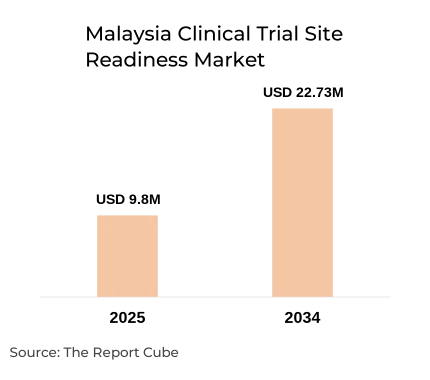
Build Your Custom Market Intelligence Report
Customize Your ReportMalaysia Clinical Trial Site Readiness Market Key Highlights
By Service Type:
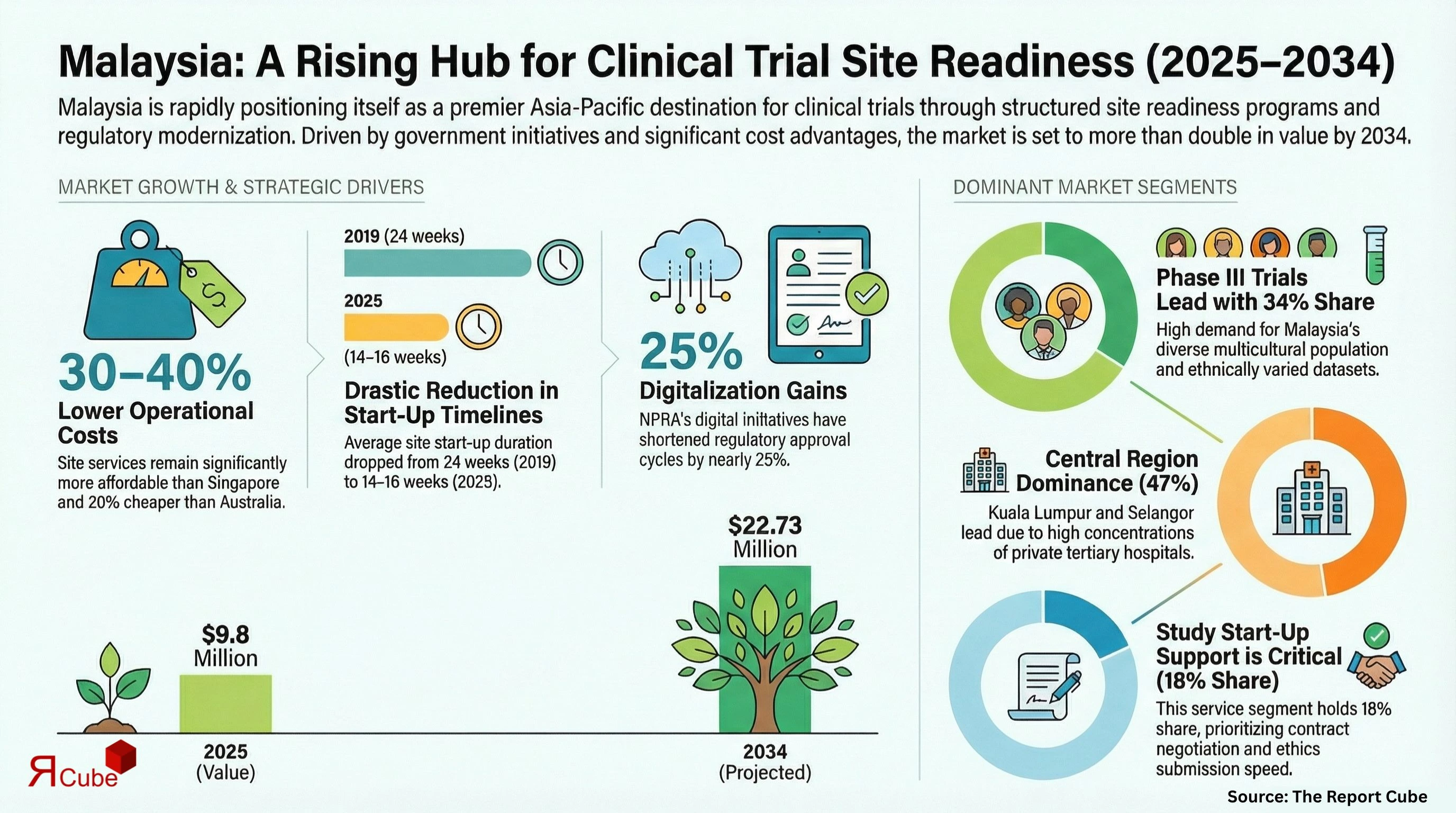
Study Start-Up Support segment leads the market, with around 18% share.
