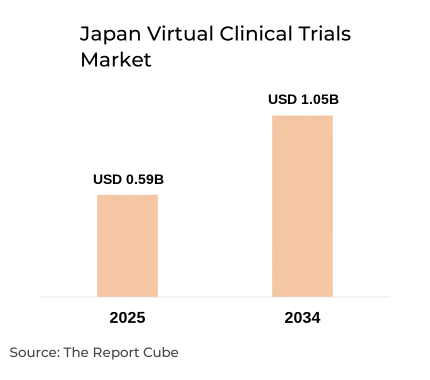
Build Your Custom Market Intelligence Report
Customize Your ReportJapan Virtual Clinical Trials Market Statistics and Insights
Market Size Statistics
- Japan Virtual Clinical Trials Market valued USD 0.59 billion during 2025
- Market projected reaching USD 1.05 billion by 2034 globally
- Market expected expanding at compound annual growth rate 6.56%
By Indication
- Oncology segment dominated market with approximately 28% share
Competition
- More than 10 companies are actively engaged in producing Japan Virtual Clinical Trials Market
- Top 5 companies acquired around 53% of the market share.
- ICON plc, Parexel International Corporation, IQVIA Inc, Laboratory Corporation of America Holdings, Medidata, and others
By Phase
- Phase III trials accounted for nearly 37% market share
Japan Virtual Clinical Trials Market Insights & Analysis
The Japan Virtual Clinical Trials Market is anticipated to register a 6.56% CAGR during 2026-2034. The market size was valued at USD 0.59 billion in 2025 and is projected to reach USD 1.05 billion by 2034. The market is experiencing steady growth as pharmaceutical companies increasingly adopt digital technologies to improve the efficiency of clinical research. Virtual clinical trials utilize digital platforms, telemedicine tools, wearable devices, and remote patient monitoring systems to conduct clinical studies without requiring participants to visit traditional trial sites frequently.
These decentralized trial models allow researchers to recruit participants from broader geographic locations, improving patient diversity and study enrollment rates. Virtual trials also reduce logistical challenges associated with patient travel and site management, making clinical studies more accessible and efficient. Japan’s advanced healthcare infrastructure and strong pharmaceutical industry support the adoption of innovative research technologies. Regulatory agencies in the country are gradually encouraging the use of digital solutions in clinical research to enhance trial efficiency and patient engagement.
Japan Virtual Clinical Trials Market Dynamics
Key Market Driver – Increasing Adoption of Decentralized Clinical Trials
- Growing adoption of decentralized clinical trial models is driving the Japan Virtual Clinical Trials Market.
- Traditional trials require patients to travel to research sites for monitoring and data collection.
- These logistical requirements can limit participation and delay study enrollment processes.
- Virtual clinical trials enable studies through digital platforms and remote monitoring technologies.
- Telemedicine consultations and electronic data capture systems support remote patient engagement.
- Participants can join clinical trials from home, improving convenience and accessibility.
Major Industry Challenge – Regulatory and Data Security Concerns
- Regulatory compliance and data security remain significant challenges for virtual clinical trials.
- Clinical studies involve sensitive patient information requiring strict privacy protection measures.
- Regulatory agencies require adherence to strict healthcare data protection standards.
- Digital trial platforms must comply with national and international clinical research regulations.
- Maintaining secure data storage systems is essential for protecting patient information.
- Integrating digital health technologies with traditional research systems can be complex.
Emerging Trend Shaping Outlook – Integration of AI and Digital Health Technologies
- Artificial intelligence integration is transforming the Japan Virtual Clinical Trials Market.
- AI-powered analytics platforms process large volumes of clinical trial data efficiently.
- These systems analyze data from remote monitoring tools and digital health platforms.
- AI algorithms help researchers identify patient patterns and treatment responses.
- Predictive analytics improves clinical trial design and research decision-making.
- AI-assisted tools support faster and more accurate patient recruitment processes.
Need Specific Data, Let’s Customize the Report for You
Request Regional DataJapan Virtual Clinical Trials Market Segment-wise Analysis
By Indication
- CNS
- Autoimmune/Inflammation
- Cardiovascular Disease
- Metabolic/Endocrinology
- Infectious Disease
- Oncology
- Genitourinary
- Ophthalmology
- Others
Indication segmentation indicates that oncology represents the largest segment in the Japan Virtual Clinical Trials Market, accounting for approximately 28% of total demand. Oncology clinical trials often involve complex treatment protocols and require extensive patient monitoring, making virtual trial technologies highly beneficial.
Remote monitoring tools allow researchers to track patient health metrics and treatment responses more effectively. These technologies also enable patients with serious illnesses to participate in clinical trials without frequent hospital visits. Clinical trials focusing on cardiovascular diseases and metabolic disorders also represent significant segments due to the high prevalence of these conditions within aging populations.
By Phase
- Phase I
- Phase II
- Phase III
- Phase IV
Phase segmentation shows that Phase III trials dominate the Japan Virtual Clinical Trials Market, accounting for approximately 37% of total market share. Phase III trials involve large patient populations and are conducted to confirm the safety and effectiveness of new therapeutic treatments before regulatory approval.
Virtual trial platforms are particularly valuable in Phase III studies because they allow researchers to recruit participants from diverse geographic regions and monitor patient outcomes remotely. Phase II trials also represent a significant portion of the market as pharmaceutical companies evaluate treatment effectiveness and determine optimal dosage levels.
