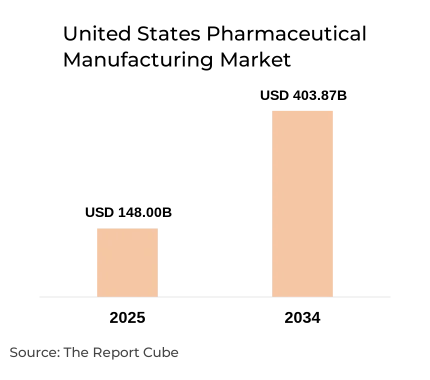
Build Your Custom Market Intelligence Report
Customize Your ReportUnited States Pharmaceutical Manufacturing Market Key Highlights
By Molecule Type:
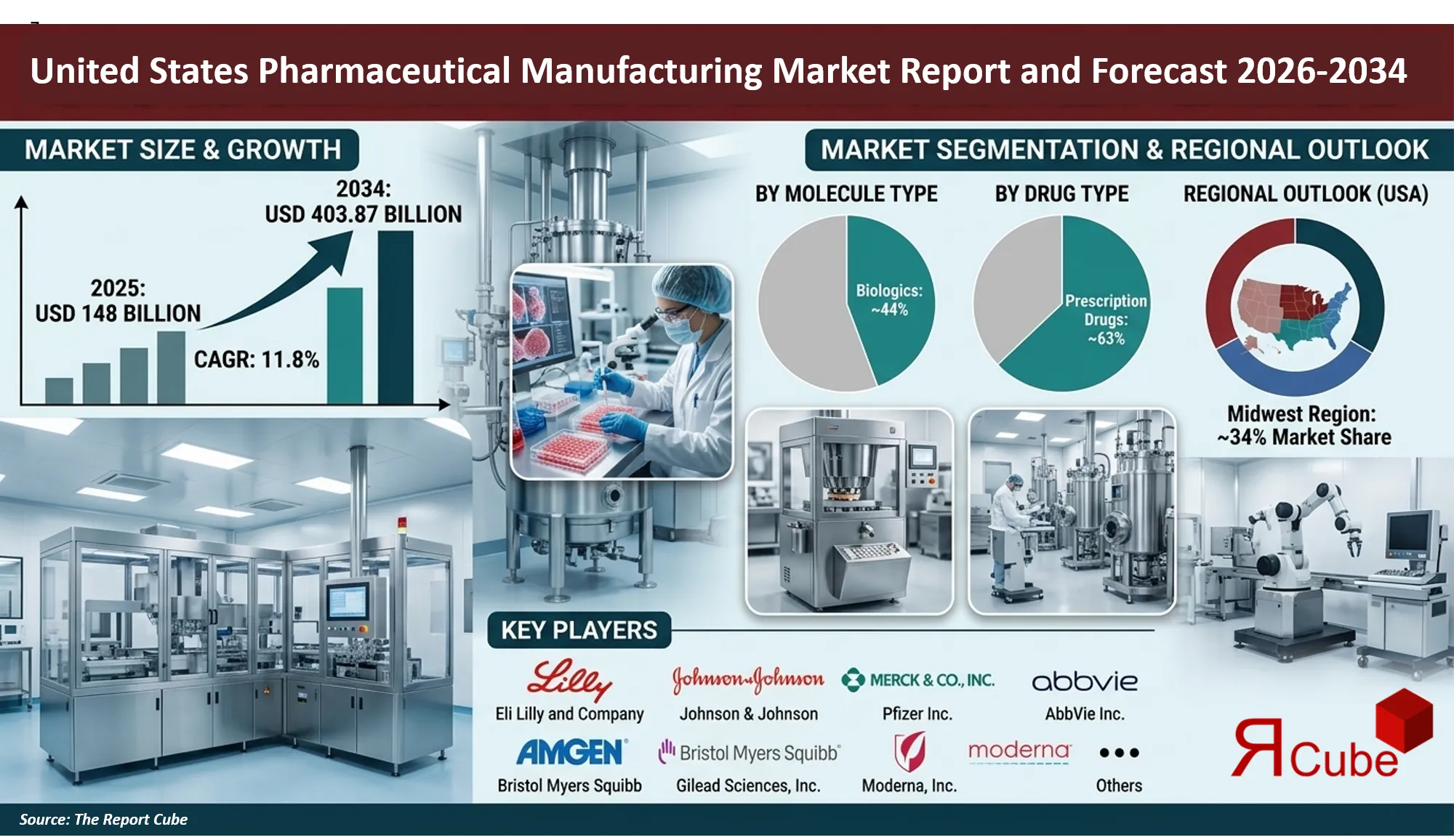
Biologics segment leads the market, with around 44% market share.
