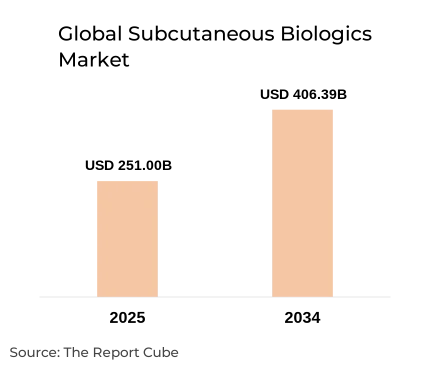
Build Your Custom Market Intelligence Report
Customize Your ReportGlobal Subcutaneous Biologics Market Insights & Analysis
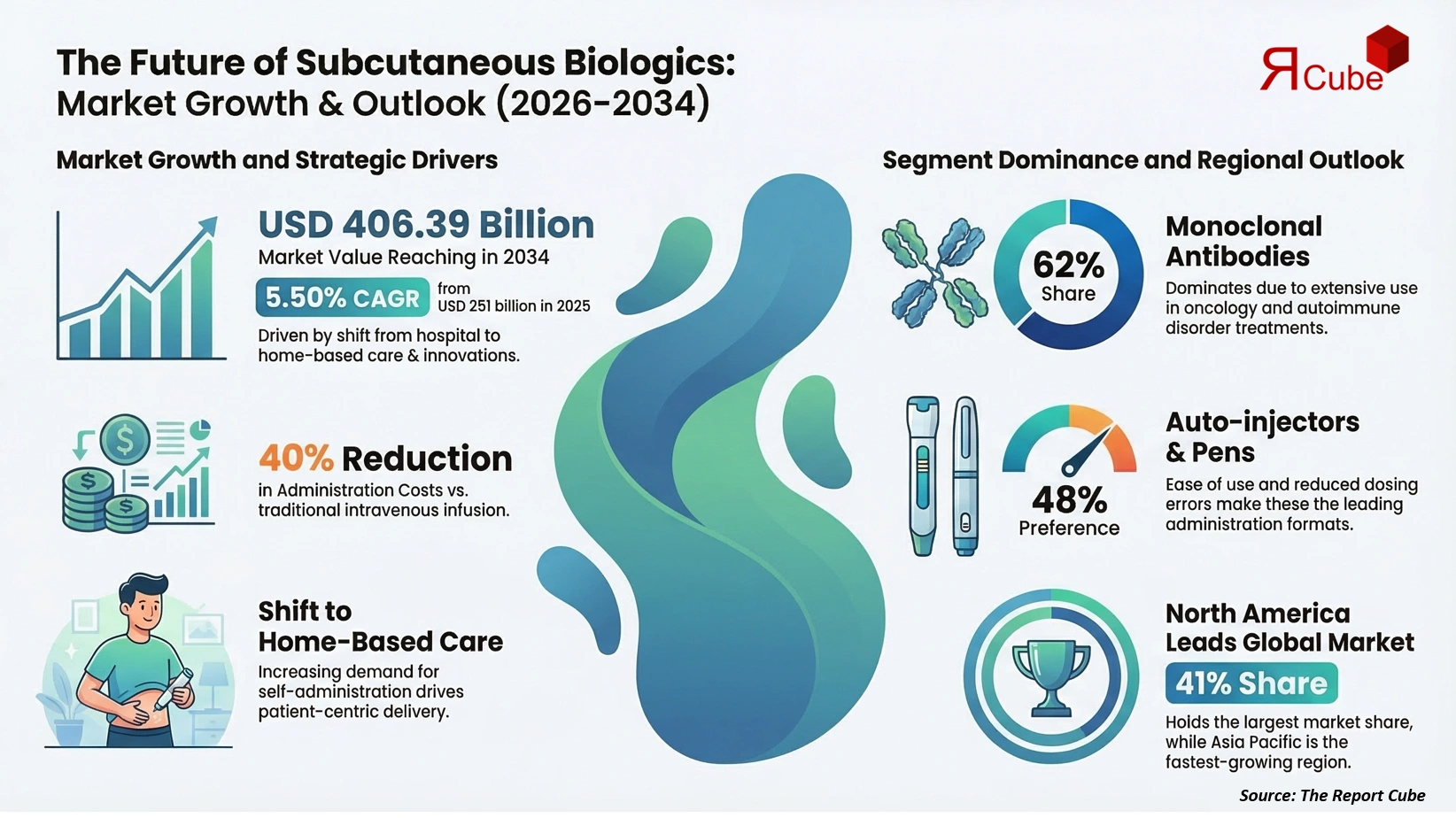
The Global Subcutaneous Biologics Market is anticipated to register a CAGR of 5.50% during 2026-2034. The market size was valued at USD 251 billion in 2025 and is projected to reach USD 406.39 billion by 2034. This strong and sustained expansion is primarily driven by the growing clinical preference for subcutaneous drug delivery over intravenous administration, particularly for chronic and long-term therapies. Subcutaneous biologics offer improved patient convenience, reduced hospital dependency, and lower overall treatment costs, making them increasingly attractive for both healthcare providers and patients. The market benefits from rising prevalence of autoimmune disorders, oncology indications, rare diseases, and metabolic conditions, where biologics have become the standard of care.
