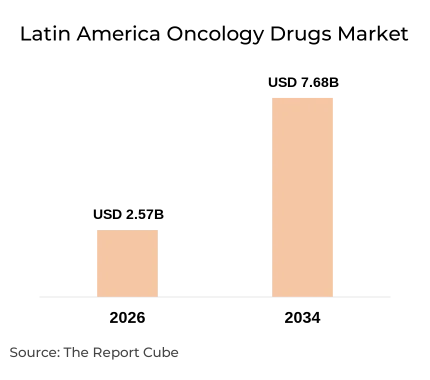
Build Your Custom Market Intelligence Report
Customize Your ReportKey Takeaways
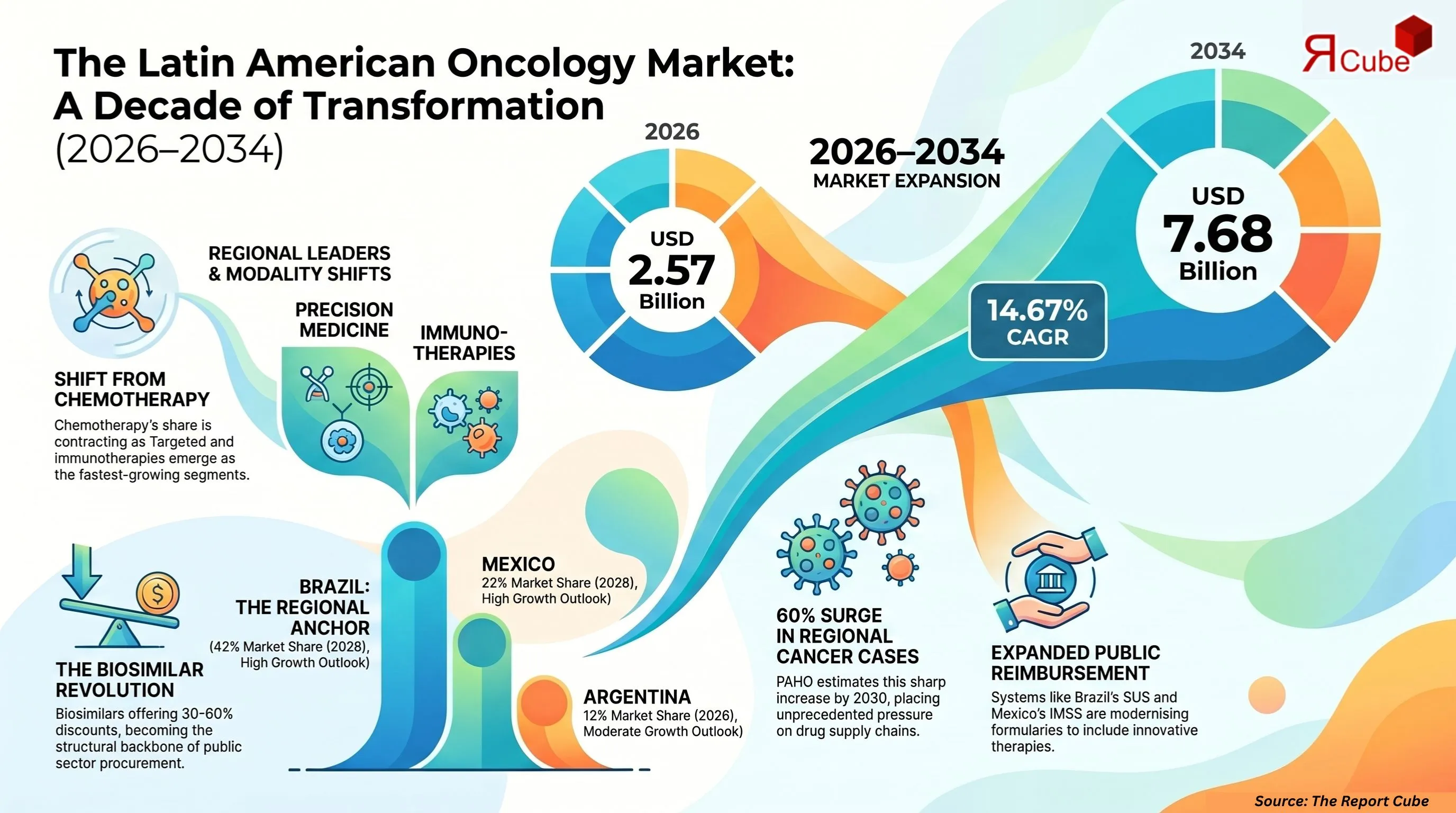
- The market is on a strong growth trajectory, expanding from USD 2.57 billion in 2026 to USD 7.68 billion by 2034, reflecting a solid 14.67% CAGR.
