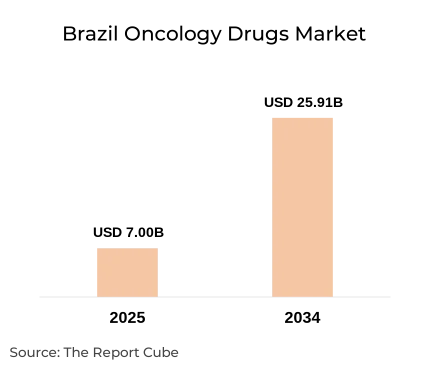
Build Your Custom Market Intelligence Report
Customize Your ReportBrazil Oncology Drugs Market Insights and Analysis
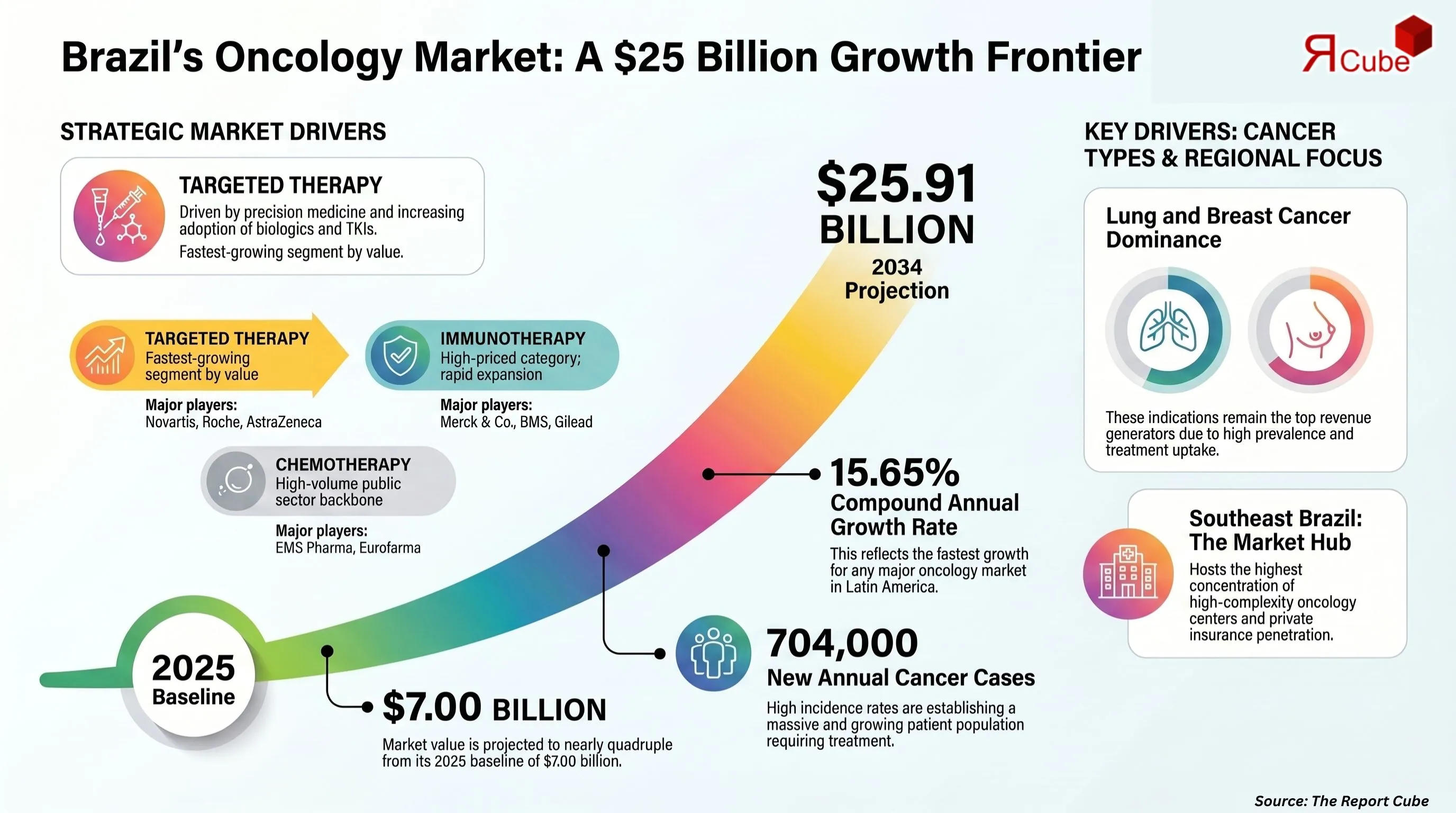
The Brazil oncology drugs market is the fastest-growing major oncology market in Latin America, expanding from USD 7.00 billion in 2025 to a projected USD 25.91 billion by 2034, at a robust CAGR of 15.65% during 2026 to 2034. Three converging forces are propelling this growth arc: a rising cancer incidence documented by INCA (Instituto Nacional de Câncer), continued expansion of the SUS (Sistema Único de Saúde) formulary through CONITEC approvals, and accelerating private health insurance coverage under ANS-mandated oncology protocols.
