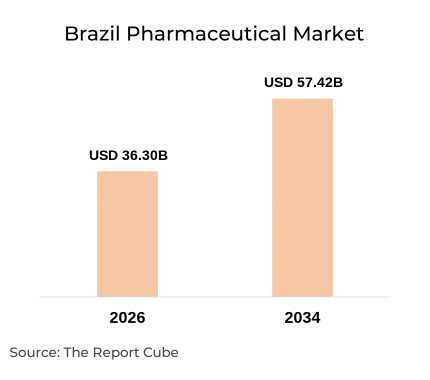
Build Your Custom Market Intelligence Report
Customize Your ReportKey Takeaways - Brazil Pharmaceutical Market
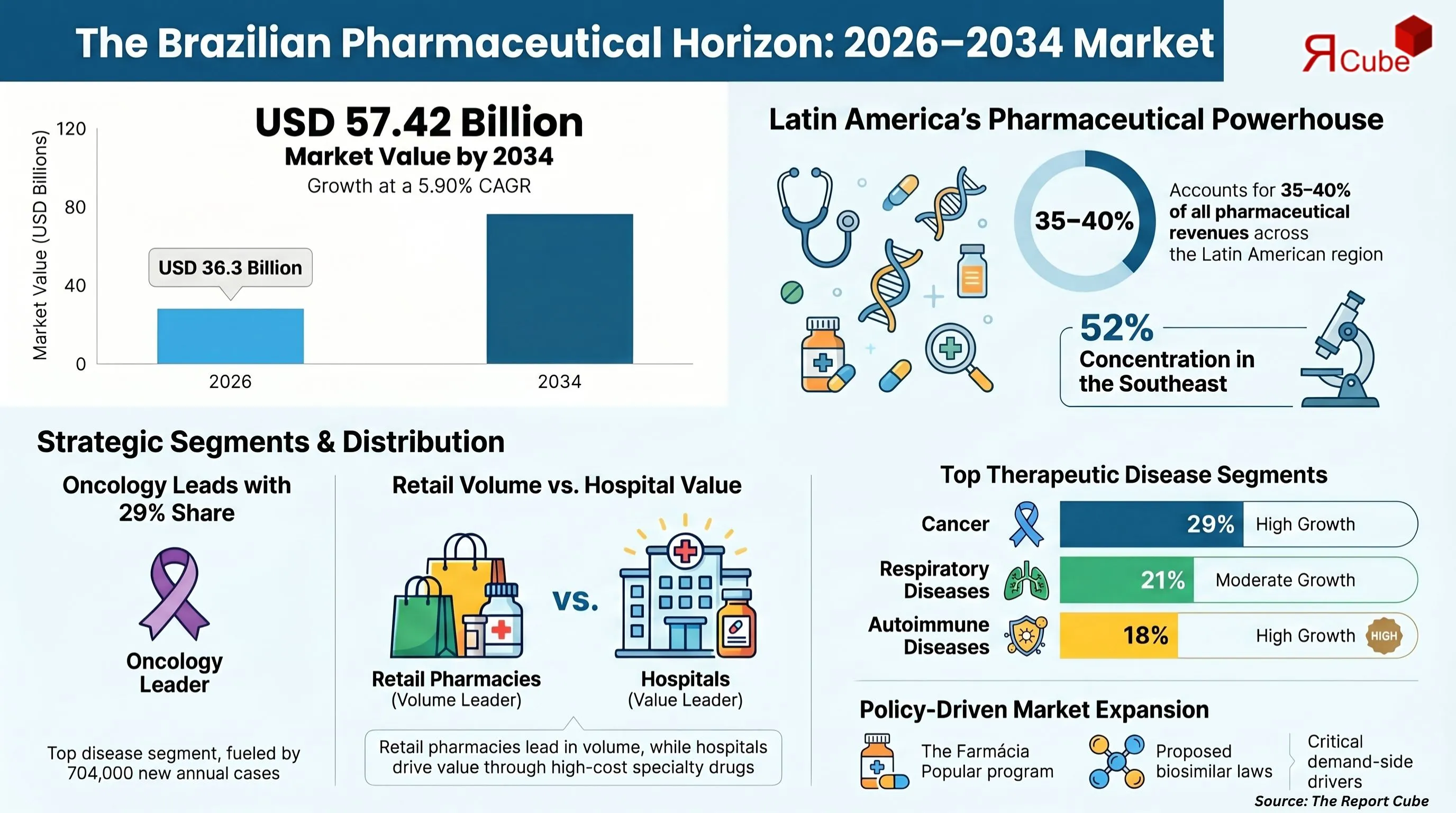
- The market is set to grow from USD 36.3 billion in 2026 to USD 57.42 billion by 2034, registering a steady 5.90% CAGR.
