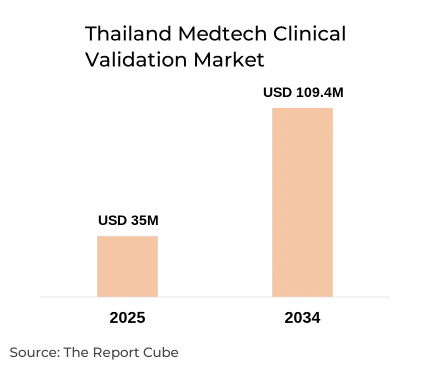
Build Your Custom Market Intelligence Report
Customize Your ReportThailand Medtech Clinical Validation Market Key Highlights
By Validation Phase:
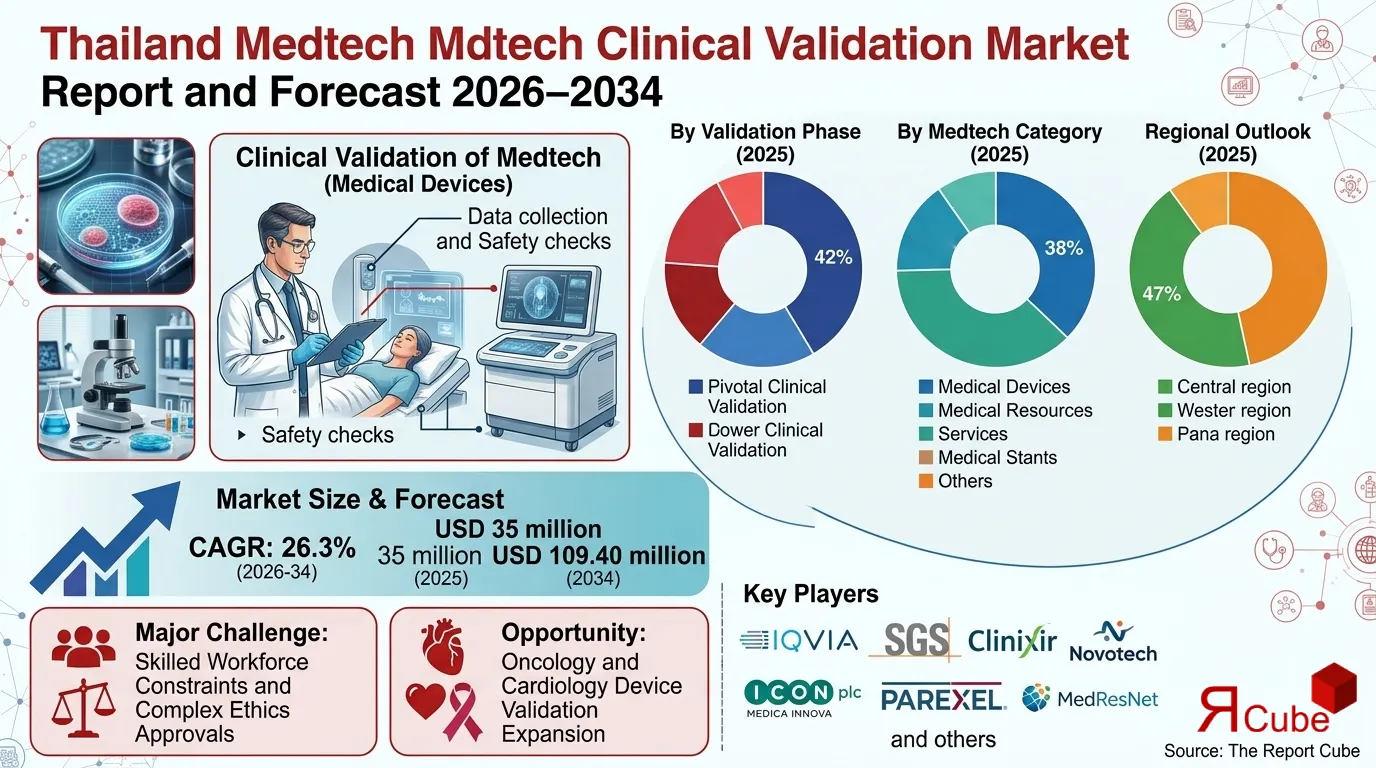
Pivotal Clinical Validation segment leads the market, with around 42% share.
