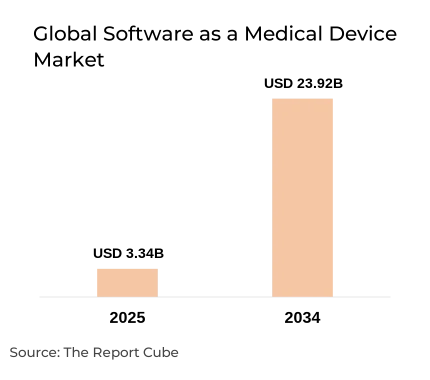
Build Your Custom Market Intelligence Report
Customize Your ReportGlobal Software as a Medical Device Market Statistics and Insights
Market Insight
- The Global Software as a Medical Device Market was valued at USD 3.34 billion in 2025.
- The market is projected to reach USD 23.92 billion by the end of 2034.
- The market is anticipated to grow at a CAGR of 24.45% during 2026–2034.
By Device Type
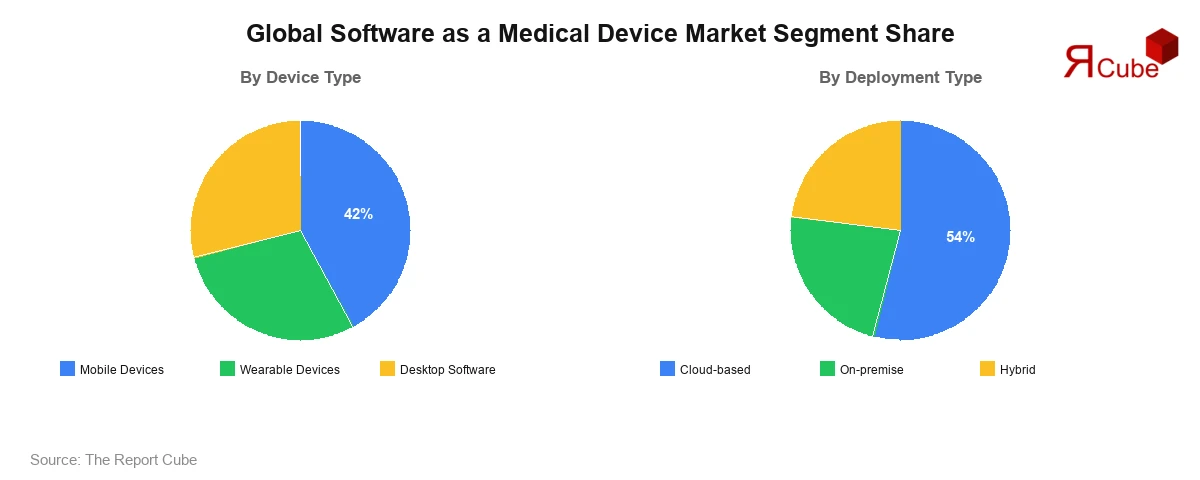
- Mobile devices hold the dominant position with approximately 42% market share in 2025.
Competitive Landscape
- More than 10 companies are actively engaged in producing Software as a Medical Device solutions globally.
- Top 5 companies acquired around 58% of the market share.
- Medtronic, Philips, Siemens Healthineers, GE Healthcare, IBM Watson Health, and others.
By Deployment Type
- Cloud-based deployment leads the market with around 54% share in 2025.
Global Software as a Medical Device Market Insights & Analysis
The Global Software as a Medical Device Market is anticipated to register a 24.45% CAGR during 2026–2034. The market size was valued at USD 3.34 billion in 2025 and is projected to reach USD 23.92 billion by 2034. The numbers look large, but the underlying story is fairly straightforward. Healthcare is digitizing fast, regulators are catching up faster than most expected, and software-based diagnostics are now taken seriously by procurement teams that once dismissed them as research tools.
The U.S. FDA had cleared over 500 AI/ML-based SaMD products by late 2024 through its Digital Health Center of Excellence, a real volume of cleared products, not a vague pipeline. The EU's MDR framework brought clinical decision-support software under CE-marking requirements, which raised standards but also made the category easier to finance. Venture funding into digital health platforms with SaMD integration crossed USD 6.1 billion globally in 2024. Asia-Pacific is now the fastest-growing region for new product registrations, not just the largest manufacturing base.