By Drug Type:
Prescription drugs segment dominates the market, accounting for nearly 63% of total revenue.
Regional Outlook:
Midwest region dominates the US pharmaceutical manufacturing market, with around 34% market share.
United States Pharmaceutical Manufacturing Market Insights & Analysis
The United States Pharmaceutical Manufacturing Market is anticipated to register a 11.8% during 2026-2034. the market size was valued at USD 148 billion in 2025 and is projected to reach USD 403.87 billion by 2034. The pharmaceutical manufacturing industry in the United States remains one of the most advanced and innovation-driven healthcare production ecosystems globally. With more than 1,800 pharmaceutical manufacturing facilities operating across the country and over 400 FDA-approved drug manufacturing plants located domestically, the U.S. continues to serve as a global hub for both small molecule drug production and advanced biologics manufacturing.
Investment momentum across the sector has accelerated significantly in recent years. According to industry estimates, pharmaceutical companies operating in the United States invested more than USD 80 billion in research, development, and manufacturing infrastructure in 2024 alone. Major manufacturers such as Eli Lilly and Company, Pfizer Inc., and Johnson & Johnson have announced multi-billion-dollar expansion projects to strengthen biologics and injectable drug production capacity. For instance, several large-scale biologics manufacturing plants are being developed across Indiana, North Carolina, and Massachusetts, reflecting the industry’s shift toward complex biologic therapies and personalized medicine.
United States Pharmaceutical Manufacturing Market Dynamics
Key Driver: Expanding Biologics and Specialty Drug Manufacturing Demand
The most influential factor driving growth in the United States pharmaceutical manufacturing market is the increasing demand for biologics and specialty medicines. Biologic therapies now account for nearly 46% of total pharmaceutical spending in the United States, a significant increase from 28% a decade earlier. These therapies include monoclonal antibodies, gene therapies, and recombinant proteins that require highly specialized manufacturing facilities and advanced bioprocessing technologies. As chronic diseases such as cancer, autoimmune disorders, and rare genetic conditions continue to rise, the demand for targeted biologic treatments has surged.
Pharmaceutical companies are responding to this demand by expanding biologics manufacturing infrastructure across the country. Large-scale facilities capable of producing cell-based therapies and mRNA medicines are being developed in biotechnology clusters such as Boston, San Diego, and the Research Triangle in North Carolina. These investments not only increase manufacturing capacity but also improve supply reliability for high-value therapies. With biologic drug pipelines representing more than half of new clinical trials in the U.S., manufacturing demand for these advanced therapies continues to accelerate.
Book your FREE 30-minute expert consultation today
Contact UsIndustry Trends: Adoption of Continuous Manufacturing and Digital Production Technologies
A major technological trend reshaping pharmaceutical manufacturing is the transition from traditional batch production to continuous manufacturing systems. Continuous manufacturing allows pharmaceutical companies to produce medicines in a constant flow process rather than separate batch cycles, improving efficiency and reducing production costs. The U.S. Food and Drug Administration has strongly encouraged this transition by issuing regulatory guidance and providing accelerated approvals for drugs produced using continuous manufacturing technology.
Digitalization is also transforming pharmaceutical production operations. Many manufacturers now integrate artificial intelligence, predictive maintenance systems, and automated quality control platforms into their facilities. These technologies monitor production parameters in real time and detect manufacturing deviations before they affect product quality. As pharmaceutical companies strive to improve compliance and efficiency, digital manufacturing infrastructure is becoming a critical competitive advantage across the industry.
