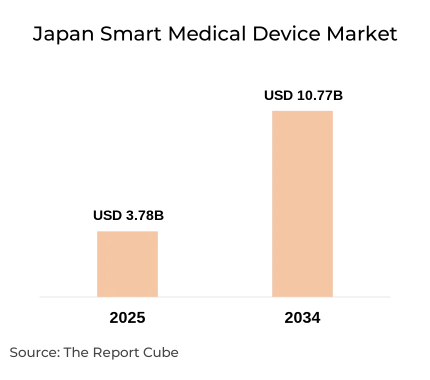
Build Your Custom Market Intelligence Report
Customize Your ReportJapan Smart Medical Device Market Statistics and Insights
Market Size Statistics
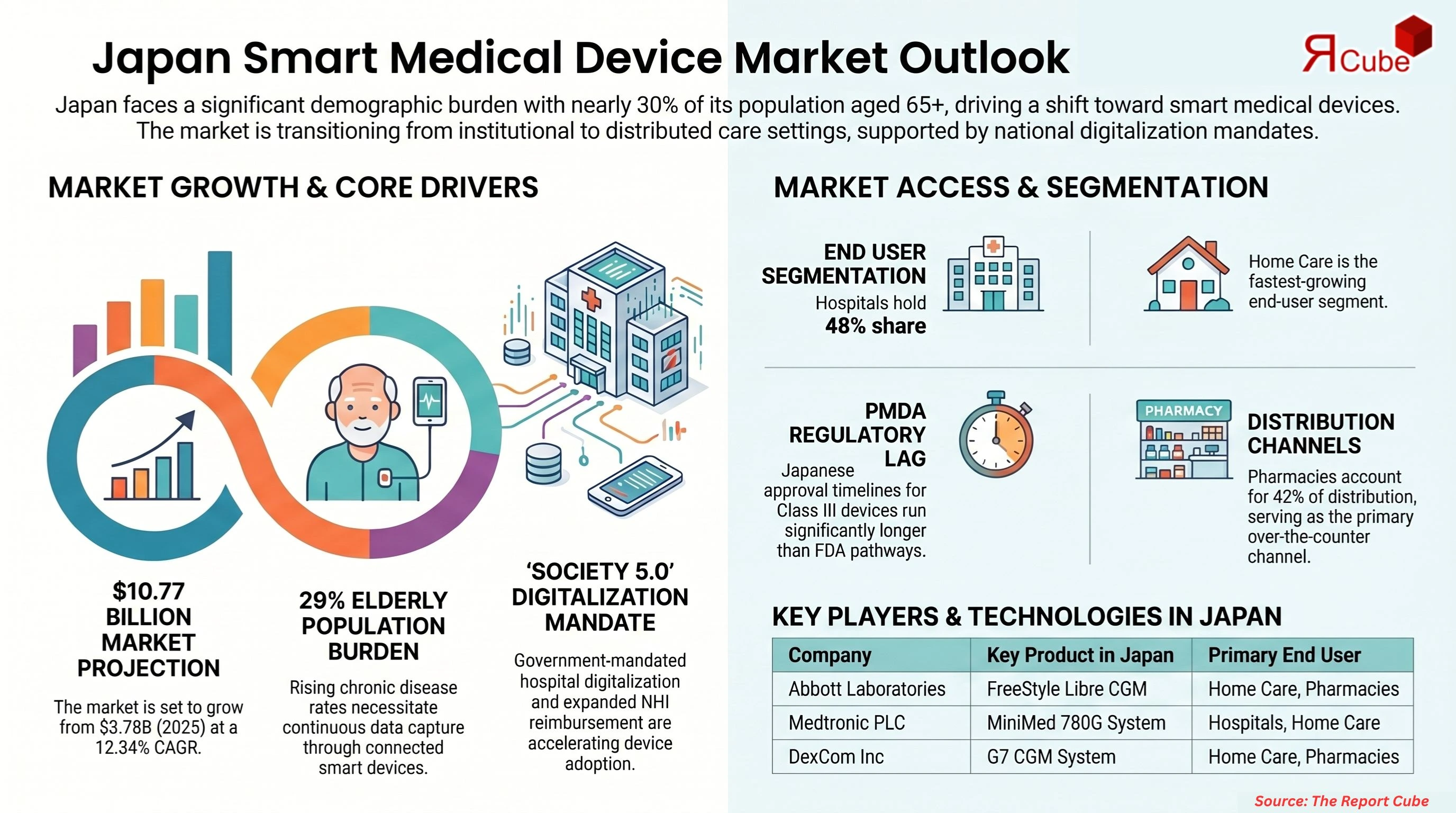
- Japan Smart Medical Devices Market reached USD 3.78 billion 2025
- Market projected to reach USD 10.77 billion by 2034.