Regional Projection of Japan Virtual Clinical Trials Market
- Kanto Region
- Kansai Region
- Chubu Region
- Other Regions
The Kanto region represents the largest share of the Japan Virtual Clinical Trials Market, accounting for approximately 39% of total demand. The region includes major pharmaceutical research centers and academic medical institutions located in Tokyo and surrounding areas. Kansai region also contributes significantly to the market due to the presence of biotechnology companies and leading healthcare research institutions.
Chubu region supports growing clinical research activities through its network of hospitals and research centers involved in pharmaceutical trials. Other regions across Japan are gradually adopting digital clinical research technologies as pharmaceutical companies expand decentralized trial models across multiple healthcare facilities.
Japan Virtual Clinical Trials Market: Recent Development
- 2025: IQVIA Inc expanded decentralized clinical trial services supporting digital research operations in Japan.
- 2025: Parexel International Corporation launched advanced virtual trial management platforms improving patient recruitment.
- 2025: Medidata introduced cloud-based clinical data management systems supporting remote patient monitoring.
- 2025: Medable Inc expanded digital clinical trial technologies supporting decentralized healthcare research initiatives.
Japan Virtual Clinical Trials Market Future Outlook (2034)
The Japan Virtual Clinical Trials Market is expected to experience steady expansion through 2034 as pharmaceutical research increasingly integrates digital technologies and decentralized clinical trial models. With a projected CAGR of 6.56%, the market is anticipated to reach approximately USD 1.05 billion by 2034.
Future growth will largely depend on advancements in digital health technologies, artificial intelligence, and cloud-based clinical data platforms. These technologies will improve patient monitoring, data collection accuracy, and clinical trial efficiency. Pharmaceutical companies are expected to continue adopting decentralized trial models to accelerate drug development and improve patient participation rates.
Why Choose This Report?
- Provides a comprehensive overview of the overall market analysis, encompassing key trends, consumer behavior analysis, and risk assessment to support strategic decision-making.
- Provides accurate, up-to-date insights into market size, segmentation, and emerging opportunities, helping to minimize risk & capitalizing on growth.
- Gives deep understanding of target audience preferences, investment habits, and communication channels for enhanced product development & marketing effectiveness.
- Delivers competitive analysis & benchmarking, uncovering the strengths & weaknesses of market competitors to guide strategies.
- Consolidate comprehensive market intelligence, reducing reasoning & streamlining research efforts.
- Facilitates customized market segmentation & risk mitigation strategies, fine-tuned to the business objectives.
- Aids in identifying both market challenges & untapped opportunities within the industry to drive long-term business growth.
- Provides valuable information based on actual customer data & search trends.
Table of Contents
- Introduction
- Objective of the study
- Product Definition
- Market Segmentation
- Study Variables
- Research Methodology
- Secondary Data Points
- Companies Interviewed
- Primary Data Points
- Break Down of Primary Interviews
- Secondary Data Points
- Executive Summary
- Market Dynamics
- Drivers
- Challenges
- Opportunity Assessment
- Recent Trends and Developments
- Policy and Regulatory Landscape
- Japan Virtual Clinical Trials Market Overview (2021-2034)
- Market Size, By Value (in USD Billions)
- Market Share, By Study
- Interventional
- Observational
- Expanded Access
- Market Share, By Indication
- CNS
- Autoimmune/Inflammation
- Cardiovascular Disease
- Metabolic/Endocrinology
- Infectious Disease
- Oncology
- Genitourinary
- Ophthalmology
- Others
- Market Share, By Phase
- Phase I
- Phase II
- Phase III
- Phase IV
- Market Share, By Region
- Kanto Region
- Kansai Region
- Chubu Region
- Other Regions
- Market Share, By Competitors
- Competition Characteristics
- Revenue Shares
- Japan Interventional Virtual Clinical Trials Market Overview, 2021-2034F
- By Value (USD Million/Billion)
- By Indication- Market Size & Forecast 2021-2034, USD Million/Billion
- By Phase- Market Size & Forecast 2021-2034, USD Million/Billion
- Japan Observational Virtual Clinical Trials Market Overview, 2021-2034F
- By Value (USD Million/Billion)
- By Indication- Market Size & Forecast 2021-2034, USD Million/Billion
- By Phase- Market Size & Forecast 2021-2034, USD Million/Billion
- Japan Expanded Access Virtual Clinical Trials Market Overview, 2021-2034F
- By Value (USD Million/Billion)
- By Indication- Market Size & Forecast 2021-2034, USD Million/Billion
- By Phase- Market Size & Forecast 2021-2034, USD Million/Billion
- Competitive Outlook (Company Profile - Partial List)
- ICON, plc
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- Parexel International (MA) Corporation
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- IQVIA Inc
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- Laboratory Corporation of America® Holdings
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- LEO Innovation LabLEO Innovation Lab
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- Medidata
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- Oracle
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- NOW Foods
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- Medable Inc
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- Others
- ICON, plc
- Contact Us & Disclaimer
Top Key Players & Market Share Outlook
- ICON, plc
- Parexel International (MA) Corporation
- IQVIA Inc
- Laboratory Corporation of America® Holdings
- LEO Innovation LabLEO Innovation Lab
- Medidata
- Oracle
- NOW Foods
- Medable Inc
- Others
Frequently Asked Questions






