Build Your Custom Market Intelligence Report
Customize Your ReportAustralia Adeno-associated Virus Gene Therapy Market Statistics and Insights
Market Insight
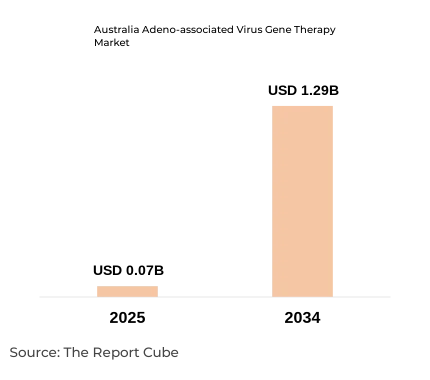
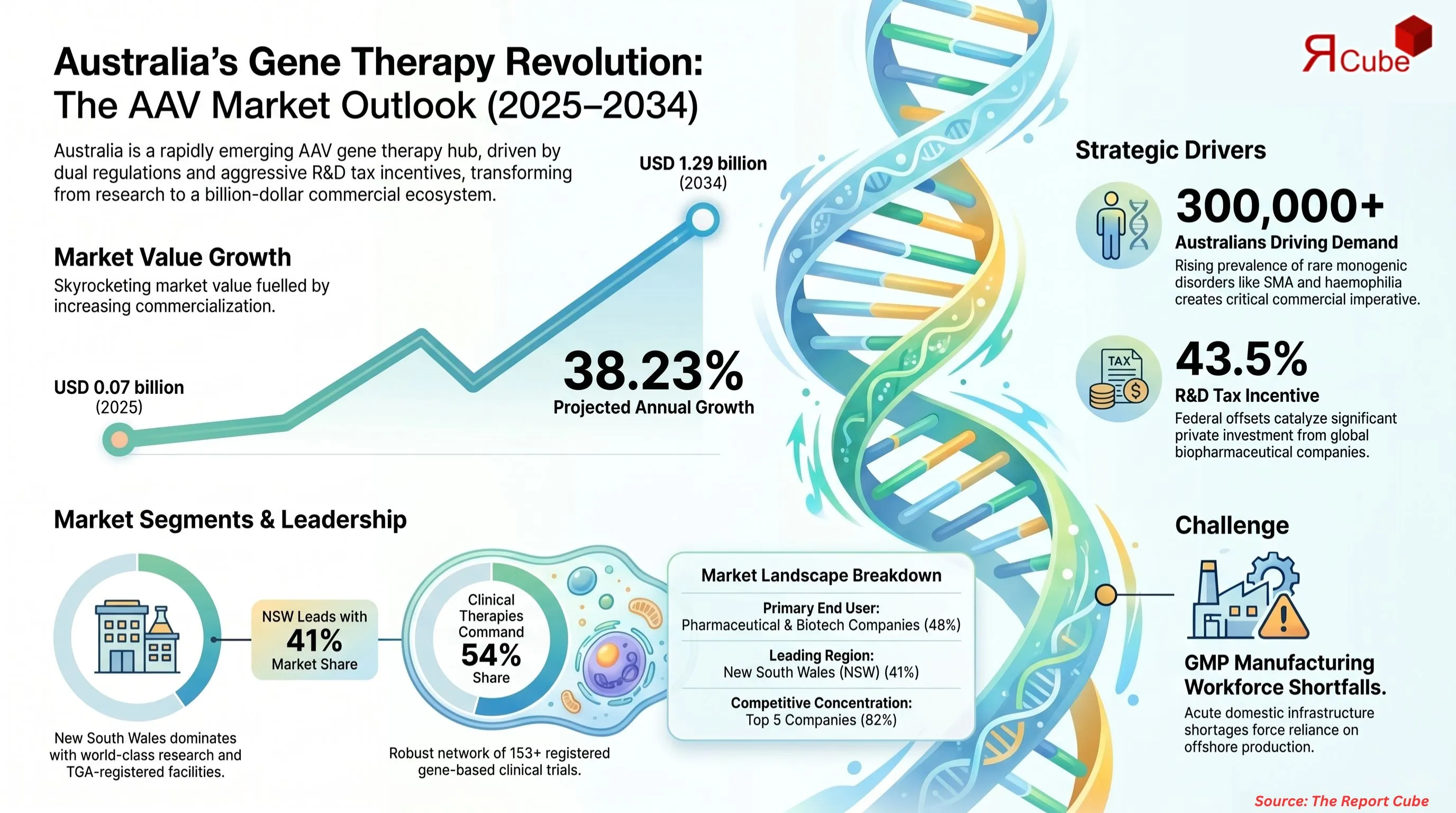
- The Australia Adeno-associated Virus Gene Therapy Market was valued at USD 0.07 billion in 2025.