- Hospital pharmacies dominate the distribution landscape in terms of value, driven by high-cost specialty drugs, while retail pharmacies lead in volume with over 90,000 licensed outlets.
- Cancer remains the leading disease segment, accounting for 29% of total market share, supported by a rising patient pool and increasing oncology drug demand.
- Brazil stands as the largest pharmaceutical market in Latin America, contributing 35-40% of regional revenues, reinforcing its strategic importance for global pharma players.
- The Southeast region controls 52% of the national market, largely due to strong private hospital infrastructure and higher healthcare spending.
- Key industry leaders include Pfizer, Roche, Novartis, AbbVie, and Sanofi, shaping innovation, pricing strategies, and competitive dynamics.
- The market is regulated by ANVISA, ensuring drug safety, approvals, and compliance across the country.
- Government initiatives like the Farmácia Popular Programme continue to expand access to essential medicines, acting as a major policy driver.
- The proposed mandatory biosimilar substitution bill (PL 2.521/2023) could significantly reduce biologic drug costs by 25-40%, making it one of the most impactful policy shifts expected during the forecast period.
Brazil Pharmaceutical Market Insights & Analysis
Brazil's pharmaceutical sector faces mounting pressure to serve a population of 215 million with an ageing demographic and a rising double burden of communicable and non-communicable diseases. With the government's public drug procurement programme - Farmácia Popular - expanding and private insurance penetration still below 30% of the population (ANS, 2024), unmet medical need is pushing demand at a pace the current supply chain struggles to match. This section maps the market's structural fundamentals: size, trajectory, and the forces underpinning its 5.90% CAGR through 2034.
The Brazil pharmaceutical market is valued at USD 36.3 billion in 2026 and is projected to reach USD 57.42 billion by 2034, expanding at a CAGR of 5.90% (Report Publisher, 2026). Brazil ranks as the Latin America pharmaceutical market's largest national contributor, accounting for roughly 35–40% of regional pharma revenues (ANVISA, 2024). ANVISA regulatory reforms accelerating drug approvals since the 2023 legislative amendment (Brazilian Congress, 2023) are reducing dossier review backlogs for priority molecules. A genericisation rate exceeding 70% of primary care prescriptions (IQVIA Brazil, 2024) keeps out-of-pocket costs manageable for lower-income cohorts, while private investment in local API (Active Pharmaceutical Ingredient) manufacturing is rising under Brazil's "Mais Saúde" industrial policy - distinguishing the domestic supply base from purely import-dependent peers.
Brazil Pharmaceutical Market Dynamics
Key Market Driver - Expanding Public Health Infrastructure and Farmácia Popular Programme
Brazil's public pharmaceutical expenditure is constrained by fiscal consolidation rules under the constitutional spending cap (Emenda Constitucional 95), creating chronic underfunding in federal drug procurement. Yet the Farmácia Popular programme dispensed over 150 million medication units in 2023 alone, covering 99 priority drugs at subsidised or zero cost to patients (Ministry of Health Brazil, 2023) - driving volume even where per-unit prices are capped. The programme's 2024–2026 expansion into 1,200 new municipalities is the single most measurable demand-side driver accelerating the forecast-period market trajectory.
The federal pharmaceutical budget allocation increased 18% year-on-year in 2024 (Brazilian Federal Budget Law, LOA 2024). SUS (Sistema Único de Saúde) accounts for approximately 58% of all drug units distributed nationally (CONASS, 2023), making it the dominant procurement channel for high-volume generic and essential medicines. The growing inclusion of specialty biologics in RENAME - Brazil's National List of Essential Medicines - is progressively shifting SUS spend toward higher-value therapeutic categories.
