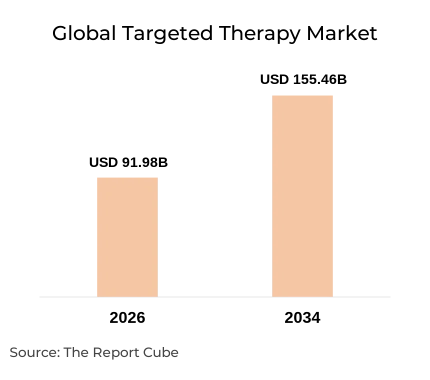
Build Your Custom Market Intelligence Report
Customize Your ReportKey Takeaways
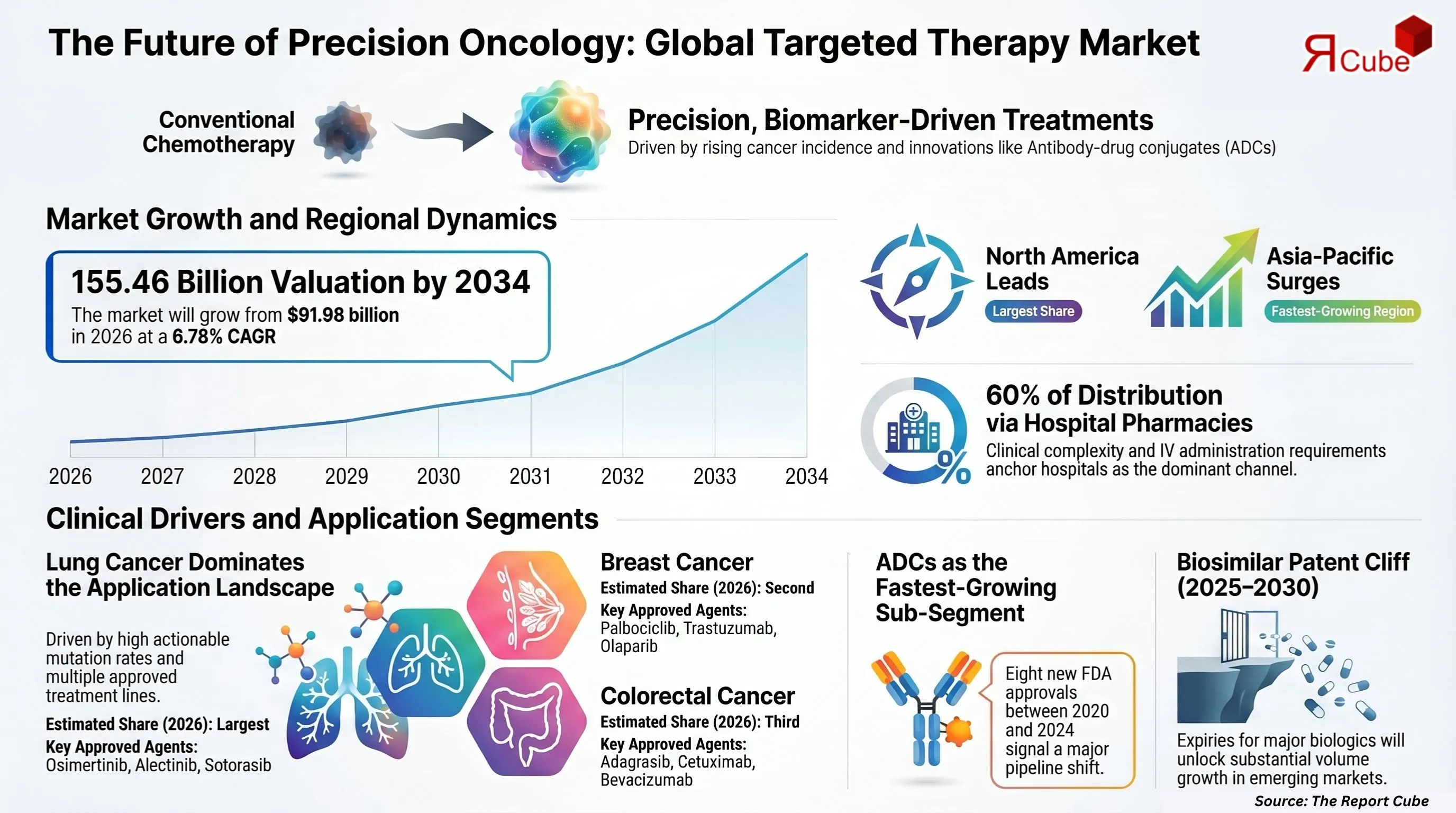
- The global targeted therapy market is valued at USD 91.98 billion in 2026, expanding to USD 155.46 billion by 2034 at a CAGR of 6.78%.