- The market is projected to reach USD 1.29 billion by 2034.
- The market is forecast to expand at a CAGR of 38.23% during 2026–2034.
By End User
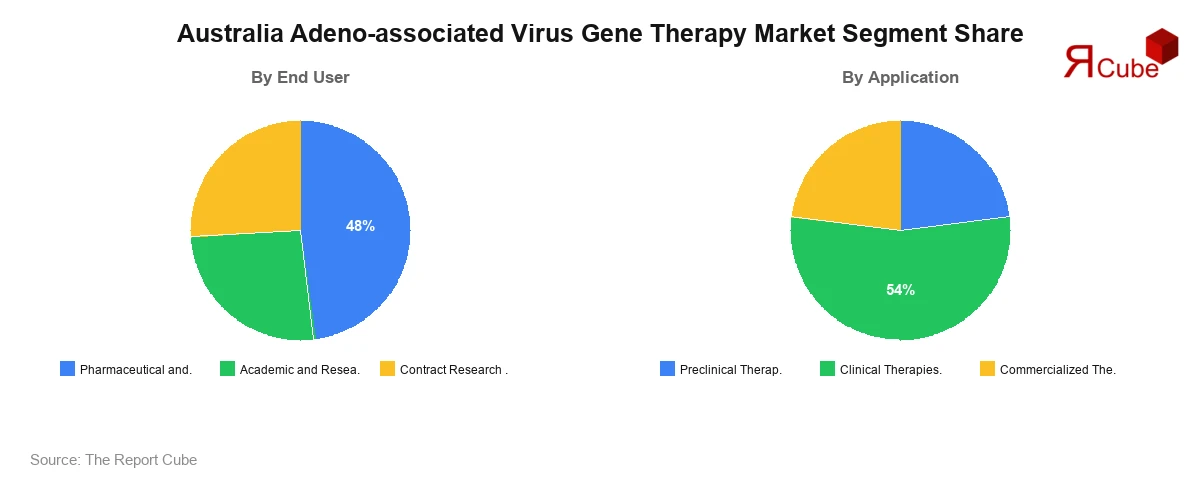
- The Pharmaceutical and Biotechnology Companies segment leads with approximately 48% market share.
Competitive Landscape
- More than 10 companies are actively engaged in producing Australia Adeno-associated Virus Gene Therapy products.
- Top 5 companies acquired around 62% of the market share.
- F. Hoffmann-La Roche Ltd., Sanofi S.A., Novartis AG, Astellas Gene Therapies Inc., Biogen Inc., and others.
By Application
- The Clinical Therapies segment dominates, accounting for approximately 54% market share.
Australia Adeno-associated Virus Gene Therapy Market Insights & Analysis
The Australia Adeno-associated Virus Gene Therapy Market is anticipated to register a 38.23% CAGR during 2026–2034. The market size was valued at USD 0.07 billion in 2025 and is projected to reach USD 1.29 billion by 2034. Australia has emerged as a compelling destination for AAV gene therapy development, supported by a well-established dual regulatory framework administered by the Therapeutic Goods Administration (TGA) and the Office of the Gene Technology Regulator (OGTR).
The country currently hosts over 153 registered clinical trials involving gene-based therapies, including pivotal AAV-based treatments such as Zolgensma and Luxturna, both approved by the TGA. Australia's federal R&D tax incentive scheme, offering offsets of up to 43.5% on eligible expenditure, has catalyzed significant private sector investment from global biopharmaceutical companies. The Australian Genomics Health Futures Mission has further earmarked dedicated funding to advance gene-related therapy research, with AU$5 million allocated specifically toward market-enabling research infrastructure under Aim 3 of the Gene-Related Therapies Incubator Project. These combined policy and financial dynamics continue to underpin the market's impressive growth trajectory.
Australia Adeno-associated Virus Gene Therapy Market Dynamics
Key Market Driver: Rising Prevalence of Rare Genetic Disorders
Australia's expanding rare disease patient population has created a compelling clinical and commercial imperative for AAV gene therapy solutions. Monogenic disorders such as spinal muscular atrophy (SMA), haemophilia A and B, and inherited retinal dystrophies are collectively diagnosed in more than 300,000 Australians, with limited conventional treatment alternatives.
AAV vectors, owing to their non-integrating nature, established safety record, and serotype versatility, are uniquely positioned to address these conditions through durable single-administration therapies. Clinical data from approved therapies like Zolgensma, which demonstrated sustained motor neuron recovery in SMA Type 1 patients, has strengthened physician and payer confidence in the platform, driving further investments into AAV-based pipeline candidates and supporting steady market expansion across research and commercial applications.
