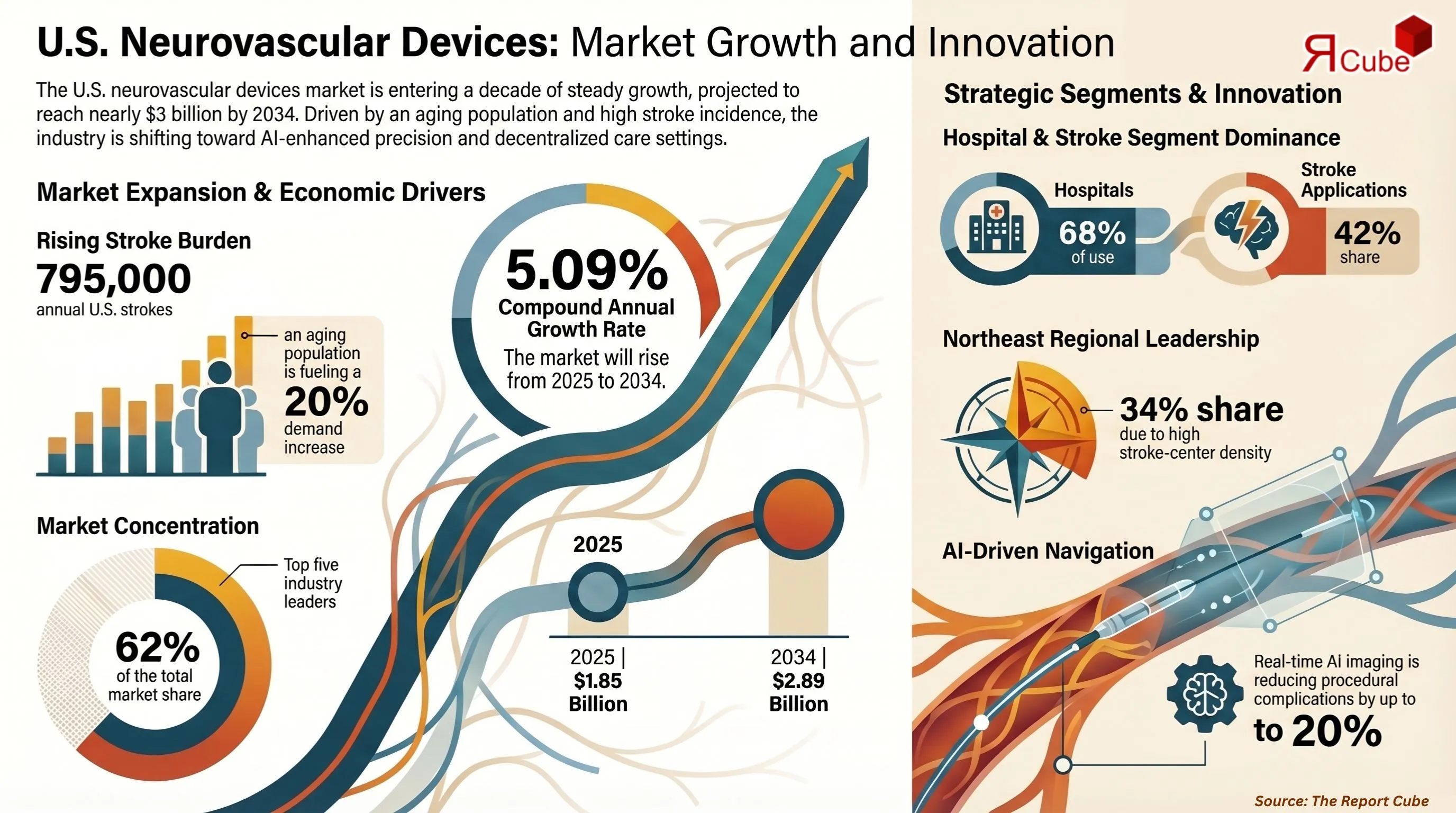
- The U.S. Neurovascular Devices Market was valued at USD 1.85 billion in 2025.