By Clinical Trial Phase:
Phase III segment dominates the market, accounting for nearly 34% of total revenue.
Regional Outlook:
Central region dominates the malaysia clinical trial site readiness market, with around 47% share.
Government Initiatives & Policies
- 12th Malaysia Plan (RMK-12) Healthcare & Research Strengthening Framework (Updated 2025 Review): Emphasizes expansion of clinical research capacity, digital health integration, and public-private collaboration to position Malaysia as a regional clinical trials hub. Official Portal: https://rmk12.epu.gov.my
- National Pharmaceutical Regulatory Agency (NPRA) Enhanced Clinical Trial Application (CTA) Digitalization Initiative 2025: Streamlines regulatory review timelines and improves site activation efficiency through electronic submission platforms. NPRA Portal: https://npra.gov.my
Malaysia Clinical Trial Site Readiness Market Insights & Analysis
The Malaysia Clinical Trial Site Readiness Market is anticipated to register a CAGR of 9.8% during 2026-34. The market size was valued at USD 9.8 million in 2025 and is projected to reach USD 22.73 million by 2034. Malaysia is strengthening its position as a preferred destination for multicenter clinical trials in Asia Pacific through structured site readiness programs, regulatory modernization, and public-private research partnerships.
Malaysia’s healthcare infrastructure, supported by over 150 Ministry of Health hospitals and a growing number of private tertiary care centers, provides a strong foundation for clinical research expansion. The country conducts more than 1,500 industry-sponsored and investigator-initiated trials annually, with oncology, infectious diseases, and metabolic disorders leading therapeutic demand. Government-backed initiatives led by Clinical Research Malaysia (CRM) and the Institute for Clinical Research (ICR) have significantly improved trial activation timelines, reducing average site start-up duration from 24 weeks in 2019 to nearly 14-16 weeks in 2025. Faster ethics approvals and centralized contracting frameworks have improved sponsor confidence.
Malaysia’s multicultural population of over 33 million enhances patient recruitment diversity, particularly for Phase III global studies requiring ethnically varied datasets. Electronic health record adoption in major hospitals such as Subang Jaya Medical Centre and Sunway Medical Centre enables structured patient identification, accelerating enrollment rates. On average, oncology sites in Central Malaysia report recruitment rates 18-22% faster than regional Southeast Asian peers.
In addition, Malaysia benefits from competitive operational costs, which remain approximately 30-40% lower than Singapore and 20% lower than Australia for comparable site services. Contract research organizations such as IQVIA Malaysia, Labcorp Drug Development, Parexel Malaysia, and Novotech Malaysia are expanding integrated site readiness support, including regulatory navigation, SOP development, and quality management systems. This coordinated ecosystem, supported by digital transformation and trained clinical investigators, is driving rapid market expansion.
Malaysia Clinical Trial Site Readiness Market Dynamics
Key Driver: Government-Backed Ecosystem Strengthening and Faster Regulatory Timelines
Malaysia’s structured approach to clinical research governance is a primary growth catalyst. CRM’s national site network model ensures standardized feasibility assessments, contracting templates, and centralized budgeting processes. Regulatory reforms under NPRA’s digital CTA system have shortened approval cycles by nearly 25% compared to pre-2022 benchmarks. Ethics committee harmonization across public hospitals has reduced duplicative reviews, improving predictability for sponsors. These enhancements reduce trial delays and lower start-up costs, making Malaysia highly attractive for Phase II and Phase III multinational trials. Furthermore, tax incentives for R&D and reinvestment allowances for healthcare facilities encourage private hospital participation in industry-sponsored trials.