Major Industry Challenge: GMP Manufacturing Workforce and Capacity Shortfalls
One of the most structurally limiting challenges facing the Australia Adeno-associated Virus Gene Therapy Market is the acute shortage of Good Manufacturing Practice (GMP)-trained professionals and scalable domestic manufacturing infrastructure. The complexity of upstream AAV production, including baculovirus expression systems, triple transfection platforms, and high-titer purification protocols, requires highly specialized technical expertise that remains in short supply locally.
This constraint forces many Australian biotechnology companies and research institutions to rely on offshore contract development and manufacturing organizations (CDMOs), elevating logistical complexity and timelines. Inconsistent vector yields, stringent quality control thresholds, and the capital-intensive nature of GMP-compliant facilities further compound the challenge, limiting Australia's capacity to independently scale AAV therapy programs from early-stage research to commercial-grade production.
Emerging Trend Shaping Outlook: Next-Generation Capsid Engineering and Tropism Optimization
The Australia AAV gene therapy landscape is increasingly shaped by advanced capsid engineering technologies designed to overcome the immunogenicity and targeting limitations of natural AAV serotypes. Rational design approaches, including directed evolution, computational capsid modeling, and chimeric serotype construction, are enabling the development of novel vectors with enhanced tissue tropism, reduced neutralizing antibody reactivity, and improved transduction efficiency at lower doses.
Academic institutions such as the University of Sydney and the Centenary Institute are actively collaborating with biotechnology partners to explore these next-generation vector platforms. This trend not only broadens the therapeutic applicability of AAV across indications including neurological disorders and cardiology, but also positions Australia as a contributor to globally competitive intellectual property in the AAV vector development space
Need Specific Data, Let’s Customize the Report for You
Request CustomizationAustralia Adeno-associated Virus Gene Therapy Market Segment-wise Analysis
The Australia Adeno-associated Virus Gene Therapy Market is segmented by end user and application, each reflecting distinct demand dynamics, investment intensities, and developmental priorities. Understanding these segment-level distinctions is essential for stakeholders seeking to identify the most commercially viable and strategically significant growth pathways within the market.
By End User
- Pharmaceutical and Biotechnology Companies
- Academic and Research Institutions
- Contract Research Organizations (CROs)
Pharmaceutical and Biotechnology Companies constitute the dominant end-user segment, commanding approximately 48% of the Australia AAV gene therapy market in 2025. Companies such as Novartis, Biogen, and Sarepta Therapeutics have established active commercial and clinical presences in Australia, leveraging the country's streamlined TGA approval pathways and generous R&D tax incentive framework to advance their AAV pipeline programs. These enterprises drive the highest investment volumes in vector manufacturing, pre-clinical toxicology studies, and late-phase clinical trial execution.
Academic and Research Institutions represent the second-largest end-user category, accounting for approximately 33% of market revenues. Institutions such as the Centenary Institute and Murdoch University's Centre for Molecular Medicine and Innovative Therapeutics (CMMIT) are at the forefront of translational AAV research, particularly in haemophilia and neuromuscular disease. Contract Research Organizations (CROs) hold the remaining share, with organizations like Avance Clinical increasingly specialized in supporting cell and gene therapy study design, regulatory submissions, and APAC region site management, a growing niche as global sponsors expand AAV trial footprints into Australia.
By Application
- Preclinical Therapies
- Clinical Therapies
- Commercialized Therapies
Clinical Therapies represent the largest application segment, accounting for approximately 54% of the Australia AAV gene therapy market. Australia hosts a robust network of active AAV gene therapy trials across indications including SMA, haemophilia B, and retinal dystrophies, supported by the TGA's abbreviated Clinical Trial Notification (CTN) pathway that enables faster trial initiation compared to equivalent U.S. and European processes.
This regulatory efficiency attracts global sponsors seeking to generate internationally recognized clinical data at lower cost. Commercialized Therapies constitute approximately 28% of the market, driven by TGA-approved products including Zolgensma (Novartis), Luxturna (Novartis), and Hemgenix (CSL Behring), all of which leverage AAV vectors and are available through the Pharmaceutical Benefits Scheme (PBS) for eligible patient populations. Preclinical Therapies account for approximately 18% of revenues, reflecting Australia's growing role in early-stage AAV research supported by NHMRC grants and the Genomics Health Futures Mission funding.