- The market is projected to reach USD 2.89 billion by the year 2034.
- The market is anticipated to expand at a CAGR of 5.09% during 2026–2034.
By Application
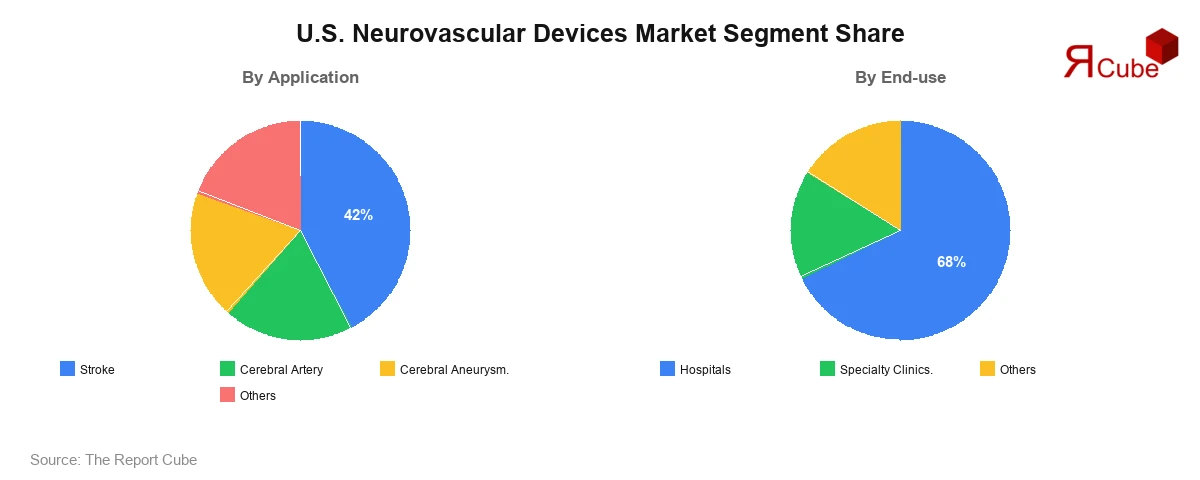
- The Stroke segment leads the market, commanding approximately 42% share.
Competitive Landscape
- More than 10 companies are actively engaged in producing U.S. Neurovascular Devices.
- Top 5 companies acquired around 62% of the market share.
- Medtronic, Johnson & Johnson Services Inc., Penumbra Inc., Stryker, and Microvention Inc. (Terumo Corporation), and others.
By End-use
- The Hospitals segment dominates end-use, accounting for approximately 68% share.
U.S. Neurovascular Devices Market Insights & Analysis
The U.S. Neurovascular Devices Market is anticipated to register a 5.09% CAGR during 2026–2034. The market size was valued at USD 1.85 billion in 2025 and is projected to reach USD 2.89 billion by 2034. The United States accounts for a dominant share of the global neurovascular devices industry, underpinned by its advanced healthcare infrastructure, high procedural volumes, and a well-established reimbursement framework under Medicare and Medicaid.
According to the Centers for Disease Control and Prevention, approximately 795,000 people in the U.S. experience a stroke annually, while the Brain Aneurysm Foundation reports that over 6.5 million Americans live with an unruptured brain aneurysm. This substantial patient burden is a consistent catalyst for device adoption. Federal funding from the National Institutes of Health (NIH) has been directed toward stroke research and interventional neurology innovation, further accelerating clinical development pipelines. In January 2025, Medtronic entered an exclusive U.S. distribution agreement with Contego Medical for the Neuroguard IEP combined stent-balloon-filter platform, reinforcing the trend of strategic consolidation and access expansion that is reshaping the competitive landscape of this market.