Global Software as a Medical Device Market Dynamics
Key Market Driver Faster FDA Clearances Are Moving Markets
The FDA's Pre-Submission Program and De Novo pathway cut average SaMD review timelines from 31 months to under 12 months for qualifying digital diagnostics. That is not a marginal improvement, it changes capital allocation. Health systems now include dedicated SaMD procurement lines in annual budgets, which was unusual before 2020. Radiology AI tools went from small pilot programs to cleared deployments across 2,400+ U.S. hospitals between 2021 and 2024. When a hospital CFO can see a predictable regulatory timeline, the software stops looking like a research experiment and starts looking like a capital investment. That shift in perception is what's actually driving commercial adoption at scale right now.
Major Industry Challenge Real-World Performance Gaps Erode Clinical Trust
A 2024 study in The Lancet Digital Health found that 64% of CE-marked or FDA-cleared SaMD products lacked post-market performance data beyond their initial validation cohort. The problem has a structural cause: most algorithms are validated on data from large academic medical centers, then deployed in community hospitals with different patient populations, EHR architectures, and clinical workflows. The gap between controlled trial performance and real-world results is often invisible until a clinician encounters a misclassification that shouldn't have happened. One high-profile failure in a department is enough to freeze adoption for months. Rebuilding that trust takes time that most commercial teams don't budget for.
Emerging Trend Shaping Outlook Federated Learning Removes the Data-Sharing Barrier
Federated learning lets SaMD models improve continuously without pooling raw patient data across institutions. Each hospital trains the model locally and shares only encrypted gradient updates, no patient records leave the source system. A diagnostic algorithm for detecting early-stage diabetic retinopathy can get better across 300 clinics globally without triggering a single data transfer agreement. That matters in markets where privacy regulations (Germany's DSGVO, India's DPDPA) make cross-border data sharing legally complex. Siemens Healthineers and several academic consortia moved federated pipelines into clinical production in 2024. The approach is not experimental anymore; it's a compliance workaround that actually works.

Need Specific Data, Let’s Customize the Report for You
Request CustomizationGlobal Software as a Medical Device Market Segment-wise Analysis
The market splits across two dimensions that affect pricing, regulation, and adoption differently: the device type the software runs on, and the infrastructure that hosts it. Both choices carry downstream consequences for vendors and buyers alike.
By Device Type
- Mobile Devices
- Wearable Devices
- Desktop Software
Mobile devices hold roughly 42% of global SaMD revenue in 2025. The reasons are practical. Smartphones are already in clinical settings and patient homes. The FDA published dedicated guidance for mobile medical applications years ago. App store distribution lowers go-to-market costs significantly compared to enterprise software sales cycles. Cardiology apps that detect atrial fibrillation via consumer smartwatches are now FDA-cleared and covered as benefits by some U.S. health insurers.
Wearable devices contribute approximately 28% of segment revenue, growing fast on the back of continuous glucose monitors and ECG-capable smartwatches that produce clinical-grade data streams, all of which require SaMD layers to turn raw signals into actionable outputs. Desktop software holds around 30% share and is not going anywhere in radiology AI, computational pathology, and hospital-based clinical decision support, where processing demands and EHR integration requirements are non-negotiable.
By Deployment Type
- Cloud-based
- On-premise
- Hybrid
Cloud-based deployment holds approximately 54% market share in 2025. Health organizations that prefer operating expenditure models, faster update cycles, and scalability without hardware commitments are driving this. Multi-tenant SaaS diagnostic tools are now standard among mid-size health systems in North America and Western Europe. On-premise holds about 22% share, mainly in markets with strict data localization rules. Germany's DSGVO enforcement and India's Digital Personal Data Protection Act are keeping patient-level inference workloads on local servers for a meaningful subset of providers. Hybrid deployment at roughly 24%, and growing, reflects a practical compromise: model inference runs on-premise to satisfy compliance, while training, updates, and analytics pipelines run in the cloud to keep IT overhead manageable. It's an architecture that tends to satisfy both the legal and technology teams, which is rare.
Regional Projection of Global Software as a Medical Device Market
- North America
- Europe
- Asia-Pacific
- Latin America
- Middle East & Africa
North America accounts for approximately 41% of global SaMD revenue in 2025. The U.S. drives the bulk of that figure. Advanced health IT infrastructure, the highest per-capita health expenditure in the world, employer-sponsored insurance programs that now cover AI-assisted diagnostics, and a venture ecosystem that has been funding digital health at scale since 2018, all of this compounds. Europe holds the second-largest share, with Germany, the UK, and France leading regional adoption. Asia-Pacific is the fastest-growing region, with China and Japan investing in domestic SaMD development for regulatory and strategic reasons. Latin America and the Middle East & Africa are earlier-stage markets but are seeing mobile-first SaMD gain traction, particularly in settings where legacy clinical infrastructure is thin.