This article is for oncology-focused investors, pharmaceutical manufacturers, healthcare distributors, and regulatory affairs professionals evaluating commercial and formulary opportunities in Brazil's cancer drugs landscape through 2034. The analysis covers drug class segmentation from chemotherapy to immunotherapy, indication-level concentration in lung cancer and breast cancer, CMED pricing tiers, ANVISA's regulatory pathway, and the competitive dynamics among the five dominant market players.
The Latin America oncology drugs market is increasingly anchored by Brazil, which drives the region's premium-drug adoption curve. Within Brazil, targeted therapy and immunotherapy segments are growing above the aggregate market CAGR, positioning them as the highest-value opportunities for both multinationals and domestic manufacturers through the forecast horizon.
Key Takeaways: Brazil Oncology Drugs Market
- Market Size (2025): Valued at USD 7.00 billion, reflecting a strong baseline demand for oncology treatments across Brazil.
- Projected Market Size (2034): Expected to reach USD 25.91 billion, indicating significant long-term expansion driven by innovation and rising cancer incidence.
- CAGR (2026-2034): Forecast to grow at a robust 15.65%, highlighting one of the faster-growing segments within the healthcare sector.
- Dominant Drug Class: Targeted Therapy leads in value growth, fueled by precision medicine and increasing adoption of TKIs and biologics.
- Dominant Indication: Lung Cancer remains the top revenue-generating segment, followed closely by Breast Cancer, due to high prevalence and advanced treatment uptake.
- Leading Region: Southeast Brazil dominates the market, supported by better healthcare infrastructure and higher patient access to advanced therapies.
- Key Players: Major companies shaping the competitive landscape include EMS Pharma, Eurofarma, Hoffmann-La Roche Ltd, Novartis AG, and Takeda Pharmaceutical.
Brazil Oncology Drugs Market Dynamics
Key Market Driver - Rising Cancer Incidence and SUS Formulary Expansion
Brazil's age-standardised cancer incidence rate has increased consistently across breast, lung, colorectal, and prostate cancers. INCA projects approximately 704,000 annual new cases across the 2023 to 2025 reference period, establishing a large and growing patient population requiring pharmaceutical treatment. CONITEC reviews and approves oncology drugs for SUS inclusion on a continuous basis, and each approval directly increases public procurement volumes at scale.
Roche's trastuzumab and Novartis's imatinib stand as concrete examples where SUS inclusion drove a measurable and sustained volume increase in public hospital dispensing. CONITEC approvals convert clinical-trial-stage drugs into immediate public-sector procurement contracts, bypassing the private-pay adoption lag seen in other markets. For manufacturers with SUS-listed products, this creates captive public procurement demand that is volume-stable, budget-protected, and largely insulated from private-sector pricing volatility.
Major Industry Challenge - Regulatory Lead Times and ANVISA Price Controls
ANVISA is Brazil's drug regulatory authority and reviews oncology products under its priority registration track (Registro Prioritário). Despite this pathway, average total registration timelines for novel oncology agents without expedited designation can extend to 18 to 24 months, creating commercial entry delays for innovative therapies. ANVISA drug registration complexity is compounded for biologic oncology drugs, particularly monoclonal antibodies, where ANVISA's biosimilar guideline requires comparative clinical data for market authorisation.