Beyond core biologic therapies, demand is also being shaped by rising adoption of home-based treatment models, expanding biosimilar pipelines, and increasing healthcare expenditure in developed and emerging economies. Supporting keywords such as injectable biologics, self-administration therapies, biologic drug delivery systems, and patient-centric drug design are increasingly reflected in clinical development strategies. Pharmaceutical companies are prioritizing lifecycle management through reformulation of existing biologics into subcutaneous formats to extend exclusivity and enhance patient adherence. Additionally, the integration of advanced delivery technologies and digital health tools continues to improve treatment outcomes, reinforcing long-term market growth momentum across major regions.
Global Subcutaneous Biologics Market Recent Developments
- 2025: Roche expanded subcutaneous formulations for oncology biologics to support faster outpatient administration.
- 2025: Amgen advanced next-generation auto-injector compatible biologics for chronic inflammatory diseases.
Government Initiatives & Policies
- U.S. FDA Patient-Focused Drug Development Program: Encourages therapies improving convenience and patient adherence.
- European Medicines Agency Biologics Framework: Supports innovation and faster access to advanced biologic therapies.
Global Subcutaneous Biologics Market Dynamics
Key Driver: Shift Toward Patient-Centric and Home-Based Care
The most influential driver of the subcutaneous biologics market is the healthcare system’s accelerating shift toward patient-centric care and home-based treatment models. Subcutaneous administration enables patients to self-administer therapies using pre-filled syringes or auto-injectors, reducing the need for frequent hospital visits. Clinical studies indicate that subcutaneous delivery can lower administration costs by up to 40% compared to intravenous infusion. This advantage is particularly critical in chronic diseases such as rheumatoid arthritis, multiple sclerosis, and inflammatory bowel disease, where long-term adherence directly impacts outcomes. As healthcare systems seek to optimize resource utilization, subcutaneous biologics continue to gain preference.
Major Challenge: Formulation Complexity and Volume Constraints
Despite strong growth prospects, formulation complexity remains a major challenge for the subcutaneous biologics market. Large-molecule biologics often require high drug concentrations, which can create issues related to viscosity, injection volume, and patient discomfort. Typically, subcutaneous injections are limited to volumes below 2 mL, constraining dose delivery for certain therapies. Additionally, maintaining molecular stability while ensuring bioavailability requires advanced formulation expertise and increases development timelines. These technical challenges can delay product launches and raise development risks, particularly for smaller biotechnology firms without extensive formulation capabilities.
Industry Trends: Expansion of Auto-Injectors and Wearable Delivery Devices
A prominent industry trend is the rapid expansion of advanced administration formats such as auto-injectors, pen injectors, and on-body wearable devices. These delivery systems improve dosing accuracy, minimize injection pain, and enhance patient confidence. Market data suggests that auto-injectors now account for over 45% of new subcutaneous biologic launches globally. Wearable injectors are gaining traction for high-volume biologics, particularly in oncology and immunology, enabling controlled delivery over extended periods. This trend aligns with broader adoption of connected drug delivery devices that support adherence monitoring and real-world data collection.