- Targeted therapy is emerging as the fastest-growing segment, signaling a shift toward precision-based treatment approaches.
- While chemotherapy remains the dominant therapy in 2026, its lead is expected to gradually narrow as advanced therapies gain traction.
- Blood cancer continues to hold the largest share across the forecast period, with breast cancer also contributing significantly to demand.
- Brazil stands out as the leading regional market, accounting for approximately 42% of total drug spending, making it the primary revenue driver.
- The market landscape is evolving rapidly, supported by increasing healthcare investments, rising cancer prevalence, and improved access to innovative treatments.
- The analysis is based on 2026 as the base year, with projections covering the period from 2026 to 2034, offering a long-term outlook on market expansion.
Latin America Oncology Drugs Market Insights & Analysis
The Latin America oncology drugs market is valued at USD 2.57 billion in 2026 and is projected to reach USD 7.68 billion by 2034, expanding at a CAGR of 14.67% over the forecast period. Cancer incidence across Latin America is accelerating faster than healthcare infrastructure can absorb - particularly in Brazil, Mexico, and Argentina, where diagnosis-to-treatment delays remain a critical barrier. The Pan American Health Organization estimates that Latin America will see a 60% increase in cancer cases by 2030 (PAHO, 2023), placing unprecedented pressure on already strained oncology drug supply chains. This section decodes the structural forces behind the market's 14.67% CAGR - from expanding public reimbursement frameworks to the entry of biosimilars and next-generation targeted agents.
Rising cancer burden across the region is one of the primary demand anchors. Colorectal, breast, cervical, and haematological malignancies are driving volume demand at an accelerating rate. Governments are responding: Brazil's SUS (Sistema Único de Saúde) and Mexico's IMSS/ISSSTE frameworks have expanded their oncology drug reimbursement lists, directly increasing procurement volumes for both originator and biosimilar drugs.
The private hospital sector is growing alongside public systems. Medical tourism within the region - particularly in Mexico and Colombia - is increasing uptake of premium targeted therapies among patients who bypass public system wait times. Biosimilar penetration is simultaneously reducing per-unit cost and widening patient access, especially in Brazil and Chile. Regulatory convergence across ANVISA (Brazil), COFEPRIS (Mexico), and ANMAT (Argentina) is compressing approval timelines, enabling faster market entry for both innovative and biosimilar agents.
What makes the latin america cancer drugs market structurally distinctive is its dual-speed nature: premium targeted therapies are growing in private-pay urban centres, while biosimilar chemotherapy is expanding access across public health systems simultaneously. This bifurcation is the central growth engine that competitors have failed to quantify.
Latin America Oncology Drugs Market Dynamics
Key Market Driver: Rising Cancer Incidence and Government Reimbursement Expansion
Governments across Latin America face a compounding problem: cancer mortality rates are rising while per-capita oncology drug spend remains among the lowest in the Americas. Brazil alone recorded over 704,000 new cancer cases in 2023 (INCA, 2023), yet public oncology drug budgets cover fewer than 40% of first-line treatment protocols for haematological cancers. Expanding SUS reimbursement lists and IMSS formulary additions are directly unlocking volume demand for both originator and biosimilar oncology drugs across the region.
Mexico's IMSS modernisation plan (2023–2025) has added targeted oncology agents to its national formulary, significantly expanding the addressable patient population for checkpoint inhibitors and EGFR inhibitors in public settings. Chile's GES (Garantías Explícitas en Salud) programme expanded coverage to 17 cancer types in 2022, pulling government drug procurement volumes upward across breast and colorectal indications. A rising middle class across the region is driving a structural shift from public to private oncology drug consumption, further expanding the premium drug market.