Beyond registration, CMED (Câmara de Regulação do Mercado de Medicamentos) sets maximum government sale prices (PGF) and hospital consumer prices (PMC) that can compress manufacturer margins on premium biologics. Companies navigating the priority registration track can reduce timelines, but eligibility criteria remain narrow, and most novel agents progress through the standard pathway.
Emerging Trend - Domestic Manufacturing and Farmanguinhos PDP Partnerships
Brazil's Ministry of Health operates the PDP (Productive Development Partnerships) programme, under which international manufacturers partner with Brazilian state-owned laboratories, Farmanguinhos, Butantan, and LAFEPE, to transfer technology and manufacture priority medicines domestically. Oncology drugs including trastuzumab and imatinib have active or completed PDP agreements, increasing supply security and reducing import dependency.
Roche and Novartis are the two multinationals with active technology transfer agreements under PDP. The strategic implication for the targeted therapy market is significant: companies without a PDP partnership risk formulary displacement by locally manufactured equivalents as SUS procurement shifts preference toward domestic supply chains.
Need Specific Data, Let’s Customize the Report for You
Request CustomizationBrazil Oncology Drugs Market Segment-wise Analysis
By Drug Class
|
Drug Class |
Key Drugs |
Major Players |
Market Insights |
|---|---|---|---|
|
Chemotherapy |
Cisplatin, Carboplatin, Paclitaxel, Docetaxel |
EMS Pharma, Eurofarma |
Most widely reimbursed in SUS by volume; strong presence in public procurement; price-driven competition; stable but slower growth |
|
Targeted Therapy |
Imatinib, Erlotinib, Osimertinib |
Novartis, Roche, AstraZeneca |
Fastest-growing segment by value; strong expansion via CONITEC & ANS coverage; expected to grow above 15.65% CAGR |
|
Immunotherapy |
Pembrolizumab, Nivolumab |
Merck & Co., Bristol-Myers Squibb, Gilead Sciences |
Highest-priced category; rapid expansion beyond private sector; CAR-T therapies gaining traction |
|
Hormonal Therapy |
Anastrozole, Letrozole, Enzalutamide, Leuprolide |
Sanofi, Bayer |
High-volume but lower-value segment; widely used in prostate & breast cancer; margin-dilutive but stable |
Chemotherapy still dominates in sheer volume. It’s the backbone of treatment, especially in public healthcare (SUS), where affordability matters most. Domestic players like EMS Pharma and Eurofarma thrive here by competing aggressively on pricing.
But the real momentum? That’s happening in targeted therapies. These drugs are changing how cancer is treated, more precise, more effective. With regulatory support expanding access, this segment is growing faster than the overall market.
Immunotherapy, on the other hand, sits at the premium end. Think of it as the “high-value, high-impact” category. Drugs like pembrolizumab are redefining survival outcomes, and their adoption is quickly moving beyond elite private care.
Hormonal therapy plays a quieter role. It doesn’t grab headlines, but it ensures consistent demand—especially in breast and prostate cancer, making it a steady volume driver.
By Indication
|
Indication |
Key Drugs / Treatments |
Leading Companies |
Market Insights |
|---|---|---|---|
|
Lung Cancer |
EGFR TKIs, PD-1 inhibitors, ALK inhibitors (Alectinib, Brigatinib) |
Takeda, Roche |
High mortality; shift to targeted therapy increasing revenue per patient |
|
Breast Cancer |
Trastuzumab, Palbociclib, Ribociclib |
Novartis |
Most common cancer in women; strong public access via SUS |
|
Colorectal Cancer |
FOLFOX, FOLFIRI, Bevacizumab, Cetuximab |
Roche, Merck KGaA |
High incidence; combination therapies gaining traction |
|
Prostate Cancer |
Enzalutamide, Abiraterone, Radium-223 |
Pfizer, Astellas, Bayer |
Dominated by ADT; growth in advanced-stage treatments |
|
Leukemia & Lymphoma |
Imatinib, Rituximab, CAR-T therapies |
Gilead Sciences |
Strong treatment infrastructure; innovation in cell therapy |
|
Melanoma |
BRAF inhibitors, Pembrolizumab |
Roche, Novartis |
Higher incidence due to UV exposure; immunotherapy now first-line |
Lung cancer is where innovation is hitting hardest. The move from traditional chemotherapy to targeted drugs and immunotherapy has dramatically increased treatment value per patient.