Major Challenge: Complex Regulatory Compliance and High Manufacturing Costs
One of the most significant challenges facing the United States pharmaceutical manufacturing market is the high cost associated with regulatory compliance and facility development. Pharmaceutical manufacturing facilities must adhere to stringent FDA Good Manufacturing Practice (GMP) standards, which require extensive validation procedures, documentation, and quality assurance systems. Establishing a new biologics manufacturing plant can require investment exceeding USD 1 billion and may take five to seven years to become fully operational.
Regulatory oversight is essential for ensuring drug safety and quality, yet the complexity of compliance requirements increases operational costs for pharmaceutical manufacturers. Companies must continuously upgrade equipment, implement advanced monitoring systems, and maintain rigorous documentation processes. Smaller biotechnology firms often rely on contract manufacturing organizations because building proprietary manufacturing facilities can be financially prohibitive. Managing regulatory expectations while maintaining operational efficiency remains a critical challenge for industry participants.
Opportunity: Growth of Contract Development and Manufacturing Organizations
The rapid growth of contract development and manufacturing organizations represents one of the most promising opportunities in the pharmaceutical manufacturing market. CDMOs provide specialized manufacturing services including biologics production, sterile injectable formulation, and clinical trial material manufacturing. As drug pipelines become more complex, pharmaceutical companies increasingly partner with contract manufacturers to access advanced production capabilities.
The United States currently hosts several of the world’s leading pharmaceutical contract manufacturers, many of which are expanding capacity to meet growing demand. Biopharmaceutical startups and mid-size pharmaceutical companies rely heavily on CDMO partnerships to accelerate product development and commercialization. As personalized medicine and gene therapies continue to gain traction, the role of contract manufacturers in supporting pharmaceutical innovation will expand significantly across the industry.
United States Pharmaceutical Manufacturing Market Segment-wise Analysis
By Molecule Type
- Small Molecule Drugs
- Biologics
- Biosimilars
The biologics segment leads the United States pharmaceutical manufacturing market with nearly 44% market share, reflecting the industry's transition toward advanced therapeutic solutions. Biologics manufacturing requires highly specialized production environments involving cell culture systems, fermentation processes, and purification technologies. Unlike small molecule drugs that are chemically synthesized, biologics are derived from living organisms, making their manufacturing processes significantly more complex.
The rapid growth of monoclonal antibodies and gene therapies has driven massive investment in biologics manufacturing infrastructure across the United States. Pharmaceutical companies such as Amgen Inc., Regeneron Pharmaceuticals, and Moderna have expanded biologics production facilities to meet rising demand for targeted treatments. Additionally, biosimilars manufacturing is gaining traction as patents for several blockbuster biologic drugs expire, creating new opportunities for cost-effective therapeutic alternatives. As biologics pipelines continue to grow, manufacturing capacity expansion remains a priority for pharmaceutical companies.
By Drug Type
- Prescription Drugs
- Over-the-Counter Drugs
- Generic Drugs
- Branded Drugs
The prescription drugs segment dominates the pharmaceutical manufacturing market, accounting for approximately 63% of total industry revenue. Prescription medicines include specialty drugs, biologics, oncology treatments, and advanced therapies that require physician supervision. The rising prevalence of chronic diseases such as cancer, diabetes, and cardiovascular disorders has significantly increased demand for prescription medications across the United States.
Additionally, pharmaceutical innovation continues to drive the expansion of prescription drug manufacturing. Many new therapies approved by regulatory authorities involve complex biologic molecules, targeted therapies, and personalized medicines designed to treat specific genetic conditions. These treatments require advanced manufacturing technologies and strict quality control systems. As pharmaceutical research continues to produce innovative therapies, the demand for prescription drug manufacturing capacity is expected to grow steadily throughout the forecast period.
Regional Projection of United States Pharmaceutical Manufacturing Industry
- West
- East
- South
- Midwest
- Others
The Midwest region leads the United States pharmaceutical manufacturing market with nearly 34% share, supported by a strong industrial base and a highly developed pharmaceutical production ecosystem. States such as Indiana, Illinois, and Ohio host several large pharmaceutical manufacturing facilities operated by major industry players including Eli Lilly and Company and Bristol Myers Squibb. Indiana alone accounts for a substantial portion of the country’s pharmaceutical exports due to its extensive network of drug production plants.