The table below maps the reimbursement gap between Brazil - the most advanced market - and smaller economies where the next wave of growth will originate:
|
Country |
Key Reimbursement Body |
Oncology Drug Coverage Level |
Recent Policy Action |
|---|---|---|---|
|
Brazil |
SUS / CONITEC |
High |
Expanded biosimilar trastuzumab inclusion, 2023 |
|
Mexico |
IMSS / ISSSTE |
Moderate |
Formulary modernisation, 2024 |
|
Chile |
GES / FONASA |
Moderate |
GES expanded to 17 cancers, 2022 |
|
Argentina |
PAMI / IOMA |
Moderate |
Generic oncology drug prioritisation, 2023 |
|
Colombia |
SGSSS |
Low–Moderate |
Regulatory simplification underway |
Major Industry Challenge: Limited Oncology Infrastructure and Diagnostic Delays
A drug market cannot grow faster than the diagnostic systems that feed it - and across Latin America, late-stage cancer diagnosis remains the norm rather than the exception. Approximately 70% of cervical cancer cases in Latin America are diagnosed at Stage III or IV (PAHO, 2022), making many first-line drug regimens irrelevant and dramatically compressing the addressable treatment window. Infrastructure deficits - in pathology labs, oncology specialists, and cold-chain logistics - form the primary structural ceiling on how quickly drug volumes can scale.
Oncologist-to-patient ratios across Brazil, Mexico, and Colombia remain among the lowest in OECD-comparable nations, creating bottlenecks between diagnosis and prescription. Cold-chain logistics gaps limit biologics distribution outside major metropolitan areas, directly constraining immunotherapy and targeted biologic uptake in secondary cities. Patent protection disputes and import pricing conflicts are delaying biosimilar access in Argentina and Peru. Brain drain of trained oncology professionals to North America and Europe is weakening domestic treatment capacity faster than medical education pipelines can compensate.
Cold-chain logistics represents an underreported structural constraint: immunotherapy and targeted biologics - the fastest-growing therapy segments - are the modalities most directly affected. Until regional cold-chain investment catches up, the chemotherapy market in Latin America will retain volume dominance in secondary markets regardless of formulary policy changes.
Emerging Trend: Biosimilar Oncology Drug Adoption Accelerating Across Public Health Systems
Originator oncology biologics price at 8–15× the cost of equivalent biosimilars - a gap that public health payers across Latin America can no longer absorb. Brazil's ANVISA approved 14 oncology biosimilars between 2020 and 2024, yet biosimilar penetration in the public oncology formulary still sits below 25% of eligible treatment lines (ANVISA, 2024). The 2026–2034 forecast period will see biosimilar adoption move from a cost-containment experiment to the structural backbone of public oncology drug spending across the region.
Trastuzumab, bevacizumab, and rituximab biosimilars are the leading volume drivers. PAHO's Strategic Fund is enabling volume-based pricing through regional procurement alliances, compressing per-unit costs further. The private sector continues to lag the public sector on biosimilar adoption due to physician preference for originator brands - a behaviour that precision market access programmes are beginning to address in Brazil and Mexico.
|
Biosimilar Drug |
Originator Reference |
Key Market |
Regulatory Status |
Price Discount vs. Originator |
|---|---|---|---|---|
|
Trastuzumab biosimilar |
Herceptin (Roche) |
Brazil, Mexico |
Approved |
40–60% |
|
Bevacizumab biosimilar |
Avastin (Roche) |
Brazil, Colombia |
Approved |
35–55% |
|
Rituximab biosimilar |
Mabthera (Roche) |
Brazil, Argentina |
Approved |
30–50% |
|
Imatinib generic |
Gleevec (Novartis) |
Region-wide |
Multiple approvals |
70–80% |
Modelling 30% biosimilar penetration across the top 5 Latin American public health systems would generate an estimated USD 400–600 million in annual procurement savings by 2030 - capital that can be redeployed toward expanding access to targeted agents for previously untreated patient populations.