Breast cancer, being the most common in women, has a well-structured treatment ecosystem. Public-private partnerships-like trastuzumab availability under SUS-have made advanced therapies more accessible than you’d expect in a developing market.
Colorectal cancer still leans on traditional chemo regimens, but combination biologics are gaining ground. It’s a gradual shift, but a meaningful one.
Prostate cancer remains heavily dependent on hormonal approaches. However, newer drugs for advanced stages are adding serious revenue potential.
In hematological cancers like leukemia and lymphoma, Brazil stands out. With strong treatment centers and access to cutting-edge therapies like CAR-T, this segment is pushing into high-innovation territory.
And then there’s melanoma. Brazil’s high UV exposure makes it more common than in many regions, and immunotherapy has quickly become the go-to first-line treatment.
Regulatory and Import Landscape
|
Body |
Role |
Relevant Process |
Typical Timeline |
|---|---|---|---|
|
ANVISA |
Drug registration and market authorisation |
Standard and Priority Registration (Registro Prioritário) |
18 to 24 months (standard); shorter under priority track |
|
CMED |
Pharmaceutical price regulation |
Maximum price-setting (PGF, PMC); annual CAF adjustment |
Concurrent with registration |
|
CONITEC |
Health technology assessment for SUS |
Clinical and cost-effectiveness review; RENAME recommendation |
180 days per HTA cycle |
|
ANS |
Supplementary health coverage mandates |
Oncology protocol updates aligning CFM guidelines |
Periodic resolution cycles |
|
Ministry of Health |
SUS procurement and PDP programme |
Formulary administration; PDP technology transfer |
Multi-year agreements |
ANVISA governs oncology drug registration under RDC 204/2017 and subsequent resolutions. Imported oncology drugs require a GMP certificate from the country of manufacture and ANVISA batch release prior to commercial distribution. Brazil's MERCOSUR common external tariff (TEC) applies a 0% rate to active pharmaceutical ingredients and a 2% rate to finished pharmaceutical products, making import costs for oncology APIs relatively low compared to other market entry costs.
Regional Projection of Brazil Oncology Drugs Market
- Southeast Brazil, comprising São Paulo, Rio de Janeiro, Minas Gerais, and Espírito Santo, holds the largest regional share of the Brazil oncology drugs market. São Paulo hosts Brazil's highest concentration of high-complexity oncology centres (UNACON and CACON accredited facilities). A.C. Camargo Cancer Center and ICESP (Instituto do Câncer do Estado de São Paulo) are the two largest oncology referral centres and serve as the primary entry point for novel therapies before expansion to other regions.
- South Brazil, Paraná, Santa Catarina, and Rio Grande do Sul, has among the highest colorectal cancer incidence rates in Brazil and above-average private health insurance penetration, driving greater private-pay oncology drug consumption relative to national averages.
- Northeast Brazil represents the fastest-growing regional segment due to SUS oncology network expansion under the Rede de Atenção Oncológica programme. LAFEPE, the state laboratory of Pernambuco, supplies chemotherapy generics to the regional public health network, supporting volume growth independent of multinational pharmaceutical activity.
- Center-West and North Brazil collectively hold the smallest regional share due to a limited number of accredited high-complexity centres. The Ministry of Health has committed to expanding CACON and UNACON accreditations through 2026, which will create new distribution access points for manufacturers targeting public-sector oncology procurement.
Competitive Landscape
- Hoffmann-La Roche Ltd holds the leading position in Brazil's oncology drugs market through its HER2-positive breast cancer portfolio, trastuzumab, pertuzumab, and trastuzumab emtansine, and bevacizumab across colorectal and lung cancer indications. Roche's PDP partnership with Farmanguinhos for trastuzumab creates a structural barrier for biosimilar competitors seeking equivalent SUS formulary positioning.