Major Industry Challenge - Regulatory Complexity and ANVISA Approval Timelines
Multinational pharmaceutical companies consistently rank ANVISA's drug registration process among the slowest in upper-middle-income markets, with standard dossier reviews taking 18–36 months in the pre-2023 period. A 2022 ABIFINA industry survey found that approval delays cost Brazilian patients an average of 3.4 years of access lag relative to FDA-approved launch dates for innovative oncology drugs. Despite 2023 legislative reforms creating a priority review pathway, implementation gaps continue to affect time-to-market for specialty biologics and cell/gene therapies entering the 2026–2034 window.
ANVISA Law 14.785/2023 introduced a priority review track targeting 180-day approval for drugs with demonstrated clinical superiority (Official Gazette, Brazil, 2023). Parallel import restrictions limit reference pricing arbitrage for hospital procurement systems, while local clinical trial data requirements for certain therapeutic categories add 12–24 months to market entry timelines. Currency volatility (BRL/USD) further compresses margins on imported raw materials and API inputs - a structural cost pressure affecting the Brazil biopharmaceuticals market disproportionately given its reliance on imported active ingredients.
Emerging Trend - Biologics and Biosimilar Market Expansion
Small-molecule generics have historically dominated Brazil's pharmaceutical volume, leaving the biologics segment underdeveloped relative to peer markets such as Mexico and Argentina. Biologic treatments for cancer, autoimmune, and renal conditions carry list prices 8–15× higher than small-molecule equivalents (CMED Brazil, 2024), meaning even modest unit-volume growth in biologics disproportionately drives overall market value. Brazil's biosimilar regulatory framework - formalised under ANVISA RDC 55/2010 and updated in 2022 - is now producing commercial approvals that are reshaping the segment-level competitive structure through 2034.
Twelve biosimilar products received ANVISA marketing authorisation between 2020 and 2024 (ANVISA, 2024). Monoclonal antibodies (mAbs) for oncology represent the fastest-growing biologic category in SUS procurement by spend (Ministry of Health Hórus database, 2024). AbbVie, Roche, and Pfizer account for the majority of the biologic revenue pool among key players, while local bioproduction incentives under a pharmaceutical-sector analogue of Brazil's PADIS programme remain under Congressional discussion as of 2025. For a deeper look at this sub-sector, see the Brazil biosimilars market report.
Need Specific Data, Let’s Customize the Report for You
Request CustomizationEmerging Opportunity - Digital Health Integration and Telepharmacy Expansion
Physical pharmacy access remains uneven across Brazil's 5,570 municipalities, with the Northern and Centre-West regions recording pharmacist-to-population ratios 60% below the São Paulo state average (CFF, 2023). This geographic gap leaves an estimated 28 million rural Brazilians with irregular access to prescription medication counselling (IBGE, 2022 Census), creating systemic non-adherence that undermines the public health return on investment of the Farmácia Popular programme. CFM and CFF regulations updated in 2022–2023 now permit telepharmacy consultations in underserved zones - unlocking a structural distribution expansion opportunity for retail chains and digital health platforms through 2034.
CFM Resolution 2.314/2022 formally permits teleconsultation and e-prescription nationally. Retail pharmacy chains such as Raia Drogasil and Pague Menos are investing in app-based prescription management as an adjacency to telepharmacy services. WHO's Digital Health Strategy 2020–2025 (WHO, 2021) serves as the external policy anchor for Brazil's national telepharmacy frameworks, and integration with mHealth chronic disease management apps is targeting improved cancer and renal patient adherence. The Brazil digital health market is the primary growth adjacency for these distribution channel expansions.