By Medtech Category:
Medical Devices segment dominates the market, accounting for nearly 38% of total revenue.
Regional Outlook:
Central region dominates the thailand medtech clinical validation market, with around 47% share.
Government Initiatives & Policies
- Thailand 4.0 and S-Curve Medical Hub Policy (Updated 2026 Roadmap): Government strategy promoting advanced medical technology development, foreign clinical trial investments, and innovation clusters.
- Thai FDA Medical Device Act B.E. 2551 (Amended Guidelines 2025): Strengthened post-market surveillance and clinical evidence requirements for medical devices and digital health solutions.
Thailand Medtech Clinical Validation Market Insights & Analysis
The Thailand Medtech Clinical Validation Market is anticipated to register a CAGR: 26.3% during 2026-34. The market size was valued at USD 35 million in 2025 and is projected to reach USD 109.40 million by 2034. Thailand’s ambition to become a regional medical innovation hub, combined with regulatory modernization and expanding healthcare infrastructure, is accelerating demand for structured medtech clinical validation services.
Thailand has positioned itself as a clinical research gateway for Southeast Asia, supported by a strong network of tertiary hospitals, experienced investigators, and cost-efficient trial execution. Over 1,200 active clinical studies were registered in Thailand by 2025, with a rising share dedicated to medical devices, digital therapeutics, and in vitro diagnostics. The Thai FDA’s alignment with ASEAN Medical Device Directive standards has enhanced regulatory clarity, reducing approval timelines for clinical study applications by nearly 20% compared to 2022 benchmarks.
Growing complexity in medical devices, particularly AI-enabled diagnostics and robotic-assisted systems, is reshaping validation frameworks. Sponsors increasingly require multi-phase validation strategies, including feasibility pilots, pivotal trials, and post-market clinical follow-up (PMCF). The expansion of universal healthcare coverage in Thailand, serving over 70% of the population, also facilitates diverse patient recruitment pools, improving data robustness for global regulatory submissions.
Investment inflows from multinational contract research organizations (CROs) such as IQVIA (Thailand), Parexel (Thailand), ICON plc, and Novotech (Thailand) are expanding site management networks and data analytics capabilities. At the same time, local CROs and academic partnerships are strengthening decentralized and hybrid study models. With digital health adoption accelerating, particularly telemedicine and wearable monitoring systems, Thailand’s medtech clinical validation ecosystem is evolving into a high-growth, high-complexity service industry.
Thailand Medtech Clinical Validation Market Dynamics
Key Driver: Regulatory Modernization and Medical Hub Strategy
Thailand’s medical hub policy and regulatory streamlining are major growth drivers. The Thai FDA has introduced clearer clinical evidence pathways for software as a medical device (SaMD) and digital health platforms, encouraging foreign sponsors to conduct validation studies locally. Approval timelines for device trial protocols have improved, with average review durations declining from 90 days in 2022 to approximately 65-70 days in 2025. Incentives under the Board of Investment (BOI) provide tax exemptions for R&D activities and clinical research operations. This policy framework is attracting multinational medtech firms seeking regional validation sites that meet global compliance standards while maintaining cost efficiency.
Book your FREE 30-minute expert consultation today
Contact UsIndustry Trends: Rise of Digital Health and Hybrid Study Designs
Digital health solutions, including remote monitoring wearables and AI-based diagnostics, are rapidly expanding within Thailand’s validation pipeline. By 2025, digital and software-based devices represented nearly 28% of newly submitted clinical validation applications. Hybrid and decentralized study models are gaining traction, allowing remote data capture and real-time monitoring. Real-world evidence (RWE) studies are increasingly integrated into post-market validation strategies, particularly for chronic disease management technologies. These shifts are encouraging CROs to invest in advanced data management systems, cloud-based trial platforms, and AI-driven patient recruitment tools, improving efficiency and study scalability.