Need Specific Data, Let’s Customize the Report for You
Request CustomizationLatin America Oncology Drugs Market Segment-wise Analysis
By Therapy Modalities
Selecting the wrong therapy modality for a cancer type is not merely a clinical error - it determines which drug classes dominate procurement budgets and which manufacturers capture volume. Chemotherapy still accounted for the majority of oncology drug spending across Latin American public health systems in 2025, but its revenue share is contracting at 2–3 percentage points per year as targeted therapy penetration rises. The table below maps each modality's current share, growth trajectory, and the patient population driving its demand:
|
Therapy Modality |
Estimated Market Share (2026) |
Growth Outlook (2026–2034) |
Key Cancer Types Served |
|---|---|---|---|
|
Chemotherapy |
38% |
Moderate |
Blood, GI, Lung, Breast |
|
Targeted Therapy |
28% |
High |
Breast, Lung, Blood |
|
Immunotherapy (Biologic) |
18% |
Very High |
Lung, Skin, Blood |
|
Hormonal Therapy |
11% |
Stable |
Breast, Prostate |
|
Others (CAR-T, ADCs) |
5% |
Emerging |
Blood, Breast |
Targeted therapy is the fastest-growing segment, driven by EGFR inhibitors, HER2 inhibitors, and CDK4/6 inhibitors expanding rapidly across the region's private-pay markets. Checkpoint inhibitors (PD-1/PD-L1) in the immunotherapy segment are entering public formularies in Brazil and Mexico. Hormonal therapy - primarily generic aromatase inhibitors for breast and prostate cancer - maintains stable demand with high affordability in public settings. CAR-T and antibody-drug conjugates (ADCs) remain in early-phase clinical availability but will gain formulary traction after 2028.
By Type (Cancer Indication)
Not all cancer types carry equal market weight - drug complexity, treatment duration, and reimbursement status determine whether a cancer indication generates USD 50 million or USD 500 million in annual drug spend. Blood cancers (leukaemia, lymphoma, multiple myeloma) collectively represent the highest drug-cost burden per patient in Latin America, with treatment courses averaging USD 40,000–USD 120,000 annually in private settings. The table below analyses how each cancer type's incidence, treatment duration, and drug price tier shapes its share of the market:
|
Cancer Type |
Estimated Market Share (2026) |
Primary Drug Classes |
Growth Outlook |
|---|---|---|---|
|
Blood Cancer |
24% |
Targeted agents, Chemotherapy, Immunotherapy |
High |
|
Breast Cancer |
22% |
HER2 inhibitors, CDK4/6 inhibitors, Chemotherapy |
High |
|
Gastrointestinal Cancer |
18% |
Chemotherapy (FOLFOX/FOLFIRI), Bevacizumab |
Moderate |
|
Respiratory/Lung Cancer |
16% |
EGFR inhibitors, Checkpoint inhibitors |
High |
|
Skin Cancer |
7% |
Checkpoint inhibitors (PD-1/PD-L1) |
Very High |
|
Other Cancers |
13% |
Chemotherapy, Hormonal Therapy |
Moderate |
Cross-referencing GLOBOCAN incidence data against drug spend per indication reveals a persistent underfunding pattern: gastrointestinal cancer carries the third-largest patient burden in Latin America yet receives proportionally less drug investment than breast or blood cancer due to lower per-protocol drug costs. This mismatch signals a future volume opportunity as bevacizumab biosimilars and FOLFOX regimen costs decline.
Regional Projection of the Latin America Oncology Drugs Market
Latin America is not a single oncology drug market - it is five distinct market tiers separated by healthcare infrastructure maturity, reimbursement depth, and regulatory sophistication. Brazil alone accounts for an estimated 40–45% of regional oncology drug spending (PAHO, 2024), meaning that a single country's procurement policy can shift regional growth curves by 2–3 percentage points. The projections below identify where the fastest growth will emerge outside Brazil's established dominance.
- Brazil - Market leader. ANVISA's progressive regulatory environment, SUS public reimbursement, and INCA-driven cancer registry infrastructure make Brazil the region's anchor market. Biosimilar oncology integration and private hospital sector expansion fuel both volume and value growth simultaneously.