Regional Projection of Australia Adeno-associated Virus Gene Therapy Market
The Australia Adeno-associated Virus Gene Therapy Market exhibits notable geographic concentration across its major biomedical precincts:
- Victoria
- Queensland
- Western Australia
- South Australia
New South Wales leads the regional landscape, accounting for approximately 41% of the national AAV gene therapy market. NSW's dominance is anchored by Sydney's concentration of world-class research institutions, including the Centenary Institute, University of Sydney, and the Children's Medical Research Institute, alongside a dense cluster of biopharmaceutical company offices and TGA-registered manufacturing facilities. Victoria follows as the second-largest region, driven by Melbourne's established biotech ecosystem and the Doherty Institute's contributions to infectious disease-targeted gene therapy research. Queensland and Western Australia are emerging contributors, benefiting from state government-backed life sciences investment strategies and growing university-industry partnerships.
Australia Adeno-associated Virus Gene Therapy Market: Recent Development (2025)
- Novartis reported Zolgensma achieving over AUD 120 million in Australian reimbursed prescriptions, marking a significant milestone.
- Sarepta Therapeutics expanded its Australian CRO partnerships to support two new AAV-based Duchenne muscular dystrophy trial sites.
- Oxford BioMedica disclosed a 22% increase in vector manufacturing margins following process optimization across its CDMO client portfolio.
- Biogen and the University of Sydney announced a collaborative AAV research agreement targeting neurodegenerative disease gene delivery.
Australia Adeno-associated Virus Gene Therapy Market Future Outlook (2034)
By 2034, the Australia Adeno-associated Virus Gene Therapy Market is projected to reach USD 1.29 billion, expanding at a robust CAGR of 38.23% from 2026. This trajectory will be shaped by the continued commercialization of TGA-approved AAV therapies across rare disease indications, growing adoption of engineered capsid platforms, and the expansion of Australia's CDMO sector for scalable AAV manufacturing. AI-powered genomic analysis and predictive vector design tools are expected to accelerate candidate selection and trial design, compressing development timelines significantly. Federal policy support, through continued NHMRC funding, the Genomics Health Futures Mission, and R&D tax incentive refinements, will sustain long-term ecosystem investment, positioning Australia as a leading Asia-Pacific hub for next-generation AAV gene therapy development and clinical delivery by the end of the forecast period.
Why Choose This Report?
- Provides a comprehensive overview of the overall market analysis, encompassing key trends, consumer behavior analysis, and risk assessment to support strategic decision-making.
- Provides accurate, up-to-date insights into market size, segmentation, and emerging opportunities, helping to minimize risk & capitalizing on growth.
- Gives deep understanding of target audience preferences, investment habits, and communication channels for enhanced product development & marketing effectiveness.
- Delivers competitive analysis & benchmarking, uncovering the strengths & weaknesses of market competitors to guide strategies.
- Consolidate comprehensive market intelligence, reducing reasoning & streamlining research efforts.
- Facilitates customized market segmentation & risk mitigation strategies, fine-tuned to the business objectives.
- Aids in identifying both market challenges & untapped opportunities within the industry to drive long-term business growth.
- Provides valuable information based on actual customer data & search trends.
Table of Contents
- Introduction
- Objective of the study
- Product Definition
- Market Segmentation
- Study Variables
- Research Methodology
- Secondary Data Points
- Companies Interviewed
- Primary Data Points
- Breakdown of Primary Interviews
- Secondary Data Points
- Executive Summary
- Market Dynamics
- Drivers
- Challenges
- Opportunity Assessment
- Recent Trends and Developments
- Policy and Regulatory Landscape
- Australia Adeno-associated Virus Gene Therapy Market Overview and Forecast Analysis (2021-2034)
- Market Size, By Value, By growth rate (CAGR/USD Billions)
- Demand - Supply Trends
- Market Share, By Therapeutic Area
- Neurological Disorders
- Ophthalmic Disorders
- Hematologic Disorders
- Muscular Disorders
- Metabolic Disorders
- Rare Genetic Disorders
- Oncology/Cancer
- Market Share, By Vector Serotype
- AAV1
- AAV2
- AAV3
- AAV4
- AAV5
- AAV6
- AAV7
- AAV8
- AAV9
- AAV10
- AAV11
- AAV12
- Engineered/Synthetic/Hybrid Capsids
- Market Share, By Route of Administration
- Intravenous (I.V.)
- Intrathecal (I.T.)
- Intraocular (I.O.)
- Intramuscular (I.M.)