U.S. Neurovascular Devices Market Dynamics
Key Market Driver: Rising Stroke Burden Fueling Device Adoption
The escalating incidence of ischemic stroke across the aging U.S. population is among the most potent structural drivers of demand for neurovascular intervention devices. The Baby Boomer cohort, now entering its 70s and 80s, is projected to increase ischemic stroke incidence by nearly 20% over the coming decade. Medicare coverage expansion for carotid stenting and mechanical thrombectomy procedures has directly raised procedural volumes at certified stroke centers.
Furthermore, biplane angiography suite installations climbed approximately 18% in 2024, enabling hospitals to handle greater caseloads with improved precision. This institutional investment in neurovascular infrastructure translates to sustained procurement cycles for stent retrievers, aspiration catheters, and flow diverters, reinforcing a durable demand trajectory for device manufacturers operating in the U.S. market through 2034.
Major Industry Challenge: Regulatory Complexity Slowing Novel Device Approvals
The pathway for neurovascular device clearance through the U.S. Food and Drug Administration (FDA) presents a significant structural challenge for manufacturers. Devices that must navigate intracranial anatomy and meet stringent biocompatibility, radiopacity, and deliverability standards require multi-phase clinical validation, often spanning five to eight years from prototype to commercial launch. Post-market surveillance obligations and mandatory adverse event reporting further extend the resource burden on manufacturers.
The FDA's De Novo and Premarket Approval (PMA) tracks impose capital-intensive study requirements that disproportionately strain mid-sized innovators. In 2024, product recalls related to microcatheter kink resistance and coil detachment mechanisms highlighted gaps between bench testing outcomes and real-world procedural demands, prompting heightened scrutiny and additional post-approval commitments that slow the commercialization of next-generation platforms.
Emerging Trend Shaping Outlook: AI-Driven Navigation Transforming Neurovascular Interventions
Artificial intelligence is redefining the precision and efficiency benchmarks for neurovascular procedures, emerging as the most transformative technological trend shaping the market's long-term outlook. AI-enhanced imaging platforms now deliver real-time vessel analysis that reduces procedural complications by up to 20%, while robotic-assisted thrombectomy systems demonstrate a measurable reduction in radiation exposure for operating teams.
Integration of AI with digital subtraction angiography (DSA) enables automated vessel segmentation, aneurysm morphology mapping, and predictive flow modeling, reducing planning time and improving first-pass recanalization rates. Companies including Viz.ai have partnered with clinical research organizations to accelerate AI-validated neurovascular pathways. As hospitals increasingly adopt intelligent care coordination platforms, AI-embedded neurovascular workflows are expected to become a standard-of-care consideration across certified comprehensive stroke centers by 2030.
Need Specific Data, Let’s Customize the Report for You
Request CustomizationU.S. Neurovascular Devices Market Segment-wise Analysis
The U.S. Neurovascular Devices Market exhibits a clearly delineated segmentation structure across application and end-use verticals, with each segment demonstrating distinct clinical adoption dynamics, reimbursement conditions, and growth trajectories. Understanding these segment-level nuances is essential for manufacturers, investors, and healthcare strategists seeking to identify high-value opportunity zones within this specialized medical device landscape.
By Application
- Stroke
- Cerebral Artery
- Cerebral Aneurysm
- Others
The Stroke segment is the dominant application category, accounting for approximately 42% of total U.S. Neurovascular Devices Market revenue in 2025. The scale of stroke incidence in the United States, with nearly 795,000 cases reported annually, creates a recurring, high-volume demand environment for mechanical thrombectomy devices, stent retrievers, and aspiration catheters. Favorable Medicare reimbursement for mechanical thrombectomy procedures and the proliferation of certified comprehensive stroke centers have broadened procedural access significantly.
The Cerebral Aneurysm segment holds the second-largest revenue share, driven by the clinical urgency of treating ruptured aneurysms and the expanded adoption of flow diverter systems such as the Pipeline Embolization Device. Cerebral Artery conditions, including arteriovenous malformations and stenosis, represent a growing sub-market as minimally invasive stenting solutions gain regulatory traction. The Others category encompasses conditions such as cerebral venous sinus thrombosis, consolidating residual procedural demand not captured by the three primary segments.
