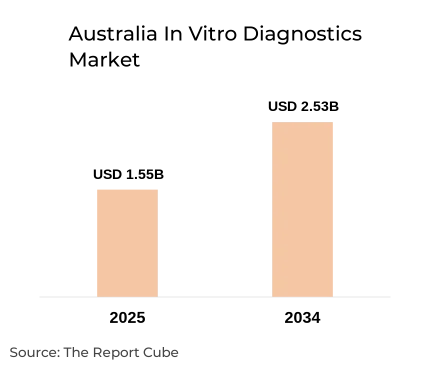
Build Your Custom Market Intelligence Report
Customize Your ReportAustralia In Vitro Diagnostics Market Insights & Analysis
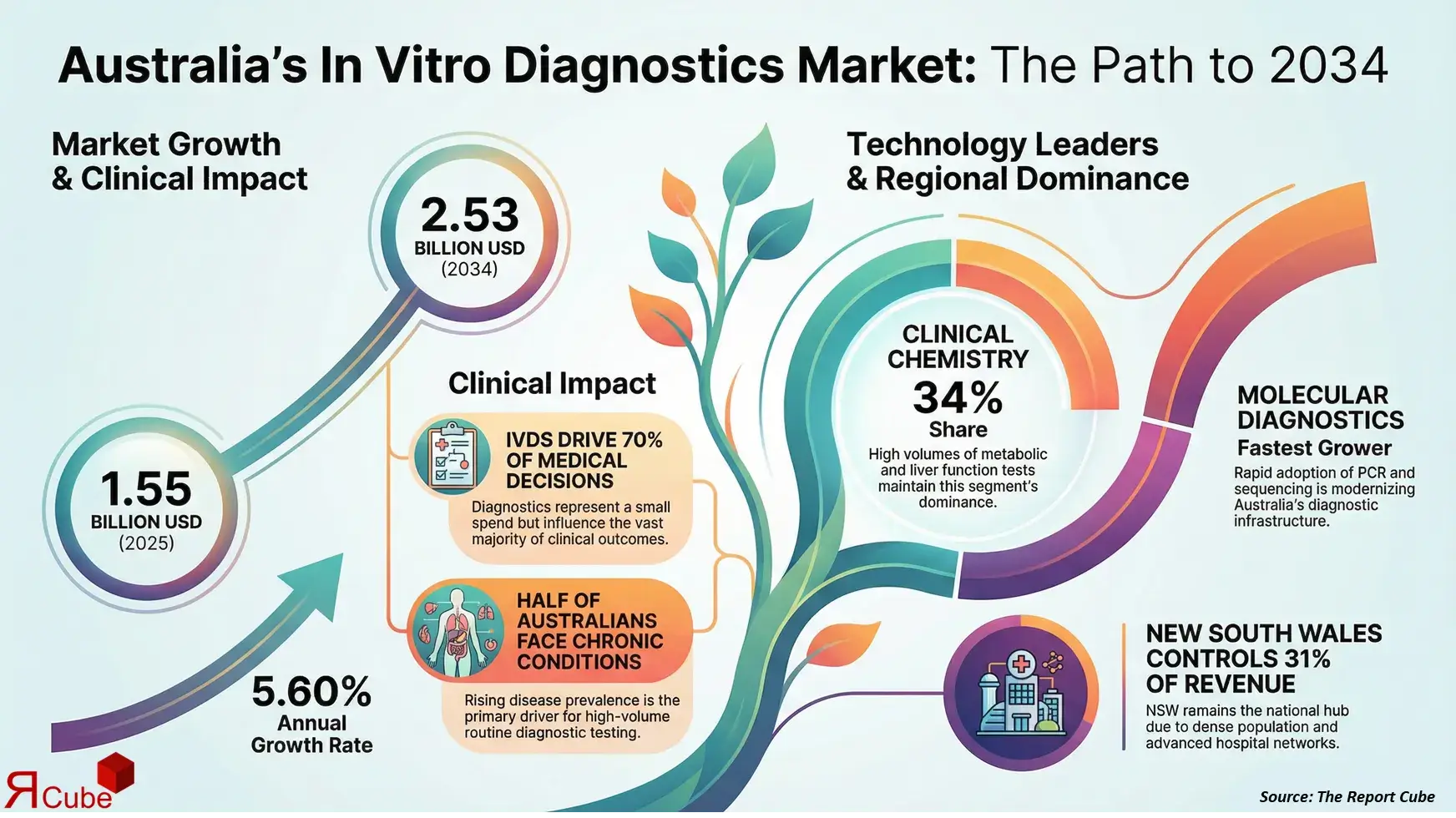
The Australia In Vitro Diagnostics Market is anticipated to register a CAGR of 5.60% during 2026-2034. The market size was valued at USD 1.55 billion in 2025 and is projected to reach USD 2.53 billion by 2034. The market is expanding steadily due to rising demand for early disease detection, increasing prevalence of chronic and infectious diseases, and strong adoption of advanced diagnostic technologies across Australia’s healthcare system. In vitro diagnostics play a critical role in clinical decision-making, accounting for nearly 70% of medical decisions while representing a relatively small share of total healthcare spending.