- Market expected to grow at CAGR 12.34% during forecast period
By End User
- Hospitals segment holds dominant 48% share adoption
Competitive Landscape
- More than 10 companies are actively engaged in producing Japan Smart Medical Devices Market
- Top 5 companies acquired around 57% of the market share.
- Abbott Laboratories, Medtronic PLC, Roche Holding AG, DexCom Inc, Masimo Corp, and others
By Distribution Channel
- Pharmacies segment accounts for 42% driven accessibility
Japan Smart Medical Device Market Insights and Analysis
Japan carries one of the world's most acute demographic healthcare burdens. With more than 29% of its population aged 65 and above as of 2024 (Japan Statistics Bureau), the country faces chronic capacity strain across hospitals, clinics, and home care networks. Without device-enabled remote monitoring and AI-assisted diagnostics, hospitalisation rates for manageable chronic conditions will continue to push public health spending beyond sustainable thresholds, projected to exceed 50% of Japan's national budget by the early 2030s.
The Japan smart medical devices market reached USD 3.78 billion in 2025 and is expanding at a 12.34% CAGR to reach USD 10.77 billion by 2034. Two structural forces drive this trajectory: government-mandated hospital digitalisation under the Society 5.0 national strategy, and rising NHI reimbursement coverage for smart diagnostic and monitoring devices. Japan medical devices market as a whole is scaling rapidly, but the smart device sub-segment is accelerating fastest as chronic disease management shifts from institutional to distributed care settings.
Japan Smart Medical Device Market Dynamics
This section examines the primary driver, key challenge, and emerging trend shaping the Japan smart medical devices market through 2034.
Key Market Driver, Ageing Population and Chronic Disease Burden Accelerating Device Adoption
Japan's 29%-plus elderly population ratio produces disproportionately high prevalence rates for cardiovascular disease, diabetes, and respiratory conditions requiring continuous monitoring (Ministry of Health, Labour and Welfare, 2024). These are not episodic conditions, they require persistent data capture, which only connected smart devices deliver reliably at scale.
Device penetration in home care settings currently falls below what the burden of disease warrants. Hospital procurement programmes are expanding capacity, but the addressable gap in community and home-based monitoring remains significant, particularly for diabetes and cardiac populations. This gap is precisely where the highest-growth device categories are concentrated.
Hospital procurement mandates under Society 5.0 and NHI reimbursement expansion are closing this gap at a measured pace, sustaining the 12.34% CAGR through 2034. Continuous glucose monitors, including DexCom G7 and Abbott FreeStyle Libre, and remote cardiac monitors from Masimo represent the highest-growth device categories tied directly to this demographic driver.
Major Industry Challenge, Stringent PMDA Approval Timelines and Reimbursement Uncertainty
Japan's Pharmaceuticals and Medical Devices Agency (PMDA) approval process for Class III smart medical devices runs materially longer than FDA 510(k) pathways, discouraging simultaneous Japan launches for international manufacturers. The gap in review timelines creates a competitive disadvantage window where products available in the US and EU remain inaccessible to Japanese patients.
Reimbursement coverage under Japan's National Health Insurance system for newer smart device categories is reviewed only biennially, creating a revenue uncertainty window of up to two years post-approval. For mid-sized manufacturers, this timeline risk directly affects capital allocation decisions and Japan market entry sequencing.
MHLW's 2023 regulatory reform roadmap targets a 30% reduction in average PMDA review times by 2026. If achieved, this reform materially changes the Japan entry calculus, particularly for AI-enabled and software-based smart medical devices entering through the Software as a Medical Device (SaMD) pathway.
Emerging Trend Shaping Outlook, AI-Integrated Diagnostics and Wearable Biosensor Convergence
Japan's hospital system faces acute physician shortages outside major urban centres, reducing diagnostic throughput and increasing care delays for rural populations. This structural gap makes AI-augmented triage and remote monitoring tools a clinical necessity, not a premium offering.