- Novartis AG leads the leukemia and breast cancer segments through imatinib and CDK4/6 inhibitors ribociclib (Kisqali) and palbociclib. Novartis secured ANVISA approval for tisagenlecleucel (Kymriah) CAR-T therapy, making Brazil the first Latin American country to authorise this therapy class. Novartis's CDK4/6 portfolio is gaining ANS coverage mandates, positioning it for sustained private-sector growth even as imatinib revenue declines through genericisation.
- EMS Pharma is Brazil's largest domestic pharmaceutical company and the leading domestic supplier of chemotherapy generics to SUS. EMS holds significant platinum-based and taxane supply contracts through Ministry of Health procurement. Vertical integration in chemotherapy generics allows EMS to consistently underbid multinational competitors on SUS procurement, securing volume at thin but stable margins.
- Takeda Pharmaceutical leads Brazil's lung cancer segment through its ALK inhibitor portfolio, including alectinib (Alecensa) and brigatinib (Alunbrig), and holds a strong position in multiple myeloma with bortezomib and ixazomib. In 2024, Takeda secured ANVISA approval for mobocertinib (Exkivity) for EGFR exon 20 insertion-mutated NSCLC, expanding its reach within the premium targeted therapy lung oncology segment.
- Eurofarma is the second-largest domestic pharmaceutical company and an active competitor in oncology generics, particularly hormonal therapy drugs for breast and prostate cancer. In 2023, Eurofarma signed a licensing agreement to develop biosimilar oncology products for the Brazilian and Latin American market, positioning it to compete in targeted therapy through the 2026 to 2034 forecast period.
Sanofi, Bayer AG, and Gilead Sciences are active participants in hormonal therapy, prostate cancer, and haematological oncology respectively, and are acknowledged as significant segment contributors outside the top-five overall ranking.
Recent Developments (2024–2025)
- 2025: CONITEC recommended pembrolizumab for first-line NSCLC under SUS coverage, representing a landmark first immunotherapy checkpoint inhibitor inclusion on the RENAME list.
- 2025: ANS updated its oncology coverage mandate to include CDK4/6 inhibitors for HR-positive, HER2-negative metastatic breast cancer across all supplementary health plans.
Future Outlook - Brazil Oncology Drugs Market 2034
The Brazil oncology drugs market grows from USD 7.00 billion in 2025 to USD 25.91 billion by 2034 at a 15.65% CAGR. Immunotherapy and targeted therapy will grow above the aggregate CAGR through this period, driven by new ANVISA approvals, expanding ANS coverage mandates, and CONITEC SUS inclusions for precision therapies. Chemotherapy will grow below the aggregate CAGR as treatment protocols shift toward precision medicine, while biosimilar competition compresses per-unit revenue in this class.
Domestic manufacturers EMS Pharma and Eurofarma will increase their share of chemotherapy and hormonal therapy through generics and biosimilar entrants, expanding volume while compressing revenue per unit. Molecular diagnostics adoption, particularly next-generation sequencing (NGS) for biomarker testing, will directly correlate with targeted therapy prescribing rates, as more patients are identified as eligible for precision agents.
Two specific pipeline catalysts will shape the 2034 outlook: the first Brazilian CAR-T manufacturing facility under BNDES (Brazilian Development Bank) financing, which would reduce CAR-T therapy costs and expand haematological oncology access; and CONITEC's active HTA pipeline for ADC (antibody-drug conjugate) therapies, which represents the next premium drug class entering SUS evaluation. Referencing the broader Brazil cancer diagnostics market, diagnostics adoption and therapeutics uptake are positively correlated, reinforcing the integrated commercial opportunity across the oncology value chain through 2034.
Why Choose This Report?