Major Challenge: Skilled Workforce Constraints and Complex Ethics Approvals
Although Thailand offers strong clinical infrastructure, the rapid rise in medtech studies is creating pressure on experienced clinical research coordinators and biostatisticians. Demand for specialized device validation expertise has increased by over 30% since 2023. Ethics committee review processes can vary between institutions, occasionally extending study initiation timelines. Multisite coordination challenges, particularly for complex robotic or implantable device trials, require enhanced technical oversight. Addressing these constraints requires expanded workforce training programs and standardized review frameworks across regions.
Opportunity: Oncology and Cardiology Device Validation Expansion
Cardiology and oncology remain priority therapeutic areas, accounting for nearly 40% of active medtech clinical validation studies. Rising non-communicable disease prevalence, including cardiovascular conditions affecting over 9% of Thailand’s adult population, supports strong demand for innovative devices. Advanced imaging systems, minimally invasive robotic platforms, and AI-enabled diagnostics are entering pivotal validation phases. Additionally, collaboration between universities and CROs is enhancing early-phase feasibility studies. Thailand’s ability to deliver high patient enrollment rates and strong retention performance creates attractive opportunities for sponsors targeting Asia-Pacific market entry.
Thailand Medtech Clinical Validation Market Segment-wise Analysis
By Validation Phase:
- Feasibility and Pilot Validation
- Pivotal Clinical Validation
- Post-Market Clinical Follow-up
Pivotal clinical validation accounts for approximately 42% of total market revenue in 2025. These large-scale confirmatory trials are essential for regulatory submissions across ASEAN, the EU, and the US. Thailand’s tertiary hospitals in Bangkok and surrounding provinces support high patient enrollment capacity, with average recruitment rates 15-20% faster than regional averages. Sponsors favor Thailand for pivotal trials involving implantable cardiac devices, advanced diagnostic systems, and robotic surgical platforms due to strong investigator expertise and structured monitoring frameworks.
By Medtech Category:
- Medical Devices
- In Vitro Diagnostics
- Digital Health Solutions
- Software as a Medical Device
- Wearables and Remote Monitoring
- Robotic and Minimally Invasive Systems
Medical devices represent nearly 38% of total validation activity, driven by demand for cardiovascular implants, orthopedic solutions, and minimally invasive systems. In vitro diagnostics and digital health solutions are rapidly expanding, particularly AI-powered imaging tools and remote patient monitoring platforms. Robotic-assisted surgical systems are entering multi-center validation studies, reflecting Thailand’s advanced hospital infrastructure. The integration of software validation requirements is also increasing complexity, prompting CROs to enhance cybersecurity, interoperability testing, and data integrity protocols.
Regional Projection of Thailand Medtech Clinical Validation Industry
- Northern
- Northeastern (Isan)
- Central
- Eastern
- Western
- Southern
The Central region, including Bangkok and surrounding provinces, leads with approximately 47% market share. Major teaching hospitals, research universities, and regulatory headquarters are concentrated in this region, enabling efficient study coordination and regulatory interactions. The Eastern Economic Corridor (EEC) is emerging as a medtech manufacturing and innovation zone, supporting clinical validation activities. Northern and Northeastern regions are expanding participation through university hospital networks, enhancing geographic diversity in patient recruitment. Southern Thailand contributes to infectious disease and tropical medicine device trials, leveraging regional healthcare strengths.
Thailand Medtech Clinical Validation Market Recent Developments
- 2025: Clinixir signed a strategic Memorandum of Understanding with Burapha University to strengthen the BUU-CRC and expand high-impact clinical research capacity, enhancing Thailand’s institutional collaboration ecosystem.
- 2025: Clinixir advanced a multinational Phase II/III immunotherapy trial for hepatocellular carcinoma across nine regional sites, enrolling over 90% of targeted patients, demonstrating strong oncology trial execution capabilities.