Australia’s well-established healthcare infrastructure, combined with high healthcare expenditure per capita, supports the widespread use of laboratory-based and point-of-care diagnostic solutions. Public and private pathology laboratories continue to invest in automation and high-throughput systems to improve efficiency and accuracy. At the same time, growing awareness of preventive healthcare and routine health screenings is driving test volumes across both hospital and independent diagnostic settings.
Furthermore, technological advancements in molecular diagnostics, immunoassays, and digital pathology are reshaping diagnostic workflows. The integration of data analytics and connected diagnostic platforms is improving turnaround times and clinical outcomes. These interconnected factors are expected to sustain long-term growth of the Australia In Vitro Diagnostics Market through 2034.
Australia In Vitro Diagnostics Market Recent Developments
- 2025: Roche Diagnostics expanded its automated immunoassay portfolio for oncology and infectious disease testing in Australia.
- 2025: Abbott Laboratories introduced next-generation point-of-care molecular testing systems for rapid diagnostics in remote regions.
Government Initiatives & Policies
- Medical Research Future Fund (MRFF): Supports diagnostics innovation and translational medical research across Australia.
- National Pathology Accreditation Advisory Council (NPAAC) Standards: Ensures quality, safety, and reliability of diagnostic testing nationwide.
Australia In Vitro Diagnostics Market Dynamics
Key Driver: Growing Burden of Chronic and Infectious Diseases
The rising burden of chronic and infectious diseases is a primary driver of the Australia In Vitro Diagnostics Market. Conditions such as cardiovascular diseases, diabetes, cancer, and autoimmune disorders continue to increase, requiring frequent diagnostic testing for early detection and disease monitoring. Nearly half of Australians live with at least one chronic condition, significantly boosting demand for routine laboratory tests. In parallel, periodic outbreaks of infectious diseases and seasonal illnesses sustain the need for rapid and accurate diagnostic solutions. This growing testing volume reinforces the importance of reliable in vitro diagnostics within both hospital-based and independent laboratory settings.
Book your FREE 30-minute expert consultation today
Contact UsIndustry Trends: Rapid Adoption of Molecular and High-Sensitivity Diagnostics
A key trend shaping the market is the accelerated adoption of molecular diagnostics and high-sensitivity testing platforms. Technologies such as PCR, next-generation sequencing, and advanced immunoassays are increasingly used for oncology, genetic screening, and infectious disease detection. These technologies offer higher accuracy and faster turnaround times compared to conventional methods. In 2025, molecular diagnostics accounted for approximately 28% of total IVD revenues in Australia. Laboratories are also investing in automation and digital connectivity to manage rising workloads while maintaining quality standards, further modernizing diagnostic infrastructure.
Major Challenge: High Cost of Advanced Diagnostic Technologies
The high cost associated with advanced diagnostic instruments and reagents presents a notable challenge for the Australia In Vitro Diagnostics Market. Molecular and automated platforms require significant capital investment, which can strain budgets of smaller laboratories and regional healthcare facilities. Additionally, ongoing costs related to consumables, maintenance, and skilled personnel add to operational expenses. Reimbursement limitations for certain advanced tests further restrict adoption, particularly in cost-sensitive settings. These financial barriers may slow the penetration of cutting-edge diagnostics outside major urban centers.
Opportunity: Expansion of Preventive and Personalized Medicine
The growing emphasis on preventive healthcare and personalized medicine offers substantial growth opportunities for the market. Increasing use of companion diagnostics, genetic testing, and biomarker-based assays enables tailored treatment decisions and improved patient outcomes. Preventive screening programs for cancer, cardiovascular diseases, and metabolic disorders are expanding nationwide, increasing test volumes. As healthcare providers focus on early intervention and precision medicine, demand for sophisticated in vitro diagnostic solutions is expected to rise steadily through the forecast period.