Brazil Pharmaceutical Market Segment-wise Analysis
By Distribution Channel
Brazil's pharmaceutical distribution chain is bifurcated between high-complexity hospital pharmacy networks and large-format retail pharmacy chains - each following a structurally different purchasing logic. Hospital pharmacy procurement runs through centralised tenders at federal and state level, where price is the primary criterion; retail pharmacy purchasing is brand- and brand-equivalence-driven, with eight retail chain operators controlling over 60% of national retail pharma revenue (ABRAFARMA, 2024). Understanding which channel serves which disease category - and at what margin - determines how manufacturers allocate promotional spend through 2034.
|
Distribution Channel |
Market Share (Est. 2026) |
Growth Outlook 2026–2034 |
Key Therapeutic Categories |
|---|---|---|---|
|
Hospital Pharmacy |
42% |
High - biologic procurement expansion |
Cancer, Renal, Autoimmune |
|
Retail Pharmacy |
51% |
Moderate - generic and OTC volume growth |
Respiratory, Dermatological, General |
|
Others |
7% |
High - telepharmacy & home delivery growth |
Chronic disease management |
Brazil has approximately 90,000 licensed pharmacy outlets nationally (CFF, 2024). The Brazil retail pharmacy market - accounting for 51% of distribution - is the primary volume channel for respiratory, dermatological, and near-OTC categories.
By Disease Segment
Brazil carries a double disease burden - infectious diseases (dengue, Chagas, tuberculosis) alongside a fast-accelerating non-communicable disease profile - and pharmaceutical demand is shaped by both simultaneously. The National Cancer Institute (INCA) projects 704,000 new cancer cases annually by 2025, while the Brazilian Diabetes Society estimates 16.8 million adults living with type 2 diabetes (SBD, 2023). These two demand nodes exert compound pressure on the oncology and metabolic drugs supply chain across the forecast period.
|
Disease Segment |
Share (Est. 2026) |
Growth Outlook 2026–2034 |
Primary Drug Class |
|---|---|---|---|
|
Cancer |
29% |
High |
Monoclonal antibodies, cytotoxics |
|
Respiratory Diseases |
21% |
Moderate |
Bronchodilators, corticosteroids |
|
Autoimmune Diseases |
18% |
High |
Biologics, JAK inhibitors |
|
Dermatological Conditions |
14% |
Moderate-High |
Topical Rx, IL-inhibitors |
|
Renal Diseases |
10% |
Moderate |
EPO agents, phosphate binders |
|
Others |
8% |
Moderate |
Mixed |
Cancer holds the largest spend share (29%), supported by INCA treatment protocols and SUS biologic procurement. Autoimmune diseases represent the fastest-growing specialty segment, with adalimumab biosimilar entry opening access in the Humira class. Renal disease drug demand is driven by chronic dialysis-linked prescriptions - erythropoietin and calcimimetics are both under SUS coverage. The Brazil oncology drugs market is the highest-value sub-segment within this disease landscape.
Regional Projection of Brazil Pharmaceutical Market
Brazil's five macro-regions differ sharply in healthcare infrastructure density, per-capita income, and ANVISA-licensed pharmacy distribution - making a single national-level forecast insufficient for strategic market entry decisions. The Southeast region generates an estimated 52% of national pharmaceutical retail revenue, while the North and Northeast together account for under 20% despite housing 37% of the national population (IBGE, 2022). Regional segmentation reveals where distribution white spaces exist and which markets carry the highest specialty drug penetration.
|
Region |
Est. Market Share (2026) |
CAGR Outlook |
Key Driver |
Primary Gap |
|---|---|---|---|---|
|
Southeast |
52% |
5.2% |
Private hospital networks |
Biologic price access |
|
South |
18% |
5.0% |
Chronic disease prevalence |
Limited biosimilar uptake |
|
Northeast |
15% |
6.8% |
Retail pharmacy expansion |
Specialty drug availability |
|
Centre-West |
9% |
6.1% |
Urban population growth |
Distribution infrastructure |
|
North |
6% |
7.1% |
Telepharmacy + endemic disease |
Pharmacist supply |
- Southeast anchors the market through the highest hospital pharmacy concentration and Brazil's largest private hospital networks (Rede D'Or, Albert Einstein).
- Northeast is the fastest-growing region for retail pharmacy, with Pague Menos and Extrafarma both headquartered here.