Global Software as a Medical Device Market: Recent Development (2025)
- Medtronic reported a 17% operating margin on its SaMD-integrated diabetes management portfolio in Q1 2025.
- Philips deployed AI-powered diagnostic software across 60 European hospital sites through a new network partnership.
- Siemens Healthineers received FDA clearance for its next-generation radiology triage software in February 2025.
- GE Healthcare posted USD 340 million in SaMD-related revenue, a 22% increase year on year.
Global Software as a Medical Device Market Future Outlook (2034)
The Global Software as a Medical Device Market is on a credible path to USD 23.92 billion by 2034 at a 24.45% CAGR. The growth is not just volume, the structure of the market is changing. HL7 FHIR interoperability standards will make SaMD integration with EHR systems routine rather than a multi-year IT project. AI diagnostic tools are moving from single-condition focus to multi-modal, whole-patient risk profiles. Reimbursement reform in the U.S. and EU, CPT codes and DRG adjustments that recognize AI-assisted clinical pathways, will pull SaMD deeper into standard care. And in Southeast Asia and Sub-Saharan Africa, mobile-first SaMD has a real shot at leapfrogging legacy infrastructure where fixed clinical systems never fully took hold.
Why Choose This Report?
- Provides a comprehensive overview of the overall market analysis, encompassing key trends, consumer behavior analysis, and risk assessment to support strategic decision-making.
- Provides accurate, up-to-date insights into market size, segmentation, and emerging opportunities, helping to minimize risk & capitalizing on growth.
- Gives deep understanding of target audience preferences, investment habits, and communication channels for enhanced product development & marketing effectiveness.
- Delivers competitive analysis & benchmarking, uncovering the strengths & weaknesses of market competitors to guide strategies.
- Consolidate comprehensive market intelligence, reducing reasoning & streamlining research efforts.
- Facilitates customized market segmentation & risk mitigation strategies, fine-tuned to the business objectives.
- Aids in identifying both market challenges & untapped opportunities within the industry to drive long-term business growth.
- Provides valuable information based on actual customer data & search trends.
Table of Contents
- Introduction
- Objective of the study
- Product Definition
- Market Segmentation
- Study Variables
- Research Methodology
- Secondary Data Points
- Companies Interviewed
- Primary Data Points
- Breakdown of Primary Interviews
- Secondary Data Points
- Executive Summary
- Market Dynamics
- Drivers
- Challenges
- Opportunity Assessment
- Recent Trends and Developments
- Policy and Regulatory Landscape
- Global Software as a Medical Device Market Overview (2021-2034)
- Market Size, By Value (in USD Billions)
- Market Share, By End Users
- Hospitals
- Clinics
- Home Care
- Pharmaceuticals
- Market Share, By Application
- Diagnostic Software
- Therapeutic Software
- Monitoring Software
- Consultation Software
- Market Share, By Device Type
- Mobile Devices
- Wearable Devices
- Desktop Software
- Market Share, By eployment Type
- Cloud-based
- On-premise
- Hybrid
- Market Share, By Region
- North America
- Europe
- Asia-Pacific
- Latin America
- Middle East & Africa
- Market Share, By Company
- Revenue Shares
- Competition Characteristics
- North America Software as a Medical Device Market Overview (2021-2034)
- Market Size, By Value (in USD Billions)
- Market Share, By End Users
- Market Share, By Application
- Market Share, By Device Type
- Market Share, By eployment Type
- By Country
- The US
- Canada
- Mexico
- The US Software as a Medical Device Market Overview (2021-2034)
- Market Share, By End Users
- Market Share, By Application
- Market Share, By Device Type
- Market Share, By eployment Type
- Canada Software as a Medical Device Market Overview (2021-2034)
- Market Share, By End Users
- Market Share, By Application
- Market Share, By Device Type
- Market Share, By eployment Type
- Mexico Software as a Medical Device Market Overview (2021-2034)
- Market Share, By End Users
- Market Share, By Application
- Market Share, By Device Type
- Market Share, By eployment Type
- South America Software as a Medical Device Market Overview (2021-2034)
- Market Size, By Value (in USD Billions)
- Market Share, By End Users
- Market Share, By Application
- Market Share, By Device Type
- Market Share, By eployment Type
- By Country
- Brazil
- Argentina
- Rest of South America