Australia In Vitro Diagnostics Market Segment-wise Analysis
By Technology:
- Immunoassay
- Clinical Chemistry
- Hematology
- Molecular Diagnostics
Clinical Chemistry leads the market by technology, accounting for approximately 34% of total market share in 2025. This dominance is driven by the high volume of routine tests related to metabolic panels, liver function, and renal assessment. These tests are fundamental to both preventive screening and chronic disease management, ensuring consistent demand. Immunoassays follow closely, supported by their widespread use in hormone testing, infectious disease detection, and oncology diagnostics.
By Test Type:
- Clinical Chemistry
- Immunodiagnostics
- Molecular Diagnostics
- Hematology Tests
By test type, Clinical Chemistry remains the leading segment, contributing nearly 36% of total test volumes in 2025. Its leadership is supported by frequent utilization across hospitals, diagnostic laboratories, and outpatient settings. Molecular diagnostics, while holding a smaller share, represent the fastest-growing segment due to increasing adoption in infectious disease and genetic testing. This balanced segment mix ensures both stability and innovation-driven growth within the market.
Regional Projection of Australia In Vitro Diagnostics Industry
- New South Wales
- Victoria
- Queensland
- Western Australia
New South Wales leads the Australia In Vitro Diagnostics Market, accounting for approximately 31% of total revenue in 2025. The region benefits from a dense population, advanced hospital networks, and a high concentration of diagnostic laboratories. Strong public and private healthcare spending supports early adoption of advanced diagnostic technologies. Victoria follows closely, driven by robust research activity and academic medical centers. While New South Wales remains dominant, Queensland is emerging as a high-growth region due to expanding healthcare infrastructure and rising diagnostic awareness.
Need insights for a specific region within this market?
Request Regional DataWhy Choose This Report?
- Provides a comprehensive overview of the overall market analysis, encompassing key trends, consumer behavior analysis, and risk assessment to support strategic decision-making.
- Provides accurate, up-to-date insights into market size, segmentation, and emerging opportunities, helping to minimize risk & capitalizing on growth.
- Gives deep understanding of target audience preferences, investment habits, and communication channels for enhanced product development & marketing effectiveness.
- Delivers competitive analysis & benchmarking, uncovering the strengths & weaknesses of market competitors to guide strategies.
- Consolidate comprehensive market intelligence, reducing reasoning & streamlining research efforts.
- Facilitates customized market segmentation & risk mitigation strategies, fine-tuned to the business objectives.
- Aids in identifying both market challenges & untapped opportunities within the industry to drive long-term business growth.
- Provides valuable information based on actual customer data & search trends.
Table of Contents
- Introduction
- Objective of the Study
- Product and Category Definition
- Market Segmentation
- Study Variables
- Research Methodology
- Secondary Data Points
- Breakdown of Secondary Sources
- Primary Data Points
- Breakdown of Primary Interviews
- Secondary Data Points
- Executive Summary
- Market Dynamics
- Drivers
- Challenges
- Opportunity Assessment
- Recent Trends and Developments
- Regulatory and Policy Landscape
- Australia In Vitro Diagnostics Market Overview (2020-2034)
- Market Size, By Value (USD Billion)
- Market Share, By Technology
- Immunoassay
- Instruments
- Reagents
- Others
- Clinical Chemistry
- Instruments
- Reagents
- Others
- Hematology
- Instruments
- Reagents
- Others Coagulation
- Instruments
- Reagents
- Others
- Molecular Diagnostics
- Instruments
- Reagents
- Others
- Microbiology
- Instruments
- Reagents
- Others
- Others
- Instruments
- Reagents
- Others
- Immunoassay
- Market Share, By Test Type
- Clinical Chemistry
- Immunodiagnostics
- Molecular Diagnostics
- Hematology Tests
- Other Tests
- Market Share, By Product Type
- Instruments
- Reagents
- Other Products
- Market Share, By Technology
- PCR