Book your FREE 30-minute expert consultation today
Contact UsIndustry Trends: Hybrid Site Readiness Models and AI-Enabled Trial Optimization
The market is witnessing rapid adoption of hybrid deployment models combining onsite readiness audits with centralized remote monitoring. Remote feasibility assessments and digital document management platforms are reducing administrative burdens by up to 30%. AI-driven patient matching systems integrated within hospital databases improve screening accuracy and minimize screen failure rates. Sponsors increasingly demand centralized site readiness hubs capable of training multiple hospitals simultaneously, improving scalability. Oncology and rare disease studies particularly benefit from digital pre-screening tools, which enhance enrollment efficiency and data quality. As decentralized trials expand, Malaysia’s readiness services are adapting to include telemedicine workflows and remote data verification capabilities.
Major Challenge: Investigator Capacity Constraints and Uneven Regional Infrastructure
Although Malaysia has strong urban hospital infrastructure, certain northern and east coast regions face limited investigator availability and research-dedicated staff. Principal investigators often balance heavy clinical workloads, constraining trial throughput. Smaller hospitals may lack advanced laboratory or imaging capabilities required for complex Phase I oncology trials. While CRM provides training and certification programs, scaling experienced coordinators remains a challenge. Additionally, competition from neighboring countries such as Thailand and South Korea increases pressure to maintain competitive turnaround times. Addressing workforce development and regional infrastructure disparities is critical to sustaining long-term growth.
Opportunity: Expansion of Early-Phase Trials and Biotech-Focused Studies
Malaysia is increasingly targeting Phase I and bioequivalence studies through upgraded clinical pharmacology units. Early-phase trial volumes are projected to grow at over 30% annually through 2030. Biotech sponsors seeking cost-efficient Asia Pacific entry points are leveraging Malaysia’s regulatory predictability and English-speaking workforce. Dedicated clinical research centers and academic medical institutions are expanding laboratory accreditation to meet global Good Clinical Practice standards. Additionally, partnerships between CROs and tertiary hospitals are creating end-to-end readiness frameworks that improve repeat business from global pharmaceutical sponsors.
Malaysia Clinical Trial Site Readiness Market Segment-wise Analysis
By Service Type:
- Site Feasibility Assessment
- Site Selection Support
- Site Infrastructure Setup
- Standard Operating Procedure Development
- Regulatory Readiness Support
- Quality Management System Setup
- Staff Training and Certification
- Patient Recruitment and Engagement Support
- Study Start-Up Support
- Ongoing Site Performance Management
- Others
Study Start-Up Support accounts for approximately 18% of total market revenue, reflecting sponsors’ prioritization of activation speed. Services include contract negotiation, ethics submission preparation, investigator meetings, and regulatory dossier compilation. Reducing activation time by even four weeks can yield cost savings of 8-12% per trial. CRM-led standardized contracting templates have accelerated start-up cycles across public hospitals. High sponsor demand for predictable launch timelines positions this segment as the most commercially critical readiness service.
By Clinical Trial Phase:
- Phase I
- Phase II
- Phase III
- Phase IV
- Bioequivalence and Observational Studies
Phase III dominates with nearly 34% market share due to multinational pharmaceutical demand for large patient populations and diverse ethnicity representation. Oncology, cardiovascular, and metabolic disorder studies represent the largest share within this phase. Malaysian sites consistently achieve enrollment targets within 90-110% of projected timelines, enhancing global sponsor trust. Phase I growth is accelerating but remains secondary to late-phase global confirmatory trials.
Regional Projection of Malaysia Clinical Trial Site Readiness Industry
- Central
- Northern
- Southern
- East
- Others
The Central region, encompassing Kuala Lumpur and Selangor, leads with around 47% market share. The presence of major tertiary hospitals such as Subang Jaya Medical Centre, Sunway Medical Centre, and Pantai Hospital Kuala Lumpur enhances research capacity. Strong private hospital participation, concentration of CRO offices, and proximity to regulatory authorities accelerate site coordination. Northern and Southern regions are expanding steadily, while East Malaysia is gaining traction through CRC Sarawak General Hospital initiatives.