- Brazil Software as a Medical Device Market Overview (2021-2034)
- Market Share, By End Users
- Market Share, By Application
- Market Share, By Device Type
- Market Share, By eployment Type
- Argentina Software as a Medical Device Market Overview (2021-2034)
- Market Share, By End Users
- Market Share, By Application
- Market Share, By Device Type
- Market Share, By eployment Type
- Europe Software as a Medical Device Market Overview (2021-2034)
- Market Size, By Value (in USD Billions)
- By End Users
- By Application
- By Device Type
- By eployment Type
- By Country
- Germany
- The UK
- France
- Spain
- Italy
- Rest of Europe
- Germany Software as a Medical Device Market Overview (2021-2034)
- Market Share, By End Users
- Market Share, By Application
- Market Share, By Device Type
- Market Share, By eployment Type
- France Software as a Medical Device Market Overview (2021-2034)
- Market Share, By End Users
- Market Share, By Application
- Market Share, By Device Type
- Market Share, By eployment Type
- The UK Software as a Medical Device Market Overview (2021-2034)
- Market Share, By End Users
- Market Share, By Application
- Market Share, By Device Type
- Market Share, By eployment Type
- Spain Software as a Medical Device Market Overview (2021-2034)
- Market Share, By End Users
- Market Share, By Application
- Market Share, By Device Type
- Market Share, By eployment Type
- Italy Software as a Medical Device Market Overview (2021-2034)
- Market Share, By End Users
- Market Share, By Application
- Market Share, By Device Type
- Market Share, By eployment Type
- The Middle East & Africa Software as a Medical Device Market Overview (2021-2034)
- Market Size, By Value (in USD Billions)
- By End Users
- By Application
- By Device Type
- By eployment Type
- By Country
- The UAE
- Saudi Arabia
- South Africa
- Rest of the Middle East & Africa
- The UAE Software as a Medical Device Market Overview (2021-2034)
- Market Share, By End Users
- Market Share, By Application
- Market Share, By Device Type
- Market Share, By eployment Type
- Saudi Arabia Software as a Medical Device Market Overview (2021-2034)
- Market Share, By End Users
- Market Share, By Application
- Market Share, By Device Type
- Market Share, By eployment Type
- South Africa Software as a Medical Device Market Overview (2021-2034)
- Market Share, By End Users
- Market Share, By Application
- Market Share, By Device Type
- Market Share, By eployment Type
- Asia-Pacific Software as a Medical Device Market Overview (2021-2034)
- Market Size, By Value (in USD Billions)
- By End Users
- By Application
- By Device Type
- By eployment Type
- By Country
- China
- India
- Japan
- South Korea
- Australia
- Rest of Asia-Pacific
- China Software as a Medical Device Market Overview (2021-2034)
- Market Share, By End Users
- Market Share, By Application
- Market Share, By Device Type
- Market Share, By eployment Type
- India Software as a Medical Device Market Overview (2021-2034)
- Market Share, By End Users
- Market Share, By Application
- Market Share, By Device Type
- Market Share, By eployment Type
- Japan Software as a Medical Device Market Overview (2021-2034)
- Market Share, By End Users
- Market Share, By Application
- Market Share, By Device Type
- Market Share, By eployment Type
- South Korea Software as a Medical Device Market Overview (2021-2034)
- Market Share, By End Users
- Market Share, By Application
- Market Share, By Device Type
- Market Share, By eployment Type
- Austraila Software as a Medical Device Market Overview (2021-2034)
- Market Share, By End Users
- Market Share, By Application
- Market Share, By Device Type
- Market Share, By eployment Type
- Competitive Outlook (Company Profile - Partial List)
- Medtronic
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- Philips
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- Siemens Healthineers
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- GE Healthcare
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- IBM Watson Health
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- Boston Scientific
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- Cerner Corporation
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- Roche Diagnostics
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- Johnson & Johnson
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- Others
- Medtronic
- Disclaimer
Top Key Players & Market Share Outlook
- Medtronic
- Philips
- Siemens Healthineers
- GE Healthcare
- IBM Watson Health
- Boston Scientific
- Cerner Corporation
- Roche Diagnostics
- Johnson & Johnson
- Others
Frequently Asked Questions