The region benefits from strong logistics infrastructure, skilled labor availability, and proximity to major healthcare research institutions. Pharmaceutical companies are increasingly expanding operations in the Midwest because of lower operational costs compared with coastal biotechnology hubs. Additionally, state governments provide tax incentives and manufacturing grants to attract pharmaceutical investments. As large companies continue expanding manufacturing facilities in this region, the Midwest is expected to maintain its leadership position within the national pharmaceutical production landscape.
Government Initiatives & Policies
- U.S. Biosecure Act (2025): Policy aimed at strengthening domestic biopharmaceutical supply chains and reducing reliance on foreign contract manufacturing while supporting domestic pharmaceutical production expansion.
- HHS Pharmaceutical Supply Chain Resilience Initiative (2026): Program designed to expand domestic pharmaceutical manufacturing capacity, strengthen API production in the U.S., and ensure national medicine security.
United States Pharmaceutical Manufacturing Market Recent Developments
- 2025: Eli Lilly and Company announced a multibillion-dollar expansion of its Indiana manufacturing facilities to support large-scale production of diabetes and obesity medications, significantly increasing domestic biologics and injectable drug manufacturing capacity.
- 2025: Pfizer Inc. expanded its sterile injectable manufacturing facility in North Carolina to strengthen supply chain resilience and increase production of oncology and specialty medicines.
- 2025: Johnson & Johnson invested in advanced continuous manufacturing technologies at its U.S. pharmaceutical plants to improve efficiency and accelerate commercial drug production.
- 2025: Moderna, Inc. expanded its mRNA manufacturing capabilities in Massachusetts to support vaccine production and future therapeutic development.
- 2025: Regeneron Pharmaceuticals expanded biologics manufacturing capacity in New York to support growing demand for monoclonal antibody therapies.
Need insights for a specific region within this market?
Request Regional DataWhy Choose This Report?
- Provides a comprehensive overview of the overall market analysis, encompassing key trends, consumer behavior analysis, and risk assessment to support strategic decision-making.
- Provides accurate, up-to-date insights into market size, segmentation, and emerging opportunities, helping to minimize risk & capitalizing on growth.
- Gives deep understanding of target audience preferences, investment habits, and communication channels for enhanced product development & marketing effectiveness.
- Delivers competitive analysis & benchmarking, uncovering the strengths & weaknesses of market competitors to guide strategies.
- Consolidate comprehensive market intelligence, reducing reasoning & streamlining research efforts.
- Facilitates customized market segmentation & risk mitigation strategies, fine-tuned to the business objectives.
- Aids in identifying both market challenges & untapped opportunities within the industry to drive long-term business growth.
- Provides valuable information based on actual customer data & search trends.
Table of Contents
- Introduction
- Objective of the Study
- Product and Category Definition
- Market Segmentation
- Study Variables
- Research Methodology
- Secondary Data Points
- Breakdown of Secondary Sources
- Primary Data Points
- Breakdown of Primary Interviews
- Secondary Data Points
- Executive Summary
- Market Dynamics
- Drivers
- Challenges
- Opportunity Assessment
- Recent Trends and Developments
- Regulatory and Policy Landscape
- United States Pharmaceutical Manufacturing Market Overview (2021-2034)
- Market Size, By Value (USD Billion)
- Market Share, By Molecule Type
- Small Molecule Drugs
- Biologics
- Biosimilars
- Market Share, By Drug Type
- Prescription Drugs
- Over-the-Counter Drugs
- Generic Drugs
- Branded Drugs
- Market Share, By Route of Administration
- Oral Drugs
- Injectable Drugs
- Inhalation Drugs
- Topical Drugs