- 2025: Novotech (Thailand) participated in the Clinical Trials Innovation Program 2025, highlighting its end-to-end regulatory and trial optimization expertise to accelerate medtech validation across Asia-Pacific.
Need insights for a specific region within this market?
Request Regional DataWhy Choose This Report?
- Provides a comprehensive overview of the overall market analysis, encompassing key trends, consumer behavior analysis, and risk assessment to support strategic decision-making.
- Provides accurate, up-to-date insights into market size, segmentation, and emerging opportunities, helping to minimize risk & capitalizing on growth.
- Gives deep understanding of target audience preferences, investment habits, and communication channels for enhanced product development & marketing effectiveness.
- Delivers competitive analysis & benchmarking, uncovering the strengths & weaknesses of market competitors to guide strategies.
- Consolidate comprehensive market intelligence, reducing reasoning & streamlining research efforts.
- Facilitates customized market segmentation & risk mitigation strategies, fine-tuned to the business objectives.
- Aids in identifying both market challenges & untapped opportunities within the industry to drive long-term business growth.
- Provides valuable information based on actual customer data & search trends.
Table of Contents
- Introduction
- Objective of the Study
- Product and Category Definition
- Scope of Medtech Clinical Validation
- Market Segmentation
- Study Variables and Assumptions
- Research Methodology
- Secondary Data Points
- Breakdown of Secondary Sources
- Primary Data Points
- Breakdown of Primary Interviews
- Data Triangulation and Forecasting Approach
- Secondary Data Points
- Executive Summary
- Market Dynamics
- Drivers
- Challenges
- Opportunity Assessment
- Recent Trends and Developments
- Regulatory and Policy Landscape
- Thailand Medtech Clinical Validation Market Overview (2021-2034)
- Market Size, By Value (USD Million)
- Market Share, By Validation Phase
- Feasibility and Pilot Validation
- Pivotal Clinical Validation
- Post‑Market Clinical Follow‑up
- Market Share, By Medtech Category
- Medical Devices
- In Vitro Diagnostics
- Digital Health Solutions
- Software as a Medical Device
- Wearables and Remote Monitoring
- Robotic and Minimally Invasive Systems
- Market Share, By Therapeutic Area
- Cardiology
- Oncology
- Neurology
- Orthopedics
- Endocrinology and Metabolic Disorders
- Infectious Diseases
- Others
- Market Share, By Study Design
- Interventional Studies
- Observational Studies
- Real‑World Evidence Studies
- Registries
- Market Share, By Sponsor Type
- Medtech Manufacturers
- Pharmaceutical and Biotech Companies
- Hospitals and Health Systems
- Academic and Research Institutions
- Public and Non‑profit Organizations
- Market Share, By Service Type
- Regulatory Strategy and Planning
- Clinical Trial Design and Protocol Development
- Site Identification and Management
- Patient Recruitment and Retention
- Data Management and Biostatistics
- Safety and Pharmacovigilance
- Real‑World Evidence and Health Economics
- Quality Management and Compliance
- Market Share, By End User
- Multinational Medtech Companies
- Local Medtech Manufacturers
- Digital Health and Health‑Tech Startups
- Contract Research Organizations
- Hospitals and Clinics
- Market Share, By Region
- Northern
- Northeastern (Isan)
- Central
- Eastern
- Western
- Southern
- Market Share, By Company
- Revenue Shares and Analysis
- Competitive Landscape
- Northern Thailand Medtech Clinical Validation Market
- Market Size, By Value (USD Million)
- Market Share, By Validation Phase
- Market Share, By Medtech Category
- Market Share, By Therapeutic Area
- Market Share, By Study Design
- Market Share, By Sponsor Type
- Market Share, By Service Type