Malaysia Clinical Trial Site Readiness Market Recent Developments
- 2025: Clinical Research Malaysia (CRM) launched CRM Trial Connect 2025, hosting over 1,000 delegates and 190 organizations, reinforcing Malaysia’s positioning as a regional clinical trial hub and strengthening national site collaboration frameworks.
- 2025: Novotech Malaysia announced participation in CRM Trial Connect 2025, highlighting integrated CRO solutions supporting biotech and mid-sized pharmaceutical sponsors expanding into Malaysia’s clinical research ecosystem.
- 2025: Parexel Malaysia entered a strategic AI-enabled partnership with Paradigm Health to expand technology-driven clinical trial execution models, improving access, efficiency, and scalability across Asia Pacific markets including Malaysia.
Need insights for a specific region within this market?
Request Regional DataWhy Choose This Report?
- Provides a comprehensive overview of the overall market analysis, encompassing key trends, consumer behavior analysis, and risk assessment to support strategic decision-making.
- Provides accurate, up-to-date insights into market size, segmentation, and emerging opportunities, helping to minimize risk & capitalizing on growth.
- Gives deep understanding of target audience preferences, investment habits, and communication channels for enhanced product development & marketing effectiveness.
- Delivers competitive analysis & benchmarking, uncovering the strengths & weaknesses of market competitors to guide strategies.
- Consolidate comprehensive market intelligence, reducing reasoning & streamlining research efforts.
- Facilitates customized market segmentation & risk mitigation strategies, fine-tuned to the business objectives.
- Aids in identifying both market challenges & untapped opportunities within the industry to drive long-term business growth.
- Provides valuable information based on actual customer data & search trends.
Table of Contents
- Introduction
- Objective of the Study
- Product and Category Definition
- Market Segmentation
- Study Variables
- Research Methodology
- Secondary Data Points
- Breakdown of Secondary Sources
- Primary Data Points
- Breakdown of Primary Interviews
- Secondary Data Points
- Executive Summary
- Market Dynamics
- Drivers
- Challenges
- Opportunity Assessment
- Recent Trends and Developments
- Regulatory and Policy Landscape
- Malaysia Clinical Trial Site Readiness Market Overview (2021-2034)
- Market Size, By Value (USD Million)
- Market Share, By Service Type
- Site Feasibility Assessment
- Site Selection Support
- Site Infrastructure Setup
- Standard Operating Procedure Development
- Regulatory Readiness Support
- Quality Management System Setup
- Staff Training and Certification
- Patient Recruitment and Engagement Support
- Study Start-Up Support
- Ongoing Site Performance Management
- Others
- Market Share, By Clinical Trial Phase
- Phase I
- Phase II
- Phase III
- Phase IV
- Bioequivalence and Observational Studies
- Market Share, By Therapeutic Area
- Oncology
- Cardiovascular Diseases
- Metabolic and Endocrine Disorders
- Infectious Diseases
- Central Nervous System Disorders
- Respiratory Diseases
- Autoimmune and Inflammatory Diseases
- Others
- Market Share, By Site Type
- Public Hospitals
- Private Hospitals
- Dedicated Clinical Research Centers
- Academic Medical Centers
- Specialist Clinics
- Others
- Market Share, By Sponsor Type
- Pharmaceutical Companies
- Biotechnology Companies
- Medical Device Companies
- Contract Research Organizations
- Academic and Government Sponsors
- Others
- Market Share, By Deployment Model
- Onsite Support
- Remote and Hybrid Support
- Centralized Site Readiness Hubs
- Market Share, By Region
- Northern
- Central
- Southern
- East
- Others
- Market Share, By Company
- Revenue Shares and Analysis
- Competitive Landscape