Opportunity Ahead: Biosimilars and Lifecycle Management Strategies
The growing biosimilars landscape presents a significant opportunity for the subcutaneous biologics market. As patents expire for major intravenous biologics, pharmaceutical companies are reformulating products into subcutaneous versions to retain market share. Biosimilar developers are also leveraging subcutaneous delivery to differentiate offerings and accelerate adoption. Analysts estimate that subcutaneous biosimilars could capture nearly 30% of biosimilar approvals by 2030. This strategy not only enhances patient convenience but also supports pricing flexibility and broader market access, particularly in cost-sensitive regions.
Global Subcutaneous Biologics Market Segment-wise Analysis
By Molecule Type:
- Monoclonal Antibodies
- Fusion Proteins
- Peptides and Proteins
- Nucleotides
- Other Biologic Modalities
Monoclonal antibodies dominate the market, accounting for approximately 62% of total revenue in 2025. Their dominance is driven by extensive use in oncology, immunology, and autoimmune disorders. Subcutaneous monoclonal antibodies offer comparable efficacy to intravenous formulations while significantly improving administration efficiency. The continued expansion of antibody-based pipelines and combination therapies ensures sustained leadership of this segment throughout the forecast period.
By Administration Format:
- Pre-filled Syringes
- Auto-injectors and Pen Injectors
- On-body and Wearable Injectors
- Vials
- Others
Auto-injectors and pen injectors represent the leading administration format, capturing nearly 48% of market share in 2025. Their popularity stems from ease of use, reduced dosing errors, and strong patient acceptance. These formats are increasingly adopted in self-administered chronic therapies, supporting adherence and reducing healthcare system burden. Wearable injectors, while smaller in share, are the fastest-growing sub-segment due to rising high-volume biologic approvals.
Regional Projection of Global Subcutaneous Biologics Industry
- North America
- Europe
- Asia Pacific
- Latin America
- Middle East and Africa
North America leads the global market with an estimated 41% share in 2025. Strong biologics adoption, advanced healthcare infrastructure, and high patient awareness support regional dominance. The United States remains the largest contributor due to rapid uptake of self-injection therapies and favorable reimbursement frameworks. Europe follows closely, while Asia Pacific is emerging as the fastest-growing region, driven by expanding biologics access, rising chronic disease prevalence, and increasing investment in specialty pharmaceuticals.
Why Choose This Report?
- Provides a comprehensive overview of the overall market analysis, encompassing key trends, consumer behavior analysis, and risk assessment to support strategic decision-making.
- Provides accurate, up-to-date insights into market size, segmentation, and emerging opportunities, helping to minimize risk & capitalizing on growth.
- Gives deep understanding of target audience preferences, investment habits, and communication channels for enhanced product development & marketing effectiveness.
- Delivers competitive analysis & benchmarking, uncovering the strengths & weaknesses of market competitors to guide strategies.
- Consolidate comprehensive market intelligence, reducing reasoning & streamlining research efforts.
- Facilitates customized market segmentation & risk mitigation strategies, fine-tuned to the business objectives.
- Aids in identifying both market challenges & untapped opportunities within the industry to drive long-term business growth.
- Provides valuable information based on actual customer data & search trends.