- Market Share, By End User
- Northeastern (Isan) Thailand Medtech Clinical Validation Market
- Market Size, By Value (USD Million)
- Market Share, By Validation Phase
- Market Share, By Medtech Category
- Market Share, By Therapeutic Area
- Market Share, By Study Design
- Market Share, By Sponsor Type
- Market Share, By Service Type
- Market Share, By End User
- Central Thailand Medtech Clinical Validation Market
- Market Size, By Value (USD Million)
- Market Share, By Validation Phase
- Market Share, By Medtech Category
- Market Share, By Therapeutic Area
- Market Share, By Study Design
- Market Share, By Sponsor Type
- Market Share, By Service Type
- Market Share, By End User
- Eastern Thailand Medtech Clinical Validation Market
- Market Size, By Value (USD Million)
- Market Share, By Validation Phase
- Market Share, By Medtech Category
- Market Share, By Therapeutic Area
- Market Share, By Study Design
- Market Share, By Sponsor Type
- Market Share, By Service Type
- Market Share, By End User
- Western Thailand Medtech Clinical Validation Market
- Market Size, By Value (USD Million)
- Market Share, By Validation Phase
- Market Share, By Medtech Category
- Market Share, By Therapeutic Area
- Market Share, By Study Design
- Market Share, By Sponsor Type
- Market Share, By Service Type
- Market Share, By End User
- Southern Thailand Medtech Clinical Validation Market
- Market Size, By Value (USD Million)
- Market Share, By Validation Phase
- Market Share, By Medtech Category
- Market Share, By Therapeutic Area
- Market Share, By Study Design
- Market Share, By Sponsor Type
- Market Share, By Service Type
- Market Share, By End User
- Competitive Outlook and Company Profiles
- IQVIA
- Company Overview
- Product Portfolio
- Strategic Alliances/Partnerships
- Recent Developments
- SGS
- Company Overview
- Product Portfolio
- Strategic Alliances/Partnerships
- Recent Developments
- Clinixir
- Company Overview
- Product Portfolio
- Strategic Alliances/Partnerships
- Recent Developments
- Novotech
- Company Overview
- Product Portfolio
- Strategic Alliances/Partnerships
- Recent Developments
- ICON plc
- Company Overview
- Product Portfolio
- Strategic Alliances/Partnerships
- Recent Developments
- Medica Innova
- Company Overview
- Product Portfolio
- Strategic Alliances/Partnerships
- Recent Developments
- Parexel
- Company Overview
- Product Portfolio
- Strategic Alliances/Partnerships
- Recent Developments
- MedResNet
- Company Overview
- Product Portfolio
- Strategic Alliances/Partnerships
- Recent Developments
- PPD (Thermo Fisher)
- Company Overview
- Product Portfolio
- Strategic Alliances/Partnerships
- Recent Developments
- Artixio
- Company Overview
- Product Portfolio
- Strategic Alliances/Partnerships
- Recent Developments
- Syneos Health
- Company Overview
- Product Portfolio
- Strategic Alliances/Partnerships
- Recent Developments
- B. Braun
- Company Overview
- Product Portfolio
- Strategic Alliances/Partnerships
- Recent Developments
- Fortrea
- Company Overview
- Product Portfolio
- Strategic Alliances/Partnerships
- Recent Developments
- Cloud 9 Consulting
- Company Overview
- Product Portfolio
- Strategic Alliances/Partnerships
- Recent Developments
- Everlife-RI Tech
- Company Overview
- Product Portfolio
- Strategic Alliances/Partnerships
- Recent Developments
- PSI CRO
- Company Overview
- Product Portfolio
- Strategic Alliances/Partnerships
- Recent Developments
- Others
- IQVIA
- Contact Us and Disclaimer
Top Key Players & Market Share Outlook
- IQVIA (Thailand)
- SGS (Thailand)
- Clinixir
- Novotech (Thailand)
- ICON plc
- Medica Innova
- Parexel (Thailand)
- MedResNet
- PPD (Thermo Fisher)
- Artixio (Thailand)
- Syneos Health
- B. Braun (Thailand)
- Fortrea (Thailand)
- Cloud 9 Consulting
- Everlife-RI Tech
- PSI CRO
Frequently Asked Questions