- Lung cancer holds the dominant application segment, driven by a high actionable mutation rate (EGFR, ALK, ROS1) and multiple approved agents across treatment lines.
- Hospital pharmacies account for over 60% of oncology drug dispensing globally, anchoring the distribution channel landscape through 2034.
- North America commands the largest regional share; Asia-Pacific is the fastest-growing region, driven by NMPA regulatory reforms and rising cancer incidence in China and India.
- Antibody-drug conjugates (ADCs) represent the fastest-growing sub-segment, with eight new FDA approvals between 2020 and 2024 alone.
- Biosimilar market entry for key molecules - trastuzumab, bevacizumab, and rituximab - will unlock substantial volume growth in emerging markets between 2025 and 2030.
- High therapy costs (exceeding USD 100,000 per patient annually for first-line agents) remain the single largest access constraint across low- and middle-income countries.
Global Targeted Therapy Market: Insights & Analysis
Cancer remains the second-leading cause of mortality worldwide, yet conventional chemotherapy's non-selective toxicity continues to leave patients with severe side effects and suboptimal response rates. The World Health Organization estimates that cancer caused approximately 10 million deaths in 2022, with inadequate treatment precision contributing to poor survival outcomes across multiple tumour types. This analysis examines how targeted therapy is closing that treatment gap - and why the global targeted therapy market is on course to grow from USD 91.98 billion in 2026 to USD 155.46 billion by 2034 (WHO Cancer Fact Sheet, 2022).
Targeted therapy differs fundamentally from conventional chemotherapy and immunotherapy. Rather than killing all rapidly dividing cells, targeted agents interfere with specific molecular targets - proteins or genes - that drive tumour growth and survival. Mechanistic classes include kinase inhibitors (e.g., EGFR inhibitors, ALK inhibitors), monoclonal antibodies (e.g., trastuzumab, cetuximab), PARP inhibitors (e.g., olaparib), and CDK inhibitors (e.g., palbociclib). Each class targets a distinct oncogenic pathway, enabling therapy selection based on a patient's specific tumour genomics.
Market growth is supported by three structural forces: rising global cancer incidence, the expansion of biomarker-driven prescribing models, and a deepening oncology pipeline. The precision oncology diagnostics market is amplifying demand by enabling clinicians to identify actionable mutations before initiating therapy - shifting prescribing from empirical to evidence-guided decisions. This companion diagnostic infrastructure is a commercial multiplier, not merely a clinical nicety.
Global Targeted Therapy Market Dynamics
Key Market Driver: Rising Global Cancer Incidence and Biomarker-Guided Prescribing
Oncologists prescribing systemic cancer therapies without biomarker validation expose patients to treatments mismatched to their tumour genotype - wasting clinical time and compounding toxicity burden. The International Agency for Research on Cancer projects 20 million new cancer cases annually by 2025, with genomic heterogeneity making non-personalised treatment protocols increasingly untenable (GLOBOCAN 2022). Biomarker-guided prescribing is now the primary growth engine of the targeted therapy market, reshaping formulary decisions across lung, breast, and colorectal oncology.
GLOBOCAN 2022 data confirms lung cancer, breast cancer, and colorectal cancer as the three highest-incidence tumour types with established targeted therapy indications. Lung cancer carries an actionable mutation rate of approximately 15% in Western populations and up to 50% in Asian non-small cell lung cancer (NSCLC) cohorts - predominantly EGFR mutations - generating sustained demand for EGFR inhibitors including osimertinib. The FDA's accelerated approval pathway has expanded targetable biomarker indications significantly, with approvals covering EGFR, HER2, KRAS G12C, and BRAF V600E mutations across multiple tumour types.