- Mexico - Second-largest market. COFEPRIS regulatory reforms and IMSS formulary expansion are driving access for targeted agents. The growing affluent urban population in Mexico City, Guadalajara, and Monterrey is absorbing premium therapies ahead of public system coverage timelines.
- Argentina - Volatile due to macroeconomic instability and currency controls limiting drug import capacity. Domestic manufacturing provides a partial buffer, but foreign exchange instability creates persistent pricing pressure on imported originator biologics.
- Colombia - Fast-growing mid-tier market. Private oncology clinic expansion in Bogotá and Medellín is pulling checkpoint inhibitor demand ahead of the public system's coverage timeline, positioning Colombia as the region's highest relative growth market through 2030.
- Chile - Small but high-value. Chile's GES insurance programme and high per-capita GDP support premium drug uptake across both public and private settings, creating the region's highest biosimilar-to-originator ratio by value.
- Rest of Latin America - Peru, Ecuador, Bolivia, and Central America represent early-stage markets with high unmet need, low current drug penetration, and significant long-run growth potential as regional reimbursement policies gradually harmonise.
|
Country |
Estimated Market Share (2026) |
Key Growth Driver |
Primary Regulatory Body |
Growth Outlook (2026–2034) |
|---|---|---|---|---|
|
Brazil |
42% |
Biosimilar formulary expansion, private sector |
ANVISA |
High |
|
Mexico |
22% |
IMSS modernisation, urban private hospitals |
COFEPRIS |
High |
|
Argentina |
12% |
Domestic manufacturing, generic oncology |
ANMAT |
Moderate (macro risk) |
|
Colombia |
9% |
Private oncology clinic growth |
INVIMA |
Very High |
|
Chile |
7% |
GES coverage + high per-capita GDP |
ISP |
Moderate–High |
|
Rest of LATAM |
8% |
Unmet need, early-stage market entry |
Various |
High (from low base) |
Latin America Oncology Drugs Market - Recent Developments (2024–2025)
- 2025 - F. Hoffmann-La Roche Ltd. expanded its oncology portfolio in Brazil through a strategic agreement with INCA to supply trastuzumab biosimilar for public-sector breast cancer treatment programmes - activating the public formulary channel for biosimilar HER2 therapy at scale.
- 2024 - Novartis A.G. received ANVISA approval for an updated Kymriah (tisagenlecleucel) indication covering relapsed/refractory follicular lymphoma in adult patients in Brazil, expanding CAR-T access through a private access channel.
- 2024 - Eurofarma Laboratórios S.A. announced a biosimilar oncology co-development agreement targeting the Brazilian and Mexican public health markets, signalling domestic manufacturer entry into the high-growth biosimilar segment.
- 2024 - Sanofi S.A. launched a patient access programme in Mexico for isatuximab (Sarclisa) in multiple myeloma in coordination with IMSS procurement authorities - a direct public formulary access play for a premium targeted agent.
- 2024 - Icon Plc. expanded clinical trial operations in Latin America, adding oncology-focused Phase II/III sites in São Paulo, Mexico City, and Bogotá, deepening the regional clinical trial pipeline for next-cycle approvals.
Latin America Oncology Drugs Market Future Outlook (2034)
Forecasting a market from USD 2.57 billion to USD 7.68 billion is straightforward - explaining which structural forces will sustain a 14.67% CAGR for eight consecutive years is not. Three previous Latin American pharmaceutical market expansions at comparable growth rates stalled between years 4 and 6 due to currency devaluation events, regulatory bottlenecks, or insurance coverage rollbacks - risks that are active today in Argentina and Brazil. This section projects the four forces most likely to determine whether the 2034 target is met, exceeded, or missed.
- Policy driver: Universal health coverage (UHC) commitments across Brazil, Mexico, and Colombia mandate expanded oncology drug reimbursement lists through 2030. Each incremental formulary addition directly converts unmet patient need into drug procurement volume.
- Technology driver: ADC (antibody-drug conjugate) approvals - including trastuzumab deruxtecan and sacituzumab govitecan - are projected to enter Latin American markets by 2027–2028 through ANVISA and COFEPRIS fast-track pathways, adding a new high-value revenue layer atop existing targeted therapy demand.