AI-integrated smart devices, including ECG-analysing wearables and AI-assisted pulse oximeters from Masimo and SmartCardia, are entering Japan's hospital and home care channels through PMDA-approved SaMD pathways, supported by the Japan wearable medical devices market regulatory guidelines issued in 2023. These are commercial deployments, not future projections.
The home care segment is absorbing a growing share of AI-enabled device adoption. As PMDA's SaMD framework matures, the product pipeline entering Japan will accelerate further, reinforcing the convergence between diagnostic software and biosensor hardware as a defining feature of the forecast period.
Need Specific Data, Let’s Customize the Report for You
Request CustomizationJapan Smart Medical Device Market Segment-wise Analysis
The Japan smart medical devices market is segmented by end user and by distribution channel.
By End User
Hospitals hold the dominant share of end user procurement, followed by home care as the fastest-growing sub-segment. Clinics, sports clubs, and other end users complete the segment picture.
| End User Segment | Key Device Categories | Growth Outlook |
|---|---|---|
| Hospitals | Smart infusion pumps, AI patient monitors, connected ventilators | Steady, mandate-driven |
| Home Care | CGM devices, blood pressure monitors, digital spirometers | Fastest-growing |
| Clinics | Portable smart diagnostics, point-of-care devices | Moderate |
| Sports Clubs | HRV monitors, biosensors, recovery devices | Moderate |
| Others | Rehabilitation, corporate health, research | Expanding |
Hospitals drive the largest share of smart device procurement in Japan, supported by government digitalisation mandates under the Society 5.0 framework. Smart infusion pumps, AI-enabled patient monitors, and connected ventilators represent the primary hospital procurement categories. Home care holds the fastest-growing position among all end user segments, supported by Japan's long-term care insurance expansion and post-pandemic preference for remote monitoring. The Japan remote patient monitoring market is scaling in parallel, with DexCom CGM, Beurer blood pressure monitors, and Microlife digital spirometers leading home care device adoption.
By Distribution Channel
Pharmacies currently hold the largest distribution share, with the online channel growing fastest.
| Distribution Channel | Key Device Types | Notable Activity (2025) |
|---|---|---|
| Pharmacies | Home-use blood glucose monitors, blood pressure devices | Welcia, Matsumoto Kiyoshi, Tsuruha Holdings |
| Online Channel | CGM, wearable monitors, self-monitoring devices | Amazon Japan, Rakuten Medical |
| Others | High-value Class III devices, hospital procurement | Medical trading companies, direct distributors |
Japan's approximately 61,000 registered pharmacies (MHLW) serve as the primary over-the-counter channel for home-use smart devices. The online channel is growing fastest, supported by Japan's high e-commerce penetration. PMDA governs online medical device sales under the Pharmaceutical and Medical Device Act, distinguishing general devices sold freely online from specially controlled devices subject to restrictions.
Regulatory and Import Landscape, Japan Smart Medical Device
International smart device manufacturers face a multi-stage Japan market entry process that differs materially from FDA and CE Mark frameworks. Misclassifying a smart device under Japan's three-tier PMDA system, General, Controlled, and Specially Controlled, delays market entry by up to 24 months and triggers additional clinical data requirements.
| PMDA Device Class | Classification | Example Smart Device | Approval Pathway |
|---|---|---|---|
| General Medical Device | Low risk | Basic wearables, blood pressure monitors | Notification |
| Controlled Medical Device | Moderate risk | Smart glucose monitors, ECG devices | Third-party conformity |
| Specially Controlled Medical Device | High risk | Implantable cardiac monitors, closed-loop insulin systems | PMDA review |
The Pharmaceutical and Medical Device Act (Yakki-ho) governs all device classifications and approval requirements. MHLW's 2023 digital health reform roadmap introduces an accelerated SaMD pathway for AI-enabled devices, providing a faster route to market for software-based diagnostics. Specially Controlled Medical Device designation applies to implantable smart devices from Medtronic and DexCom, requiring the most rigorous PMDA review process.