- Northern Malaysia Clinical Trial Site Readiness Market
- Market Size, By Value (USD Million)
- Market Share, By Service Type
- Market Share, By Clinical Trial Phase
- Market Share, By Therapeutic Area
- Market Share, By Site Type
- Market Share, By Sponsor Type
- Market Share, By Deployment Model
- Central Malaysia Clinical Trial Site Readiness Market
- Market Size, By Value (USD Million)
- Market Share, By Service Type
- Market Share, By Clinical Trial Phase
- Market Share, By Therapeutic Area
- Market Share, By Site Type
- Market Share, By Sponsor Type
- Market Share, By Deployment Model
- Southern Malaysia Clinical Trial Site Readiness Market
- Market Size, By Value (USD Million)
- Market Share, By Service Type
- Market Share, By Clinical Trial Phase
- Market Share, By Therapeutic Area
- Market Share, By Site Type
- Market Share, By Sponsor Type
- Market Share, By Deployment Model
- East Malaysia Clinical Trial Site Readiness Market
- Market Size, By Value (USD Million)
- Market Share, By Service Type
- Market Share, By Clinical Trial Phase
- Market Share, By Therapeutic Area
- Market Share, By Site Type
- Market Share, By Sponsor Type
- Market Share, By Deployment Model
- Competitive Outlook and Company Profiles
- Clinical Research Malaysia (CRM)
- Company Overview
- Service Portfolio
- Strategic Alliances/Partnerships
- Recent Developments
- Institute for Clinical Research (ICR)
- Company Overview
- Service Portfolio
- Strategic Alliances/Partnerships
- Recent Developments
- IQVIA Malaysia
- Company Overview
- Service Portfolio
- Strategic Alliances/Partnerships
- Recent Developments
- SGS Malaysia
- Company Overview
- Service Portfolio
- Strategic Alliances/Partnerships
- Recent Developments
- Labcorp Drug Development
- Company Overview
- Service Portfolio
- Strategic Alliances/Partnerships
- Recent Developments
- CMIC Malaysia
- Company Overview
- Service Portfolio
- Strategic Alliances/Partnerships
- Recent Developments
- Emerald Clinical Trials
- Company Overview
- Service Portfolio
- Strategic Alliances/Partnerships
- Recent Developments
- Parexel Malaysia
- Company Overview
- Service Portfolio
- Strategic Alliances/Partnerships
- Recent Developments
- ClinActis
- Company Overview
- Service Portfolio
- Strategic Alliances/Partnerships
- Recent Developments
- Subang Jaya Medical Centre (SJMC)
- Company Overview
- Service Portfolio
- Strategic Alliances/Partnerships
- Recent Developments
- Sunway Medical Centre (SunMed)
- Company Overview
- Service Portfolio
- Strategic Alliances/Partnerships
- Recent Developments
- Pantai Hospital Kuala Lumpur
- Company Overview
- Service Portfolio
- Strategic Alliances/Partnerships
- Recent Developments
- Novotech Malaysia
- Company Overview
- Service Portfolio
- Strategic Alliances/Partnerships
- Recent Developments
- PPD (Part of Thermo Fisher)
- Company Overview
- Service Portfolio
- Strategic Alliances/Partnerships
- Recent Developments
- Fortrea
- Company Overview
- Service Portfolio
- Strategic Alliances/Partnerships
- Recent Developments
- CRC Sarawak General Hospital
- Company Overview
- Service Portfolio
- Strategic Alliances/Partnerships
- Recent Developments
- Icon PLC Malaysia
- Company Overview
- Service Portfolio
- Strategic Alliances/Partnerships
- Recent Developments
- Others
- Clinical Research Malaysia (CRM)
- Contact Us and Disclaimer
Top Key Players & Market Share Outlook
- Clinical Research Malaysia (CRM)
- Institute for Clinical Research (ICR)
- IQVIA Malaysia
- SGS Malaysia
- Labcorp Drug Development
- CMIC Malaysia
- Emerald Clinical Trials
- Parexel Malaysia
- ClinActis
- Subang Jaya Medical Centre (SJMC)
- Sunway Medical Centre (SunMed)
- Pantai Hospital Kuala Lumpur
- Novotech Malaysia
- PPD (Part of Thermo Fisher)
- Fortrea
- CRC Sarawak General Hospital
- Icon PLC Malaysia
Frequently Asked Questions