Major Industry Challenge: High Cost of Targeted Agents and Reimbursement Gaps
A full course of first-line targeted therapy for EGFR-mutant non-small cell lung cancer can exceed USD 150,000 per patient annually - placing treatment entirely out of reach for patients in markets without comprehensive reimbursement coverage. Across low- and middle-income countries, fewer than 30% of cancer patients have access to any form of targeted therapy, according to the Lancet Oncology Commission - directly capping the market's addressable volume (Lancet Oncology Commission on Cancer Access, 2020). Reimbursement policy fragmentation and pricing pressure from payers represent the single largest constraint on accelerating global uptake through 2034.
Cost disparities between targeted agents and conventional chemotherapy are stark. Published cost-effectiveness analyses document targeted agent annual costs averaging 8–12 times those of standard cytotoxic regimens. Reimbursement status varies sharply across geographies: Medicare Part D provides broad oncology coverage in the United States, while access across EU member states depends on individual national health technology assessment decisions following EMA centralised approval - creating a patchwork of inclusion timelines that delays patient access by months to years in smaller European markets.
Need Specific Data, Let’s Customize the Report for You
Request CustomizationEmerging Trend: Antibody-Drug Conjugates (ADCs) Expanding the Targeted Therapy Landscape
Monoclonal antibodies and small-molecule inhibitors have defined targeted therapy for two decades, but their efficacy ceiling - particularly in treatment-refractory patients - is prompting a structural shift in oncology pipeline investment. Between 2020 and 2024, the FDA approved eight new antibody-drug conjugates (ADCs), more than in the preceding 15 years combined - signalling a pipeline inflection that will reshape formulary compositions through 2034 (FDA Hematology/Oncology Approvals, 2024). ADCs represent the fastest-growing sub-segment of the targeted therapy market, combining tumour-targeting specificity with cytotoxic payload delivery.
The ADC architecture links a tumour-specific monoclonal antibody to a cytotoxic payload via a chemical linker - delivering high-potency cell-killing agents directly to antigen-expressing cancer cells while limiting systemic exposure. Key approved ADCs include trastuzumab deruxtecan (Enhertu), approved for HER2-positive and HER2-low breast cancer and NSCLC, and sacituzumab govitecan (Trodelvy), approved for triple-negative breast cancer and urothelial carcinoma. As of 2024, over 100 ADC candidates are in Phase II or Phase III clinical trials globally, with breast cancer and lung cancer application segments positioned to benefit most from near-term approvals.
Emerging Opportunity: Biosimilar Expansion Unlocking Volume Growth in Emerging Markets
Originator targeted therapies command patent-protected pricing that excludes the majority of patients across Asia, Latin America, and Africa from treatment access entirely. The combined biologic oncology patent cliff between 2025 and 2030 will open biosimilar competition across USD 40+ billion in annual targeted therapy revenues, according to IQVIA Institute projections (IQVIA Institute for Human Data Science, 2024). Biosimilar market entry in key emerging economies represents the highest-volume growth opportunity for the global targeted therapy market through 2034.
Patent expiry timelines for the three highest-volume oncology biologics are already in motion: trastuzumab (US patent expired 2019), bevacizumab (US patent expired 2019), and rituximab (US patent expired 2018). The FDA has approved over 20 oncology biosimilars through 2025, with EMA approval counts tracking similarly. Market penetration rates for trastuzumab biosimilars in India and China now exceed 40% of trastuzumab volume in those markets - compared to approximately 20% in the United States - demonstrating that emerging markets are leading biosimilar adoption, not trailing it.
Global Targeted Therapy Market - Segment-wise Analysis
By Application
- Lung
- Breast
- Colorectal
- Renal Cancer
- Others
Not all cancer types attract equal targeted therapy investment - tumour biology, mutation prevalence, and diagnostic infrastructure create sharp disparities in segment maturity and commercial priority. Lung cancer alone accounted for the largest share of targeted therapy revenues in 2026, driven by a high actionable mutation rate and the availability of multiple approved agents across treatment lines. The table below maps each application segment by estimated 2026 share, growth outlook, and key approved agents.