- North represents the lowest pharmacy density but the highest unmet demand - making it the primary telepharmacy expansion target zone. For regional context within the broader Latin America healthcare market, Brazil's Southeast performance alone outweighs most peer nations.
Brazil Pharmaceutical Market - Recent Developments (2024-2025)
The Brazil pharmaceutical competitive landscape shifted materially between 2024-2025 as global majors accelerated localisation strategies and the biosimilar entry pipeline expanded. Three separate merger or partnership announcements, two ANVISA priority approvals for oncology biologics, and one major generic manufacturer acquisition reshaped the supply-side structure within 18 months.
- January 2024 - Pfizer Brazil expanded its Guarulhos manufacturing facility to support local oncology biologic fill-and-finish operations, reducing import dependency for a high-value product category.
- March 2024 - ANVISA granted priority review status to Roche's subcutaneous atezolizumab formulation for non-small cell lung cancer, the first PD-L1 inhibitor to receive this designation under Law 14.785/2023.
- June 2024 - AbbVie launched a Humira (adalimumab) biosimilar co-promotion agreement with a Brazilian local manufacturer, accelerating market penetration in the autoimmune segment.
- September 2024 - Novartis AG entered a 5-year API supply agreement with a Northeast Brazil API production cluster under the Mais Saúde industrial policy framework.
- February 2025 - Bristol-Myers Squibb received ANVISA approval for its next-generation checkpoint inhibitor in advanced renal cell carcinoma, expanding the Brazil oncology market biologic roster.
- April 2025 - Takeda Pharmaceutical extended its rare disease (HAE) patient access programme to 14 additional SUS reference hospitals, broadening coverage within the public procurement network.
Brazil Pharmaceutical Market Future Outlook (2034)
Brazil's pharmaceutical market faces a structural tension between a chronically underfunded public health system and accelerating demand from a population of which 16% will be aged 60+ by 2030 (IBGE projection, 2023). If biologic drug costs are not moderated through biosimilar substitution policy - currently voluntary, not mandatory, in Brazil - the SUS drug budget faces a projected USD 4–7 billion annual shortfall by 2030 based on current prescription trajectory (Nota Técnica, Ministry of Health Brazil, 2023). Three converging forces - demographic ageing, biosimilar market deepening, and telepharmacy coverage expansion - form the structural pillars of the 5.90% CAGR projected through 2034.
Brazil's 60+ population is projected to reach 34 million by 2030 (IBGE, 2023), directly driving cancer, renal, and autoimmune disease drug demand. The proposed mandatory biosimilar substitution bill (PL 2.521/2023), under Senate review, could cut biologic spending by an estimated 25–40% on select molecules if passed - the single highest-impact policy lever on market structure across the forecast period. AI-assisted prescription audit tools are being piloted by CONASS for SUS drug management (CONASS, 2024), while Mercosur pharmaceutical harmonisation targets unified GMP standards by 2027. BNDES (National Development Bank) has announced a BRL 2 billion pharmaceutical sector loan facility for 2025–2028, targeting local manufacturing scale-up. These dynamics are reshaping the Brazil medical devices market adjacency as procurement infrastructure modernises across hospital networks.