- Other Routes
- Market Share, By Formulation
- Tablets
- Capsules
- Injectables
- Suspensions
- Solutions
- Others
- Market Share, By Manufacturer Type
- Innovator Manufacturers
- Generic Manufacturers
- Contract Development
- Manufacturing Organizations
- Market Share, By Scale of Manufacturing
- Captive Manufacturing
- Contract Manufacturing
- Market Share, By Region
- West
- East
- South
- Midwest
- Others
- Market Share, By Company
- Revenue Shares & Analysis
- Competitive Landscape
- West United States Pharmaceutical Manufacturing Market
- Market Size, By Value (USD Billion/Million)
- Market Share, By Molecule Type
- Market Share, By Drug Type
- Market Share, By Route of Administration
- Market Share, By Formulation
- Market Share, By Manufacturer Type
- Market Share, By Scale of Manufacturing
- East United States Pharmaceutical Manufacturing Market
- Market Size, By Value (USD Billion/Million)
- Market Share, By Molecule Type
- Market Share, By Drug Type
- Market Share, By Route of Administration
- Market Share, By Formulation
- Market Share, By Manufacturer Type
- Market Share, By Scale of Manufacturing
- South United States Pharmaceutical Manufacturing Market
- Market Size, By Value (USD Billion/Million)
- Market Share, By Molecule Type
- Market Share, By Drug Type
- Market Share, By Route of Administration
- Market Share, By Formulation
- Market Share, By Manufacturer Type
- Market Share, By Scale of Manufacturing
- Midwest United States Pharmaceutical Manufacturing Market
- Market Size, By Value (USD Billion/Million)
- Market Share, By Molecule Type
- Market Share, By Drug Type
- Market Share, By Route of Administration
- Market Share, By Formulation
- Market Share, By Manufacturer Type
- Market Share, By Scale of Manufacturing
- Competitive Outlook and Company Profiles
- Eli Lilly and Company
- Company Overview
- Product Portfolio
- Strategic Alliances/Partnerships
- Recent Developments
- Johnson & Johnson
- Company Overview
- Product Portfolio
- Strategic Alliances/Partnerships
- Recent Developments
- Merck & Co., Inc.
- Company Overview
- Product Portfolio
- Strategic Alliances/Partnerships
- Recent Developments
- Pfizer Inc.
- Company Overview
- Product Portfolio
- Strategic Alliances/Partnerships
- Recent Developments
- AbbVie Inc.
- Company Overview
- Product Portfolio
- Strategic Alliances/Partnerships
- Recent Developments
- Amgen Inc.
- Company Overview
- Product Portfolio
- Strategic Alliances/Partnerships
- Recent Developments
- Bristol Myers Squibb
- Company Overview
- Product Portfolio
- Strategic Alliances/Partnerships
- Recent Developments
- Gilead Sciences, Inc.
- Company Overview
- Product Portfolio
- Strategic Alliances/Partnerships
- Recent Developments
- Moderna, Inc.
- Company Overview
- Product Portfolio
- Strategic Alliances/Partnerships
- Recent Developments
- Regeneron Pharmaceuticals
- Company Overview
- Product Portfolio
- Strategic Alliances/Partnerships
- Recent Developments
- Biogen Inc.
- Company Overview
- Product Portfolio
- Strategic Alliances/Partnerships
- Recent Developments
- Vertex Pharmaceuticals
- Company Overview
- Product Portfolio
- Strategic Alliances/Partnerships
- Recent Developments
- Viatris Inc.
- Company Overview
- Product Portfolio
- Strategic Alliances/Partnerships
- Recent Developments
- Incyte Corporation
- Company Overview
- Product Portfolio
- Strategic Alliances/Partnerships
- Recent Developments
- Zoetis Inc.
- Company Overview
- Product Portfolio
- Strategic Alliances/Partnerships
- Recent Developments
- Perrigo Company plc
- Company Overview
- Product Portfolio
- Strategic Alliances/Partnerships
- Recent Developments
- Others
- Eli Lilly and Company
- Contact Us and Disclaimer
Top Key Players & Market Share Outlook
- Eli Lilly and Company
- Johnson & Johnson
- Merck & Co., Inc.
- Pfizer Inc.
- AbbVie Inc.
- Amgen Inc.
- Bristol Myers Squibb
- Gilead Sciences, Inc.
- Moderna, Inc.
- Regeneron Pharmaceuticals
- Biogen Inc.
- Vertex Pharmaceuticals
- Viatris Inc.
- Incyte Corporation
- Zoetis Inc.
- Perrigo Company plc
Frequently Asked Questions