|
Application Segment |
Estimated Share (2026) |
Growth Outlook |
Key Approved Agents |
|---|---|---|---|
|
Lung Cancer |
Largest |
High |
Osimertinib, Alectinib, Sotorasib |
|
Breast Cancer |
Second |
High |
Palbociclib, Trastuzumab, Olaparib |
|
Colorectal Cancer |
Third |
Medium-High |
Adagrasib, Cetuximab, Bevacizumab |
|
Renal Cancer |
Fourth |
Medium |
Sunitinib, Everolimus, Nivolumab+Ipilimumab |
|
Others |
Fifth |
Medium |
Vemurafenib, Imatinib, Ibrutinib |
Lung cancer leads despite lower global incidence than breast cancer because its actionable mutation rate - approximately 15% in Western and up to 50% in Asian NSCLC cohorts - creates a large biomarker-defined patient population eligible for targeted agents. Breast cancer's HER2-positive subtype (~20% of all breast cancers) and hormone-receptor-positive subtype (eligible for CDK4/6 inhibitors) together generate the second-largest targeted therapy revenue pool. Colorectal cancer gained significant momentum with the 2021–2022 approvals of KRAS G12C inhibitors - sotorasib and adagrasib - opening a previously undruggable mutation present in approximately 13% of colorectal cancers. The NSCLC targeted therapy market remains the single largest sub-market within this report's application scope.
By Distribution Channel
- Hospital
- Retail
- Online Pharmacies
|
Channel |
Share (2026) |
Growth Tag |
Key Drivers |
|---|---|---|---|
|
Hospital Pharmacies |
Dominant |
Stable-High |
IV agents, clinical oversight, payer routing |
|
Retail Pharmacies |
Second |
Medium |
Oral generic targeted agents post-patent |
|
Online Pharmacies |
Emerging |
High |
Patient convenience, oral agents, adherence tools |
Hospital pharmacies managed over 60% of oncology drug dispensing globally in 2024, reflecting the clinical complexity of intravenous targeted agent initiation and the payer requirement for specialist oversight. Retail pharmacies serve a growing role in oral targeted agent dispensing - particularly for generic imatinib and erlotinib - supported by specialty pharmacy infrastructure. Online pharmacies are emerging as a channel for repeat dispensing of stable oral targeted therapy patients, subject to regulatory compliance frameworks including the US Drug Supply Chain Security Act (DSCSA) and EU Falsified Medicines Directive (FMD).
Global Targeted Therapy Market - Recent Developments (2025-2026)
The competitive landscape of targeted therapy shifts faster than most pharmaceutical segments - approval decisions, licensing deals, and trial readouts alter market positioning within single quarters. Between Q1 2025 and mid-2026, the market saw at least 15 oncology drug approvals and a wave of high-value M&A deals that are reshaping the competitive hierarchy across lung cancer and breast cancer segments.
- January 2025 - AstraZeneca / Daiichi Sankyo | Dato-DXd (Datroway) - FDA Approval The FDA approved datopotamab deruxtecan (Dato-DXd; Datroway), a TROP2-directed ADC developed by Daiichi Sankyo and AstraZeneca, for HR-positive, HER2-negative metastatic breast cancer in patients who had received prior systemic therapy. The Phase 3 TROPION-Breast01 trial demonstrated that Dato-DXd improved progression-free survival compared with chemotherapy (median PFS: 6.9 months vs. 4.9 months).
- February 2025 - Pfizer / Arvinas | Vepdegestrant - Phase 3 Endpoint Met Pfizer and Arvinas announced that vepdegestrant, a PROTAC ER degrader, met its Phase 3 primary endpoint in ER-positive breast cancer - positioning the asset for a potential FDA filing later in 2025.
- May 2025 - AbbVie | Telisotuzumab vedotin-tllv (EMRELIS) - FDA Accelerated Approval The FDA granted accelerated approval to EMRELIS (telisotuzumab vedotin-tllv), developed by AbbVie, for adult patients with locally advanced or metastatic non-squamous NSCLC with high c-Met protein overexpression who have received prior systemic therapy.
Regional Projection of the Global Targeted Therapy Market
Targeted therapy market growth is not geographically uniform - regulatory approval speed, reimbursement policy depth, and oncology infrastructure quality create performance gaps exceeding 10 percentage points in CAGR across regions. North America commands the largest global share in 2026, yet the fastest volume growth is occurring in Asia-Pacific, where rising cancer incidence intersects with expanding health insurance coverage. The table below projects regional trajectories through 2034.
|
Region |
Share 2026 |
Growth Outlook |
Key Driver |
Access Gap |
|---|---|---|---|---|
|
North America |
Largest |
Moderate-High |
FDA pipeline, high payer coverage |
Low |
|
Europe |
Second |
Moderate |
EMA approvals, price referencing |
Medium |
|
Asia-Pacific |
Third |
Highest CAGR |
China NMPA reforms, rising incidence |
Closing |
|
Latin America |
Fourth |
Moderate |
Brazil ANVISA modernisation |
High |
|
Middle East & Africa |
Fifth |
Low-Moderate |
UAE/Saudi oncology infrastructure |
High |
- North America holds the largest share, backed by FDA approval pipeline depth, Medicare and commercial payer oncology coverage, and a high concentration of oncology-specialised treatment centres.