- NGS
- Immunoassay
- Clinical Chemistry
- Other Technologies
- Market Share, By Application
- Autoimmune Diseases
- Cardiology
- Diabetes
- Drug Testing
- Infectious Diseases
- Nephrology
- Oncology
- Other applications
- Market Share, By End User
- Diagnostic Laboratories
- Hospitals Clinics
- Other End Users
- Market Share, By Testing Mode
- Laboratory Testing
- Point-of-Care
- Home Care
- Others
- Market Share, By Region
- New South Wales
- Victoria
- Queensland
- Western Australia
- South Australia
- Tasmania
- Australian Capital Territory
- Northern Territory
- Market Share, By Company
- Market Share, By Company
- Competition Characteristics
- Revenue Shares & Analysis
- New South Wales IVD Market
- Market Size, By Value (USD Billion)
- Market Share, By Technology
- Market Share, By Test Type
- Market Share, By Product Type
- Market Share, By Application
- Market Share, By End User
- Market Share, By Testing Mode
- Victoria IVD Market
- Market Size, By Value (USD Billion)
- Market Share, By Technology
- Market Share, By Test Type
- Market Share, By Product Type
- Market Share, By Application
- Market Share, By End User
- Market Share, By Testing Mode
- Queensland IVD Market
- Market Size, By Value (USD Billion)
- Market Share, By Technology
- Market Share, By Test Type
- Market Share, By Product Type
- Market Share, By Application
- Market Share, By End User
- Market Share, By Testing Mode
- Western Australia IVD Market
- Market Size, By Value (USD Billion)
- Market Share, By Technology
- Market Share, By Test Type
- Market Share, By Product Type
- Market Share, By Application
- Market Share, By End User
- Market Share, By Testing Mode
- South Australia IVD Market
- Market Size, By Value (USD Billion)
- Market Share, By Technology
- Market Share, By Test Type
- Market Share, By Product Type
- Market Share, By Application
- Market Share, By End User
- Market Share, By Testing Mode
- Tasmania IVD Market
- Market Size, By Value (USD Billion)
- Market Share, By Technology
- Market Share, By Test Type
- Market Share, By Product Type
- Market Share, By Application
- Market Share, By End User
- Market Share, By Testing Mode
- Market Share, By Testing Mode
- Australian Capital Territory IVD Market
- Market Size, By Value (USD Billion)
- Market Share, By Technology
- Market Share, By Test Type
- Market Share, By Product Type
- Market Share, By Application
- Market Share, By End User
- Market Share, By Testing Mode
- Northern Territory IVD Market
- Market Size, By Value (USD Billion)
- Market Share, By Technology
- Market Share, By Test Type
- Market Share, By Product Type
- Market Share, By Application
- Market Share, By End User
- Market Share, By Testing Mode
- Competitive Outlook and Company Profiles
- Roche Diagnostics
- Company Overview
- Product Portfolio
- Strategic Alliances and Partnerships
- Recent Developments
- Abbott Laboratories
- Company Overview
- Product Portfolio
- Strategic Alliances and Partnerships
- Recent Developments
- Siemens Healthineers
- Company Overview
- Product Portfolio
- Strategic Alliances and Partnerships
- Recent Developments
- Thermo Fisher Scientific
- Company Overview
- Product Portfolio
- Strategic Alliances and Partnerships
- Recent Developments
- Danaher Corporation
- Company Overview
- Product Portfolio
- Strategic Alliances and Partnerships
- Recent Developments
- Becton Dickinson
- Company Overview
- Product Portfolio
- Strategic Alliances and Partnerships
- Recent Developments
- bioMérieux SA
- Company Overview
- Product Portfolio
- Strategic Alliances and Partnerships
- Recent Developments
- Sysmex Corporation
- Company Overview
- Product Portfolio
- Strategic Alliances and Partnerships
- Recent Developments
- QIAGEN NV
- Company Overview
- Product Portfolio
- Strategic Alliances and Partnerships
- Recent Developments
- Bio-Rad Laboratories
- Company Overview
- Product Portfolio
- Strategic Alliances and Partnerships
- Recent Developments
- Others
- Roche Diagnostics
- Contact Us and Disclaimer
Top Key Players & Market Share Outlook
- Roche Diagnostics
- Abbott Laboratories
- Siemens Healthineers
- Thermo Fisher Scientific
- Danaher Corporation
- Becton Dickinson
- bioMérieux SA
- Sysmex Corporation
- QIAGEN NV
- Bio-Rad Laboratories
- Others
Frequently Asked Questions