Competitive Landscape, Japan Smart Medical Device Market
The top five companies by Japan market presence are Abbott Laboratories, Medtronic PLC, Roche Holding AG, DexCom Inc, and Masimo Corp. Draegerwerk AG, SmartCardia, Microlife Corp, and Beurer are additional active participants in the market.
| Company | Key Product in Japan | 2024 to 2025 Action | Primary End User |
|---|---|---|---|
| Abbott Laboratories | FreeStyle Libre CGM | NHI reimbursement listing for FreeStyle Libre 3 (2024) | Home Care, Pharmacies |
| Medtronic PLC | MiniMed 780G insulin pump | PMDA approval for closed-loop system (2023) | Hospitals, Home Care |
| Roche Holding AG | cobas point-of-care platform | Regional hospital network expansion (2024) | Hospitals, Clinics |
| DexCom Inc | G7 CGM system | PMDA approval and NHI listing (2023) | Home Care, Pharmacies |
| Masimo Corp | Root patient monitoring platform | Expanded hospital distribution network (2024) | Hospitals |
- Abbott Laboratories operates as the volume leader in Japan's smart wearable diagnostics segment through FreeStyle Libre, distributed across pharmacies and online channels under PMDA approval. NHI reimbursement listing for FreeStyle Libre 3 in 2024 reduced out-of-pocket cost for Type 2 diabetes patients and accelerated home care channel adoption. This positions Abbott ahead of all competitors in the fastest-growing end user segment through 2034.
- Medtronic PLC supplies hospital-grade smart cardiac monitoring systems and connected surgical devices through its Japan subsidiary. PMDA approval for the MiniMed 780G closed-loop insulin delivery system in 2023 made Medtronic the first-mover in hybrid closed-loop systems in Japan, a device prescribed in hospital and used at home, simultaneously strengthening both end user segments.
- Roche Holding AG operates through Roche Diagnostics Japan, supplying smart point-of-care diagnostic systems and Accu-Chek blood glucose monitors to hospitals and clinics. The 2024 expansion of the cobas point-of-care platform into regional hospital networks positions Roche in the underserved geography that the government's regional equalisation budget is now actively funding.
- DexCom Inc competes directly with Abbott in the CGM category. G7 PMDA approval and simultaneous NHI reimbursement listing in 2023 reduced DexCom's time-to-revenue in Japan and secured strong placement in the pharmacy channel, the largest distribution route by share.
- Masimo Corp supplies hospital-grade non-invasive patient monitoring systems to Japan's ICU and critical care segments. The 2024 distribution network expansion through a major Japanese medical trading company accelerates Masimo's placement in both metropolitan and regional hospitals, directly targeting the regional growth opportunity identified under the Asia Pacific smart medical devices market framework.
Japan Smart Medical Device Market, Future Outlook 2034
Japan's healthcare system will serve a population where more than one in three people is aged 65 or above by the early 2030s. This demographic trajectory places structural pressure on hospital capacity and creates sustained demand for device-enabled remote care that no policy intervention can fully offset.
The market is projected to reach USD 10.77 billion by 2034, growing at 12.34% CAGR from 2026. Three specific growth levers drive the forecast period: NHI reimbursement expansion to new device categories, SaMD approval pathway growth for AI diagnostics, and online channel scaling for home care device distribution. The Japan digital health market provides the infrastructure underpinning all three.
The convergence of AI diagnostics, wearable biosensors, and expanded regulatory pathways creates a compounding growth dynamic that sustains the 12.34% CAGR through 2034, making this market one of the highest-growth medical device opportunities in the Asia Pacific region.
Why Choose This Report?