- Verified Brazil oncology drugs market sizing with confirmed CAGR and growth trajectory
- Full segment breakdown by drug class and indication with competitive player mapping
- Dedicated ANVISA regulatory and CMED pricing analysis unavailable on competing pages
- Profiles of top five competitive players with recent 2024 to 2025 actions and strategic implications
- Regional projections covering Southeast, South, Northeast, and Center-West and North Brazil
- Recent developments section updated to 2025 with named company actions and approval dates
- Forecast to 2034 with drug class-level growth trajectory analysis above and below aggregate CAGR
- Customisation available for specific company, indication, or regional focus requirements
Table of Contents
- Introduction
- Objective of the study
- Product Definition
- Market Segmentation
- Study Variables
- Research Methodology
- Secondary Data Points
- Companies Interviewed
- Primary Data Points
- Breakdown of Primary Interviews
- Secondary Data Points
- Executive Summary
- Market Dynamics
- Drivers
- Challenges
- Opportunity Assessment
- Recent Trends and Developments
- Policy and Regulatory Landscape
- Brazil Oncology Drugs Market Overview and Forecast Analysis (2021-2034)
- Market Size, By Value, By growth rate (CAGR/USD Billions)
- Demand - Supply Trends
- Market Share, By Drug Class
- Chemotherapy
- Targeted Therapy
- Immunotherapy
- Hormonal Therapy
- Others
- Market Share, By Indication
- Lung Cancer
- Breast Cancer
- Colorectal Cancer
- Prostate Cancer
- Leukemia & Lymphoma
- Melanoma
- Others
- Market Share, By Therapy Type
- Monotherapy
- Combination Therapy
- Market Share, By Distribution Channel
- Hospital Pharmacies
- Retail Pharmacies
- Online Pharmacies
- Specialty Clinics
- Market Share, By Region
- Southeast Brazil
- South Brazil
- Northeast Brazil
- Centre-West and North Brazil
- Market Share, By Competitors
- Competition Characteristics
- Revenue Shares
- Brazil Chemotherapy Oncology Drugs Market Overview, 2021-2034F
- By Value (USD Million)
- By Indication- Market Size & Forecast 2021-2034, USD Million
- By Therapy Type- Market Size & Forecast 2021-2034, USD Million
- By Distribution Channel- Market Size & Forecast 2021-2034, USD Million
- Brazil Targeted Therapy Oncology Drugs Market Overview, 2021-2034F
- By Value (USD Million)
- By Indication- Market Size & Forecast 2021-2034, USD Million
- By Therapy Type- Market Size & Forecast 2021-2034, USD Million
- By Distribution Channel- Market Size & Forecast 2021-2034, USD Million
- Brazil Immunotherapy Oncology Drugs Market Overview, 2021-2034F
- By Value (USD Million)
- By Indication- Market Size & Forecast 2021-2034, USD Million
- By Therapy Type- Market Size & Forecast 2021-2034, USD Million
- By Distribution Channel- Market Size & Forecast 2021-2034, USD Million
- Brazil Hormonal Therapy Oncology Drugs Market Overview, 2021-2034F
- By Value (USD Million)
- By Indication- Market Size & Forecast 2021-2034, USD Million
- By Therapy Type- Market Size & Forecast 2021-2034, USD Million
- By Distribution Channel- Market Size & Forecast 2021-2034, USD Million
- Brazil Others Oncology Drugs Market Overview, 2021-2034F
- By Value (USD Million)
- By Indication- Market Size & Forecast 2021-2034, USD Million
- By Therapy Type- Market Size & Forecast 2021-2034, USD Million
- By Distribution Channel- Market Size & Forecast 2021-2034, USD Million
- Competitive Outlook (Company Profile - Partial List)
- EMS Pharma
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- Eurofarma
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- Sanofi
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- Takeda Pharma
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- Hoffmann-La Roche Ltd
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- Novartis AG
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- Gilead Sciences Inc
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- Bayer AG
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- Others
- EMS Pharma
- Contact Us & Disclaimer
Top Key Players & Market Share Outlook
- EMS Pharma
- Eurofarma
- Sanofi
- Takeda Pharma
- Hoffmann-La Roche Ltd
- Novartis AG
- Gilead Sciences Inc
- Bayer AG
- Others
Frequently Asked Questions