By End-use
- Hospitals
- Specialty Clinics
- Others
Hospitals represent the commanding end-use segment, accounting for approximately 68% of U.S. Neurovascular Devices Market revenue in 2025. The complexity and time-sensitivity of neurovascular interventions, which frequently require multidisciplinary teams, intensive monitoring, and advanced imaging infrastructure, confine the majority of procedures to tertiary care and academic medical centers equipped with neurointervention suites.
Institutions such as comprehensive stroke centers have invested heavily in biplane angiography and hybrid operating rooms, creating sustained device procurement demand. The Specialty Clinics segment is the fastest-growing end-use category, reflecting the decentralization of select neurovascular procedures to ambulatory surgery centers and outpatient neurointervention facilities. Medtronic and GE Healthcare's collaboration to serve Office-Based Labs (OBLs) and Ambulatory Surgery Centers (ASCs), announced in 2022, exemplifies this care-setting diversification. The Others segment captures academic research institutions and rehabilitation centers with diagnostic neurovascular capabilities.
Regional Projection of U.S. Neurovascular Devices Market
The U.S. Neurovascular Devices Market is geographically concentrated, reflecting disparities in healthcare access, stroke center certification density, and interventional neurology specialist availability across the country.
- Northeast
- Midwest
- South
- West
The Northeast region commands the leading market share, estimated at approximately 34% of total U.S. revenue in 2025, driven by the highest density of comprehensive stroke centers, academic medical institutions, and neurointervention specialists in states including New York, Massachusetts, and Pennsylvania. The established research infrastructure and proximity to major medical device distributors further reinforce this regional dominance. The South and West regions are gaining share as healthcare infrastructure investment accelerates and telemedicine-supported stroke triage expands access to mechanical thrombectomy capabilities in previously underserved markets.
U.S. Neurovascular Devices Market: Recent Developments (2025)
- Medtronic signed exclusive U.S. distribution deal for the Neuroguard combined stent-balloon-filter, targeting expanded stroke volumes.
- Stryker launched the AXS Lift Intracranial Base Catheter in June 2025, reporting a 12% improvement in procedural margin contribution.
- Penumbra Inc. expanded its Lightning Flash aspiration platform, with gross margins reported at approximately 58% for its neurovascular product line.
- Johnson & Johnson Services Inc. advanced its neurovascular portfolio integration following prior strategic partnership commitments with Abbott Laboratories.
U.S. Neurovascular Devices Market Future Outlook (2034)
The U.S. Neurovascular Devices Market is poised for steady and sustainable expansion, projected to reach USD 2.89 billion by 2034 at a 5.09% CAGR. Growth will be underpinned by the continued aging of the U.S. population, persistent stroke and aneurysm burden, and accelerating technological adoption across certified neurovascular centers. Next-generation flow diverters, bioresorbable embolic materials, and robotic-assisted delivery platforms represent the most consequential innovation vectors through the forecast horizon.
AI-driven imaging integration will transform pre-procedural planning, enabling personalized intervention strategies that improve first-pass recanalization rates and long-term patient outcomes. Expansion of ambulatory neurovascular care settings and supportive CMS reimbursement policies will progressively democratize access to advanced devices beyond major academic centers, broadening the total addressable market through 2034.
Why Choose This Report?
- Provides a comprehensive overview of the overall market analysis, encompassing key trends, consumer behavior analysis, and risk assessment to support strategic decision-making.
- Provides accurate, up-to-date insights into market size, segmentation, and emerging opportunities, helping to minimize risk & capitalizing on growth.
- Gives deep understanding of target audience preferences, investment habits, and communication channels for enhanced product development & marketing effectiveness.
- Delivers competitive analysis & benchmarking, uncovering the strengths & weaknesses of market competitors to guide strategies.