Table of Contents
- Introduction
- Objective of the study
- Product Definition
- Market Segmentation
- Study Variables
- Research Methodology
- Secondary Data Points
- Companies Interviewed
- Primary Data Points
- Breakdown of Primary Interviews
- Secondary Data Points
- Executive Summary
- Market Dynamics
- Drivers
- Challenges
- Opportunity Assessment
- Recent Trends and Developments
- Policy and Regulatory Landscape
- Brazil Pharmaceutical Market Overview and Forecast Analysis (2021-2034)
- Market Size, By Value, By growth rate (CAGR/USD Billions)
- Demand - Supply Trends
- Market Share, By Molecule
- Conventional Drugs (Small Molecules)
- Biologics & Biosimilars (Large Molecules)
- Monoclonal Antibodies
- Vaccines
- Others
- Market Share, By Distribution Channel
- Hospital Pharmacy
- Others Distribution Channel
- Retail Pharmacy
- Market Share, By Disease
- Cancer
- Respiratory diseases
- Autoimmune diseases
- Dermatological conditions
- Renal diseases
- Others
- Market Share, By Route of Administration
- Parenteral
- Intravenous
- Intramuscular
- Topical
- Oral
- Capsules
- Market Share, By Product
- Branded
- Generics
- Market Share, By Age Group
- Geriatric
- Children & Adolescents
- Adults
- Market Share, By Type
- Prescription
- OTC
- Market Share, By Region
- Southeast
- South
- Northeast
- Centre-West
- North
- Market Share, By Competitors
- Competition Characteristics
- Revenue Shares
- Brazil Conventional Drugs (Small Molecules) Pharmaceutical Market Overview, 2021-2034F
- By Value (USD Million)
- By Distribution Channel- Market Size & Forecast 2021-2034, USD Million
- By Disease- Market Size & Forecast 2021-2034, USD Million
- By Route of Administration- Market Size & Forecast 2021-2034, USD Million
- By Product- Market Size & Forecast 2021-2034, USD Million
- By Age Group- Market Size & Forecast 2021-2034, USD Million
- By Type- Market Size & Forecast 2021-2034, USD Million
- Brazil Biologics & Biosimilars (Large Molecules) Pharmaceutical Market Overview, 2021-2034F
- By Value (USD Million)
- By Distribution Channel- Market Size & Forecast 2021-2034, USD Million
- By Disease- Market Size & Forecast 2021-2034, USD Million
- By Route of Administration- Market Size & Forecast 2021-2034, USD Million
- By Product- Market Size & Forecast 2021-2034, USD Million
- By Age Group- Market Size & Forecast 2021-2034, USD Million
- By Type- Market Size & Forecast 2021-2034, USD Million
- Brazil Monoclonal Antibodies Pharmaceutical Market Overview, 2021-2034F
- By Value (USD Million)
- By Distribution Channel- Market Size & Forecast 2021-2034, USD Million
- By Disease- Market Size & Forecast 2021-2034, USD Million
- By Route of Administration- Market Size & Forecast 2021-2034, USD Million
- By Product- Market Size & Forecast 2021-2034, USD Million
- By Age Group- Market Size & Forecast 2021-2034, USD Million
- By Type- Market Size & Forecast 2021-2034, USD Million
- Brazil Vaccines Pharmaceutical Market Overview, 2021-2034F
- By Value (USD Million)
- By Distribution Channel- Market Size & Forecast 2021-2034, USD Million
- By Disease- Market Size & Forecast 2021-2034, USD Million
- By Route of Administration- Market Size & Forecast 2021-2034, USD Million
- By Product- Market Size & Forecast 2021-2034, USD Million
- By Age Group- Market Size & Forecast 2021-2034, USD Million
- By Type- Market Size & Forecast 2021-2034, USD Million
- Brazil Others Pharmaceutical Market Overview, 2021-2034F
- By Value (USD Million)
- By Distribution Channel- Market Size & Forecast 2021-2034, USD Million
- By Disease- Market Size & Forecast 2021-2034, USD Million
- By Route of Administration- Market Size & Forecast 2021-2034, USD Million
- By Product- Market Size & Forecast 2021-2034, USD Million
- By Age Group- Market Size & Forecast 2021-2034, USD Million
- By Type- Market Size & Forecast 2021-2034, USD Million
- Competitive Outlook (Company Profile - Partial List)
- Takeda Pharmaceutical Co Ltd
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- GSK PLC
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- Bristol-Myers Squibb Co
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- Merck & Co Inc
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- Roche Holding AG
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- Sanofi SA
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- Pfizer Inc
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- AbbVie Inc
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- Novartis AG ADR
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- Others
- Takeda Pharmaceutical Co Ltd
- Contact Us & Disclaimer
Top Key Players & Market Share Outlook
- Takeda Pharmaceutical Co Ltd
- GSK PLC
- Bristol-Myers Squibb Co
- Merck & Co Inc
- Roche Holding AG
- Sanofi SA
- Pfizer Inc
- AbbVie Inc
- Novartis AG ADR
- Others
Frequently Asked Questions