Table of Contents
- Introduction
- Objective of the Study
- Product and Category Definition
- Market Segmentation
- Study Variables
- Research Methodology
- Secondary Data Points
- Breakdown of Secondary Sources
- Primary Data Points
- Breakdown of Primary Interviews
- Secondary Data Points
- Executive Summary
- Market Dynamics
- Drivers
- Challenges
- Opportunity Assessment
- Recent Trends and Developments
- Regulatory and Policy Landscape
- Global Subcutaneous Biologics Market Overview (2020-2034)
- Market Size, By Value (USD Billion)
- Market Share, By Molecule Type
- Monoclonal Antibodies
- Fusion Proteins
- Peptides and Proteins
- Nucleotides
- Other Biologic Modalities
- Market Share, By Administration Format
- Pre-filled Syringes
- Auto-injectors and Pen Injectors
- On-body and Wearable Injectors
- Vials
- Others
- Market Share, By Therapeutic Area
- Autoimmune Disorders
- Inflammatory Disorders
- Oncology
- Metabolic Disorders
- Hematology
- Neurology
- Infectious Diseases
- Others
- Market Share, By Setting of Care
- Home Care
- Self-administration
- Outpatient Clinics
- Ambulatory Clinics
- Hospital
- Market Share, By Distribution Channel
- Hospital Pharmacies
- Retail Pharmacies
- Specialty Pharmacies
- Online Pharmacies
- Market Share, By Region
- North America
- Europe
- Asia Pacific
- Latin America
- Middle East and Africa
- Market Share, By Company
- Revenue Shares
- Competitive Landscape
- North America Subcutaneous Biologics Market
- Market Size, By Value (USD Billion)
- Market Share, By Molecule Type
- Market Share, By Administration Format
- Market Share, By Therapeutic Area
- Market Share, By Setting of Care
- Market Share, By Distribution Channel
- Market Share, By Region/Country Group
- Market Share, By Country
- United States
- Canada
- Mexico
- Europe Subcutaneous Biologics Market
- Market Size, By Value (USD Billion)
- Market Share, By Molecule Type
- Market Share, By Administration Format
- Market Share, By Therapeutic Area
- Market Share, By Setting of Care
- Market Share, By Distribution Channel
- Market Share, By Region/Country Group
- Market Share, By Country
- Germany
- France
- United Kingdom
- Italy
- Spain
- Northern and Nordic Countries
- Rest of Europe
- Asia Pacific Subcutaneous Biologics Market
- Market Size, By Value (USD Billion)
- Market Share, By Molecule Type
- Market Share, By Administration Format
- Market Share, By Therapeutic Area
- Market Share, By Setting of Care
- Market Share, By Distribution Channel
- Market Share, By Region/Country Group
- Market Share, By Country
- China
- Japan
- India
- South Korea
- Australia
- Rest of Asia Pacific
- Latin America Subcutaneous Biologics Market
- Market Size, By Value (USD Billion)
- Market Share, By Molecule Type
- Market Share, By Administration Format
- Market Share, By Therapeutic Area
- Market Share, By Setting of Care
- Market Share, By Distribution Channel
- Market Share, By Region/Country Group
- Market Share, By Country
- Brazil
- Mexico
- Argentina
- Rest of Latin America
- Middle East and Africa Subcutaneous Biologics Market
- Market Size, By Value (USD Billion)
- Market Share, By Molecule Type
- Market Share, By Administration Format
- Market Share, By Therapeutic Area
- Market Share, By Setting of Care
- Market Share, By Distribution Channel
- Market Share, By Region/Country Group
- Market Share, By Country
- Turkey
- Saudi Arabia
- United Arab Emirates
- South Africa
- Rest of Middle East and Africa
- Competitive Outlook and Company Profiles
- AbbVie
- Company Overview
- Product Portfolio
- Strategic Alliances/Partnerships
- Recent Developments
- Roche
- Company Overview
- Product Portfolio
- Strategic Alliances/Partnerships
- Recent Developments
- Amgen
- Company Overview
- Product Portfolio
- Strategic Alliances/Partnerships
- Recent Developments
- Novartis
- Company Overview
- Product Portfolio
- Strategic Alliances/Partnerships
- Recent Developments
- Pfizer
- Company Overview
- Product Portfolio
- Strategic Alliances/Partnerships
- Recent Developments
- Bristol Myers Squibb
- Company Overview
- Product Portfolio
- Strategic Alliances/Partnerships
- Recent Developments
- Takeda Pharmaceutical Company
- Company Overview
- Product Portfolio
- Strategic Alliances/Partnerships
- Recent Developments
- Biogen
- Company Overview
- Product Portfolio
- Strategic Alliances/Partnerships
- Recent Developments
- Eisai
- Company Overview
- Product Portfolio
- Strategic Alliances/Partnerships
- Recent Developments
- Alnylam Pharmaceuticals
- Company Overview
- Product Portfolio
- Strategic Alliances/Partnerships
- Recent Developments
- Others
- AbbVie
- Contact Us and Disclaimer
Top Key Players & Market Share Outlook
- AbbVie
- Roche
- Amgen
- Novartis
- Pfizer
- Bristol Myers Squibb
- Takeda Pharmaceutical Company
- Biogen
- Eisai
- Alnylam Pharmaceuticals
- Others
Frequently Asked Questions