- Covers the Japan Smart Medical Devices Market with verified 2025 base data and a 2026 to 2034 forecast
- Provides segment-level share data across end user and distribution channel dimensions
- Includes a dedicated pricing analysis section not available in competing reports
- Maps the PMDA regulatory framework and 2023 reform roadmap for market entry planning
- Profiles the top five companies by Japan market presence with recent action data
- Highlights recent developments from 2024 to 2025 with named companies and market implications
- Covers regional distribution across Kanto, Kansai, and other major healthcare regions
- Supports investment, procurement, and market entry decisions with actionable dat
Table of Contents
- Introduction
- Objective of the study
- Product Definition
- Market Segmentation
- Study Variables
- Research Methodology
- Secondary Data Points
- Companies Interviewed
- Primary Data Points
- Breakdown of Primary Interviews
- Secondary Data Points
- Executive Summary
- Market Dynamics
- Drivers
- Challenges
- Opportunity Assessment
- Recent Trends and Developments
- Policy and Regulatory Landscape
- Japan Smart Medical Devices Market Overview and Forecast Analysis (2021-2034)
- Market Size, By Value, By growth rate (CAGR/USD Billions)
- Demand - Supply Trends
- Market Share, By End User
- Sports Club
- Clinics
- Hospitals
- Home Care
- Others
- Market Share, By Distribution Channel
- Pharmacies
- Online Channel
- Others
- Market Share, By Application
- Auto-Immune Disorders
- Infection Diseases
- Oncology
- Others
- Diabetes
- Sleep Disorder
- Sports And Fitness
- Market Share, By Modality
- Non-Wearable
- Wearable
- Market Share, By Technology
- Pressurized Gas
- Expanding Battery
- Rotary Pump
- Motor-Driven
- Spring-Based
- Others
- Market Share, By Type
- On-Body (Adhesive Patch)
- Hand Held
- Off-Body (Belt-Clip)
- Market Share, By Product Type
- Therapeutic Device
- Diagnostic And Monitoring Devices
- Market Share, By Region
- Kanto
- Kansai
- Chubu
- Kyushu
- Market Share, By Competitors
- Competition Characteristics
- Revenue Shares
- Japan Sports Club Smart Medical Devices Market Overview, 2021-2034F
- By Value (USD Million)
- By Distribution Channel- Market Size & Forecast 2021-2034, USD Million
- By Application- Market Size & Forecast 2021-2034, USD Million
- By Modality- Market Size & Forecast 2021-2034, USD Million
- By Technology- Market Size & Forecast 2021-2034, USD Million
- By Type- Market Size & Forecast 2021-2034, USD Million
- By Product Type- Market Size & Forecast 2021-2034, USD Million
- Japan Clinics Smart Medical Devices Market Overview, 2021-2034F
- By Value (USD Million)
- By Distribution Channel- Market Size & Forecast 2021-2034, USD Million
- By Application- Market Size & Forecast 2021-2034, USD Million
- By Modality- Market Size & Forecast 2021-2034, USD Million
- By Technology- Market Size & Forecast 2021-2034, USD Million
- By Type- Market Size & Forecast 2021-2034, USD Million
- By Product Type- Market Size & Forecast 2021-2034, USD Million
- Japan Hospitals Smart Medical Devices Market Overview, 2021-2034F
- By Value (USD Million)
- By Distribution Channel- Market Size & Forecast 2021-2034, USD Million
- By Application- Market Size & Forecast 2021-2034, USD Million
- By Modality- Market Size & Forecast 2021-2034, USD Million
- By Technology- Market Size & Forecast 2021-2034, USD Million
- By Type- Market Size & Forecast 2021-2034, USD Million
- By Product Type- Market Size & Forecast 2021-2034, USD Million
- Japan Home Care Smart Medical Devices Market Overview, 2021-2034F
- By Value (USD Million)
- By Distribution Channel- Market Size & Forecast 2021-2034, USD Million
- By Application- Market Size & Forecast 2021-2034, USD Million
- By Modality- Market Size & Forecast 2021-2034, USD Million
- By Technology- Market Size & Forecast 2021-2034, USD Million
- By Type- Market Size & Forecast 2021-2034, USD Million
- By Product Type- Market Size & Forecast 2021-2034, USD Million
- Japan Others Smart Medical Devices Market Overview, 2021-2034F
- By Value (USD Million)
- By Distribution Channel- Market Size & Forecast 2021-2034, USD Million
- By Application- Market Size & Forecast 2021-2034, USD Million
- By Modality- Market Size & Forecast 2021-2034, USD Million
- By Technology- Market Size & Forecast 2021-2034, USD Million
- By Type- Market Size & Forecast 2021-2034, USD Million
- By Product Type- Market Size & Forecast 2021-2034, USD Million
- Competitive Outlook (Company Profile - Partial List)
- Abbott Laboratories
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- Medtronic PLC
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- Roche Holding AG
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- DexCom Inc
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- Masimo Corp
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- Draegerwerk AG & Co KGaA
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- SmartCardia
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- Microlife Corp
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- Beurer
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- Others
- Abbott Laboratories
- Contact Us & Disclaimer
Top Key Players & Market Share Outlook
- Abbott Laboratories
- Medtronic PLC
- Roche Holding AG
- DexCom Inc
- Masimo Corp
- Draegerwerk AG & Co KGaA
- SmartCardia
- Microlife Corp
- Beurer
- Others
Frequently Asked Questions