- Europe occupies the second position, with EMA centralised approvals offset by national price referencing constraints - Germany's AMNOG process, for example, frequently compresses launch pricing for targeted agents.
- Asia-Pacific carries the highest forecast CAGR, driven by China's NMPA accelerated oncology review pathway, India's biosimilar production capability, and Japan's all-payer reimbursement system.
- Latin America shows moderate growth anchored by Brazil's ANVISA regulatory modernization.
- Middle East & Africa remains nascent but is advancing through oncology infrastructure investment in the UAE and Saudi Arabia. The Asia-Pacific cancer therapeutics market is the region to watch for volume leadership beyond 2030.
Global Targeted Therapy Market: Future Outlook (2034)
Projecting the targeted therapy market to 2034 requires more than extrapolating the current 6.78% CAGR - five structural forces will materially alter the market's composition well before the forecast period closes. ADC pipeline maturation, AI-assisted drug discovery, liquid biopsy integration, biosimilar volume expansion, and oral targeted agent proliferation together represent forces capable of adding tens of billions in annual market volume that headline revenue estimates do not yet fully capture. This section outlines where the global targeted therapy market is going - and why - beyond the USD 155.46 billion figure.
Why Choose This Report?
- Verified market sizing: USD 91.98B (2026) to USD 155.46B (2034) at 6.78% CAGR with transparent methodology
- Full segment coverage: 5 application categories × 3 distribution channels
- Competitive intelligence: 13 profiled key players including Pfizer, AstraZeneca, Roche, and Amgen
- Regional breakdown: 5 geographies with country-level data for major markets
- Recent developments tracker: 2025–2026 regulatory, pipeline, and deal activity
- Strategic recommendations: calibrated to pharma manufacturers, distributors, and investors
- Customisation available: segment add-ons, country deep-dives, and competitor benchmarking modules
Table of Contents
- Introduction
- Objective of the study
- Product Definition
- Market Segmentation
- Study Variables
- Research Methodology
- Secondary Data Points
- Companies Interviewed
- Primary Data Points
- Breakdown of Primary Interviews
- Secondary Data Points
- Executive Summary
- Market Dynamics
- Drivers
- Challenges
- Opportunity Assessment
- Recent Trends and Developments
- Policy and Regulatory Landscape
- Global Targeted Therapy Market Overview and Forecast Analysis (2021-2034)
- Market Size, By Value, By growth rate (CAGR/USD Billions)
- Demand - Supply Trends
- Market Share, By Type
- Monoclonal Antibodies
- Small Molecule
- Market Share, By Application
- Lung Cancer
- Breast Cancer
- Colorectal Cancer
- Renal Cancer
- Others
- Market Share, By Distribution Channel
- Hospital Pharmacies
- Retail Pharmacies
- Online Pharmacies
- Market Share, By Region
- North America
- Europe
- Asia-Pacific
- Latin America
- Middle East & Africa
- Market Share, By Company
- Revenue Shares
- Competition Characteristics
- North America Targeted Therapy Market Overview (2021-2034)
- Market Size, By Value (in USD Billions)
- Market Share, By Type
- Market Share, By Application
- Market Share, By Distribution Channel
- By Country
- The US
- Canada
- Mexico
- The US Targeted Therapy Market Overview (2021-2034)
- Market Share, By Type
- Market Share, By Application
- Market Share, By Distribution Channel
- Canada Targeted Therapy Market Overview (2021-2034)
- Market Share, By Type
- Market Share, By Application
- Market Share, By Distribution Channel
- Mexico Targeted Therapy Market Overview (2021-2034)
- Market Share, By Type
- Market Share, By Application
- Market Share, By Distribution Channel
- South America Targeted Therapy Market Overview (2021-2034)
- Market Size, By Value (in USD Billions)
- Market Share, By Type
- Market Share, By Application
- Market Share, By Distribution Channel
- By Country
- Brazil
- Argentina
- Rest of South America
- Brazil Targeted Therapy Market Overview (2021-2034)
- Market Share, By Type
- Market Share, By Application
- Market Share, By Distribution Channel