- Intracerebral
- Subcutaneous
- Local/Direct Injection
- Market Share, By Application
- Preclinical Therapies
- Clinical Therapies
- Commercialized Therapies
- Market Share, By Manufacturing Type
- In-House Manufacturing
- CDMOs/Vector Production Facilities
- Market Share, By End User
- Pharmaceutical and Biotechnology Companies
- Academic and Research Institutions
- Contract Research Organizations (CROs)
- Market Share, By Region
- Victoria
- Queensland
- Western Australia
- South Australia
- Market Share, By Competitors
- Competition Characteristics
- Revenue Shares
- Australia Neurological Disorders Adeno-associated Virus Gene Therapy Market Overview, 2021-2034F
- By Value (USD Million)
- By Vector Serotype- Market Size & Forecast 2021-2034, USD Million
- By Route of Administration- Market Size & Forecast 2021-2034, USD Million
- By Application- Market Size & Forecast 2021-2034, USD Million
- By Manufacturing Type- Market Size & Forecast 2021-2034, USD Million
- By End User- Market Size & Forecast 2021-2034, USD Million
- Australia Ophthalmic Disorders Adeno-associated Virus Gene Therapy Market Overview, 2021-2034F
- By Value (USD Million)
- By Vector Serotype- Market Size & Forecast 2021-2034, USD Million
- By Route of Administration- Market Size & Forecast 2021-2034, USD Million
- By Application- Market Size & Forecast 2021-2034, USD Million
- By Manufacturing Type- Market Size & Forecast 2021-2034, USD Million
- By End User- Market Size & Forecast 2021-2034, USD Million
- Australia Hematologic Disorders Adeno-associated Virus Gene Therapy Market Overview, 2021-2034F
- By Value (USD Million)
- By Vector Serotype- Market Size & Forecast 2021-2034, USD Million
- By Route of Administration- Market Size & Forecast 2021-2034, USD Million
- By Application- Market Size & Forecast 2021-2034, USD Million
- By Manufacturing Type- Market Size & Forecast 2021-2034, USD Million
- By End User- Market Size & Forecast 2021-2034, USD Million
- Australia Muscular Disorders Adeno-associated Virus Gene Therapy Market Overview, 2021-2034F
- By Value (USD Million)
- By Vector Serotype- Market Size & Forecast 2021-2034, USD Million
- By Route of Administration- Market Size & Forecast 2021-2034, USD Million
- By Application- Market Size & Forecast 2021-2034, USD Million
- By Manufacturing Type- Market Size & Forecast 2021-2034, USD Million
- By End User- Market Size & Forecast 2021-2034, USD Million
- Australia Metabolic Disorders Adeno-associated Virus Gene Therapy Market Overview, 2021-2034F
- By Value (USD Million)
- By Vector Serotype- Market Size & Forecast 2021-2034, USD Million
- By Route of Administration- Market Size & Forecast 2021-2034, USD Million
- By Application- Market Size & Forecast 2021-2034, USD Million
- By Manufacturing Type- Market Size & Forecast 2021-2034, USD Million
- By End User- Market Size & Forecast 2021-2034, USD Million
- Australia Rare Genetic Disorders Adeno-associated Virus Gene Therapy Market Overview, 2021-2034F
- By Value (USD Million)
- By Vector Serotype- Market Size & Forecast 2021-2034, USD Million
- By Route of Administration- Market Size & Forecast 2021-2034, USD Million
- By Application- Market Size & Forecast 2021-2034, USD Million
- By Manufacturing Type- Market Size & Forecast 2021-2034, USD Million
- By End User- Market Size & Forecast 2021-2034, USD Million
- Australia Oncology/Cancer Adeno-associated Virus Gene Therapy Market Overview, 2021-2034F
- By Value (USD Million)
- By Vector Serotype- Market Size & Forecast 2021-2034, USD Million
- By Route of Administration- Market Size & Forecast 2021-2034, USD Million
- By Application- Market Size & Forecast 2021-2034, USD Million
- By Manufacturing Type- Market Size & Forecast 2021-2034, USD Million
- By End User- Market Size & Forecast 2021-2034, USD Million
- Competitive Outlook (Company Profile - Partial List)
- F. Hoffmann-La Roche Ltd.
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- Sanofi S.A.
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- Novartis AG
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- Astellas Gene Therapies Inc.
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- Biogen Inc.
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- Sarepta Therapeutics Inc.
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- Oxford BioMedica plc
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- Others
- F. Hoffmann-La Roche Ltd.
- Contact Us & Disclaimer
Top Key Players & Market Share Outlook
- F. Hoffmann-La Roche Ltd.
- Sanofi S.A.
- Novartis AG
- Astellas Gene Therapies Inc.
- Biogen Inc.
- Sarepta Therapeutics Inc.
- Oxford BioMedica plc
- Others
Frequently Asked Questions