- Consolidate comprehensive market intelligence, reducing reasoning & streamlining research efforts.
- Facilitates customized market segmentation & risk mitigation strategies, fine-tuned to the business objectives.
- Aids in identifying both market challenges & untapped opportunities within the industry to drive long-term business growth.
- Provides valuable information based on actual customer data & search trends.
Table of Contents
- Introduction
- Objective of the study
- Product Definition
- Market Segmentation
- Study Variables
- Research Methodology
- Secondary Data Points
- Companies Interviewed
- Primary Data Points
- Breakdown of Primary Interviews
- Secondary Data Points
- Executive Summary
- Market Dynamics
- Drivers
- Challenges
- Opportunity Assessment
- Recent Trends and Developments
- Policy and Regulatory Landscape
- U.S Neurovascular Devices Market Overview (2021-2034)
- Market Size, By Value (in USD Billions)
- Market Share, By Device
- Cerebral Embolization and Aneurysm Coiling Devices
- Cerebral Angioplasty and Stenting Systems
- Neurothrombectomy Devices
- Support Devices
- Trans Radial Access Devices
- Market Share, By Application
- Stroke
- Cerebral Artery
- Cerebral Aneurysm
- Others
- Market Share, By Size
- 0.027"
- 0.021"
- 0.071"
- 0.017"
- 0.019"
- 0.013"
- 0.058"
- 0.068"
- Others
- Market Share, By End-use
- Hospitals
- Specialty Clinics
- Others
- Market Share, By Region
- Northeast
- Midwest
- South
- West
- Market Share, By Competitors
- Competition Characteristics
- Revenue Shares
- U.S Cerebral Embolization and Aneurysm Coiling Devices Neurovascular Devices Market Overview, 2021-2034F
- By Value (USD Million)
- By Application- Market Size & Forecast 2021-2034, USD Million
- By Size- Market Size & Forecast 2021-2034, USD Million
- By End-use- Market Size & Forecast 2021-2034, USD Million
- U.S Cerebral Angioplasty and Stenting Systems Neurovascular Devices Market Overview, 2021-2034F
- By Value (USD Million)
- By Application- Market Size & Forecast 2021-2034, USD Million
- By Size- Market Size & Forecast 2021-2034, USD Million
- By End-use- Market Size & Forecast 2021-2034, USD Million
- U.S Neurothrombectomy Devices Neurovascular Devices Market Overview, 2021-2034F
- By Value (USD Million)
- By Application- Market Size & Forecast 2021-2034, USD Million
- By Size- Market Size & Forecast 2021-2034, USD Million
- By End-use- Market Size & Forecast 2021-2034, USD Million
- U.S Support Devices Neurovascular Devices Market Overview, 2021-2034F
- By Value (USD Million)
- By Application- Market Size & Forecast 2021-2034, USD Million
- By Size- Market Size & Forecast 2021-2034, USD Million
- By End-use- Market Size & Forecast 2021-2034, USD Million
- U.S Trans Radial Access Devices Neurovascular Devices Market Overview, 2021-2034F
- By Value (USD Million)
- By Application- Market Size & Forecast 2021-2034, USD Million
- By Size- Market Size & Forecast 2021-2034, USD Million
- By End-use- Market Size & Forecast 2021-2034, USD Million
- Competitive Outlook (Company Profile - Partial List)
- Medtronic
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- Johnson and Johnson Services Inc.
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- Penumbra, Inc.
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- Microport Scientific Corporation
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- Stryker
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- Microvention Inc (Terumo Corporation)
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- Codman Neuro (Integra Lifesciences)
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- Others
- Medtronic
- Contact Us & Disclaimer
Top Key Players & Market Share Outlook
- Medtronic
- Johnson and Johnson Services Inc.
- Penumbra, Inc.
- Microport Scientific Corporation
- Stryker
- Microvention Inc (Terumo Corporation)
- Codman Neuro (Integra Lifesciences)
- Others
Frequently Asked Questions