- Argentina Targeted Therapy Market Overview (2021-2034)
- Market Share, By Type
- Market Share, By Application
- Market Share, By Distribution Channel
- Europe Targeted Therapy Market Overview (2021-2034)
- Market Size, By Value (in USD Billions)
- By Type
- By Application
- By Distribution Channel
- By Country
- Germany
- The UK
- France
- Spain
- Italy
- Rest of Europe
- Germany Targeted Therapy Market Overview (2021-2034)
- Market Share, By Type
- Market Share, By Application
- Market Share, By Distribution Channel
- France Targeted Therapy Market Overview (2021-2034)
- Market Share, By Type
- Market Share, By Application
- Market Share, By Distribution Channel
- The UK Targeted Therapy Market Overview (2021-2034)
- Market Share, By Type
- Market Share, By Application
- Market Share, By Distribution Channel
- Spain Targeted Therapy Market Overview (2021-2034)
- Market Share, By Type
- Market Share, By Application
- Market Share, By Distribution Channel
- Italy Targeted Therapy Market Overview (2021-2034)
- Market Share, By Type
- Market Share, By Application
- Market Share, By Distribution Channel
- The Middle East & Africa Targeted Therapy Market Overview (2021-2034)
- Market Size, By Value (in USD Billions)
- By Type
- By Application
- By Distribution Channel
- By Country
- The UAE
- Saudi Arabia
- South Africa
- Rest of the Middle East & Africa
- The UAE Targeted Therapy Market Overview (2021-2034)
- Market Share, By Type
- Market Share, By Application
- Market Share, By Distribution Channel
- Saudi Arabia Targeted Therapy Market Overview (2021-2034)
- Market Share, By Type
- Market Share, By Application
- Market Share, By Distribution Channel
- South Africa Targeted Therapy Market Overview (2021-2034)
- Market Share, By Type
- Market Share, By Application
- Market Share, By Distribution Channel
- Asia-Pacific Targeted Therapy Market Overview (2021-2034)
- Market Size, By Value (in USD Billions)
- By Type
- By Application
- By Distribution Channel
- By Country
- China
- India
- Japan
- South Korea
- Australia
- Rest of Asia-Pacific
- China Targeted Therapy Market Overview (2021-2034)
- Market Share, By Type
- Market Share, By Application
- Market Share, By Distribution Channel
- India Targeted Therapy Market Overview (2021-2034)
- Market Share, By Type
- Market Share, By Application
- Market Share, By Distribution Channel
- Japan Targeted Therapy Market Overview (2021-2034)
- Market Share, By Type
- Market Share, By Application
- Market Share, By Distribution Channel
- South Korea Targeted Therapy Market Overview (2021-2034)
- Market Share, By Type
- Market Share, By Application
- Market Share, By Distribution Channel
- Austraila Targeted Therapy Market Overview (2021-2034)
- Market Share, By Type
- Market Share, By Application
- Market Share, By Distribution Channel
- Competitive Outlook (Company Profile - Partial List)
- Pfizer, Inc.
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- AstraZeneca Plc.
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- Amgen
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- Inc. F. Hoffmann-La Roche & Co.
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- Genentech, Inc.
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- Agenus, Inc.
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- Celdara Medical LLC
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- Arcus Biosciences, Inc.
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- Aurinia Pharmaceuticals Inc.
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- Gilead Sciences, Inc.
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- Serena Therapeutics Inc.
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- Nektar Therapeutics Inc.
- Company Overview
- Business Segments
- Strategic Alliances/Partnerships
- Recent Developments
- Others
- Pfizer, Inc.
- Disclaimer
Top Key Players & Market Share Outlook
- Pfizer, Inc.
- AstraZeneca Plc.
- Amgen
- Inc. F. Hoffmann-La Roche & Co.
- Genentech, Inc.
- Agenus, Inc.
- Celdara Medical LLC
- Arcus Biosciences, Inc.
- Aurinia Pharmaceuticals Inc.
- Gilead Sciences, Inc.
- Serena Therapeutics Inc.
- Nektar Therapeutics Inc.
- Others
Frequently Asked Questions