- Demographic driver: The 60+ age cohort in Brazil and Mexico is growing at 3.4% CAGR (PAHO, Health in the Americas 2023), directly expanding cancer incidence and drug demand volumes at a rate that will sustain procurement growth independent of policy changes.
- Risk factor: Currency volatility across BRL, ARS, and MXN against USD creates persistent pricing instability for imported originators, compressing manufacturer margins and incentivising parallel import controls that could slow premium therapy adoption.
Why Choose This Report?
- Covers the 2026–2034 forecast period with base year 2026 validation and scenario modelling
- Segment analysis across 5 therapy modalities and 6 cancer indications with market share and growth outlook
- Country-level projections for Brazil, Mexico, Argentina, Colombia, Chile, and Rest of LATAM
- Competitive landscape covering 9 named key players plus emerging regional manufacturers
- Biosimilar penetration modelling - including public vs. private sector drug spend breakdown
- Policy tracker covering ANVISA, COFEPRIS, ANMAT, and INVIMA regulatory pipelines
- Actionable data formatted for strategic planning, investor due diligence, and market entry assessment
Table of Contents
- Introduction
- Objective of the study
- Product Definition
- Market Segmentation
- Study Variables
- Research Methodology
- Secondary Data Points
- Companies Interviewed
- Primary Data Points
- Breakdown of Primary Interviews
- Secondary Data Points
- Executive Summary
- Market Dynamics
- Drivers
- Challenges
- Opportunity Assessment
- Recent Trends and Developments
- Policy and Regulatory Landscape
- Latin America Oncology Drugs Market Overview and Forecast Analysis (2021-2034)
- Market Size, By Value, By growth rate (CAGR/USD Billions)
- Demand - Supply Trends
- Market Share, By Therapy Modalities
- Chemotherapy
- Targeted Therapy
- Immunotherapy (Biologic Therapy)
- Hormonal Therapy
- Others
- Market Share, By Type
- Blood Cancer
- Breast Cancer
- Gastrointestinal Cancer
- Respiratory/Lung Cancer
- Skin Cancer
- Other Cancers
- Market Share, By Region
- Brazil
- Mexico
- Argentina
- Colombia
- Rest of LATAM
- Market Share, By Competitors
- Competition Characteristics
- Revenue Shares
- Latin America Chemotherapy Oncology Drugs Market Overview, 2021-2034F
- By Value (USD Million)
- By Type- Market Size & Forecast 2021-2034, USD Million
- Latin America Targeted Therapy Oncology Drugs Market Overview, 2021-2034F
- By Value (USD Million)
- By Type- Market Size & Forecast 2021-2034, USD Million
- Latin America Immunotherapy (Biologic Therapy) Oncology Drugs Market Overview, 2021-2034F
- By Value (USD Million)
- By Type- Market Size & Forecast 2021-2034, USD Million
- Latin America Hormonal Therapy Oncology Drugs Market Overview, 2021-2034F
- By Value (USD Million)
- By Type- Market Size & Forecast 2021-2034, USD Million
- Latin America Others Oncology Drugs Market Overview, 2021-2034F
- By Value (USD Million)
- By Type- Market Size & Forecast 2021-2034, USD Million
- Competitive Outlook (Company Profile - Partial List)
- Laboratrio Cristlia
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- Ach Laboratrios Farmacuticos S.A.
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- Icon Plc.
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- F. Hoffmann-La Roche Ltd.
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- Laboratorios IMA
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- Eurofarma Laboratrios S.A.
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- Laboratoires Pierre Fabre
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- Novartis A.G
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- Sanofi S.A
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- Others
- Laboratrio Cristlia
- Contact Us & Disclaimer
Top Key Players & Market Share Outlook
- Laboratrio Cristlia
- Ach Laboratrios Farmacuticos S.A.
- Icon Plc.
- F. Hoffmann-La Roche Ltd.
- Laboratorios IMA
- Eurofarma Laboratrios S.A.
- Laboratoires Pierre Fabre
- Novartis A.G
- Sanofi S.A
- Others
Frequently Asked Questions






